Neuralgic amyotrophy (NA) or Parsonage Turner syndrome was characterized in more than 130 patients in the mid-20th century and named “shoulder-girdle syndrome”.1 It can impact any nerve of the brachial plexus and its recurrence has been described in approximately 25% of patients at 6 years of follow-up.2 Phrenic nerve afference has numerous variations but stems from C3, C4 and C5 ventral roots and thus depends on both the brachial and cervical plexus. Therefore though not typical in NA, phrenic nerve involvement and resulting diaphragm paralysis has been described in 6–14% of series.2
Our patient is a 47-year-old male with past medical history of hemorrhagic rectocolitis (HR) diagnosed 15 years ago and grade 1 obesity as well as smoking history of 10 pack-years. There was no family history of NA. His usual treatment was mesalamine 800mg twice daily.
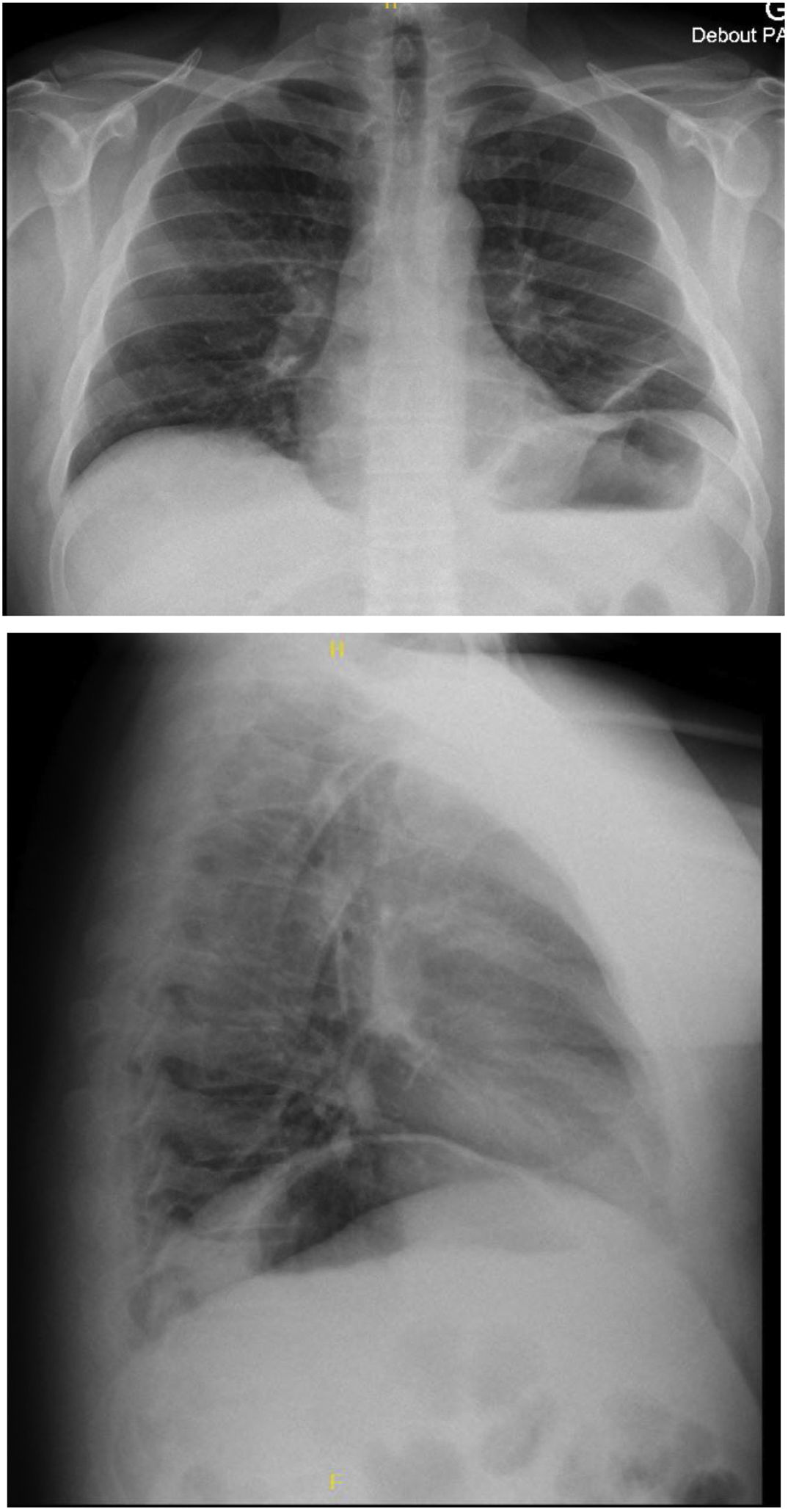
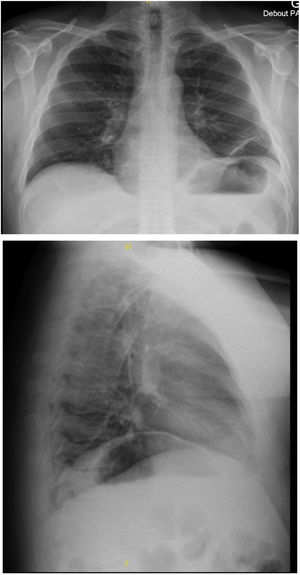
He was diagnosed with NA in 2008 following an exacerbation of hemorrhagic rectocolitis. Ten years later, he experienced a reoccurrence of acute interscapular pain associated with orthopnea. There was no trigger event for this recurrent episode of NA nor infectious nor acute episode of HR. Chest X-ray (Fig. 1) and CT-scan revealed ascension of both hemidiaphragms. The results of the pulmonary function test (PFT) showed restrictive pulmonary syndrome, and supine variation of vital capacity of 64% confirmed severe diaphragm dysfunction. The laboratory results and blood gases were normal in the prone position (Table 1). The patient was thus diagnosed with recurrent NA with bilateral phrenic nerve damage. Treatment included analgesics, corticosteroids and non-invasive ventilation (NIV).
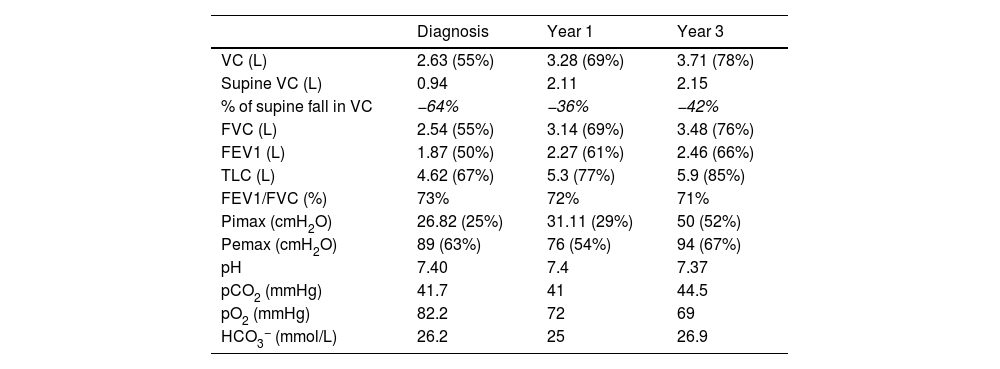
Pulmonary function test and blood gases.
| Diagnosis | Year 1 | Year 3 | |
|---|---|---|---|
| VC (L) | 2.63 (55%) | 3.28 (69%) | 3.71 (78%) |
| Supine VC (L) | 0.94 | 2.11 | 2.15 |
| % of supine fall in VC | −64% | −36% | −42% |
| FVC (L) | 2.54 (55%) | 3.14 (69%) | 3.48 (76%) |
| FEV1 (L) | 1.87 (50%) | 2.27 (61%) | 2.46 (66%) |
| TLC (L) | 4.62 (67%) | 5.3 (77%) | 5.9 (85%) |
| FEV1/FVC (%) | 73% | 72% | 71% |
| Pimax (cmH2O) | 26.82 (25%) | 31.11 (29%) | 50 (52%) |
| Pemax (cmH2O) | 89 (63%) | 76 (54%) | 94 (67%) |
| pH | 7.40 | 7.4 | 7.37 |
| pCO2 (mmHg) | 41.7 | 41 | 44.5 |
| pO2 (mmHg) | 82.2 | 72 | 69 |
| HCO3− (mmol/L) | 26.2 | 25 | 26.9 |
FEV1, forced expiratory volume in the first second; FVC, forced vital capacity; Pimax, maximal inspiratory pressure; Pemax, maximal expiratory pressure; TLC, total lung capacity; VC, vital capacity.
At 3 years of follow-up, there was no neurologic sequelae and improvements were seen on electromyography and PFT (Table 1). However, nocturnal NIV dependency remained necessary with mean daily use of 7h20 even though orthopnea has partially improved. We identified no alternative etiologies for chronic respiratory failure – particularly no active smoking and stable BMI.
The physiopathology of NA is poorly understood but some data suggest a possible association with autoimmune reaction. Current literature distinguishes between hereditary and idiopathic NA.2 Our case is the third reported case associating inflammatory bowel disease (IBD) and NA. This case is the only one in which the phrenic nerve is affected and the first one in which the underlying condition was HR.3,4
Besides the rare association with IBD, our case report shows unusual features with recurrence of NA, severe bilateral diaphragmatic dysfunction and improved PFT despite NIV dependency at 3 years. Previous studies have reported that improvement generally occurs within 3 years but that improved clinical symptoms do not necessarily mean improved paraclinical tests.5 Our case illustrates that better PFT results are not always accompanied by clinical improvement and discontinuation of NIV.
In this case report describing a novel association of NA and hemorrhagic rectocolitis, our patient presented with bilateral diaphragmatic paralysis requiring NIV dependency that persisted at 3 years of follow-up. It is worth reporting since NA is often underdiagnosed and this very particular presentation with discrepancy of NIV dependency and PFT-electromyography improvement.
Data sharingData and material are available upon request at University of Burgundy, CHU de Dijon, Dijon, France.
Informed consentThe patient was informed and had the opportunity to decline study participation.
FundingThis research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Conflict of interestsAll authors declare no competing interests.