The main purpose of staging in non-small cell lung cancer (NSCLC) is to assess mediastinal lymph node involvement, with thoracic CT being the main non-invasive test for this. However, given that up to 15% of patients who show no mediastinal lymph node involvement in the CT has lymph node metastasis during surgery, other examinations are required. Endoscopic ultrasonography guided fine-needle aspiration (EUS-FNA) was shown to be able to detect advanced disease (metastatic mediastinal lymph nodes, adrenal metastasis, mediastinal invasion by the tumor) in approximately 25% of patients with a CT that suggested a non-advanced disease.
Another situation in which CT has a very limited value is in the evaluation of the response to induction therapy, with its most limiting factor being its intrinsic inability to distinguish between a tumor and necrosis. In this context, EUS-FNA was shown to have a good performance, with a sensitivity, negative predictive value and precision of 75%, 67%, and 83%, respectively.
In conclusion, EUS-FNA may be considered a good alternative in the pre-operative staging of patients with NSCLC, with and without diseased mediastinal lymph nodes in CT, and could play an important role in the mediastinal re-staging of these patients by identifying a patient sub-group who might benefit from additional surgical treatment.
El principal objetivo de la estadificación en el cáncer de pulmón de células no pequeñas (CPCNP) es la evaluación de la afectación ganglionar mediastínica, y la TC torácica es la principal prueba no invasiva para la evaluación. Sin embargo, dado que hasta un 15% de los pacientes sin afectación ganglionar del mediastino en la TC tienen metástasis ganglionares en la cirugía, son necesarias otras exploraciones. La punción aspirativa con aguja fina guiada por ultrasonografía endoscópica (USE-PAAF) ha demostrado ser capaz de detectar enfermedad avanzada (ganglios mediastínicos metastásicos, metástasis en la suprarrenal, invasión mediastínica por el tumor) en aproximadamente el 25% de los pacientes con TC que sugiere enfermedad no avanzada.
Otra situación en la que la TC tiene un valor muy limitado es en la evaluación de la respuesta a la terapia de inducción, siendo su incapacidad intrínseca para distinguir entre tumor y necrosis el factor más limitante. En este contexto la USE-PAAF ha demostrado tener un buen rendimiento con una sensibilidad, valor predictivo negativo y precisión del 75, 67 y 83% respectivamente.
En consecuencia, la USE-PAAF puede ser considerada una buena alternativa en la estadificación preoperatoria de los pacientes con CPCNP con y sin ganglios mediastínicos patológicos en la TC y podría desempeñar un papel importante en la reestadificación mediastínica de estos pacientes mediante la identificación de un subgrupo de pacientes que se beneficiarían de tratamiento quirúrgico adicional.
The main objective of staging in non-small-cell lung cancer (NSCLC) is mediastinal evaluation. Precise mediastinal evaluation is fundamental for determining the prognosis of and treatment for these patients, as the presence of mediastinal lymph node involvement means stage IIIA or IIIB disease.1–3 This translates into the need for neoadjuvant treatment, which may or may not be followed by surgery. Given that 30%–40% of patients with newly diagnosed NSCLC have mediastinal involvement, complementary tests are necessary to evaluate the mediastinum.
There are many options for staging the mediastinum. Thoracic CT is the main diagnostic test for the mediastinal evaluation of bronchogenic carcinomas. The diagnostic criteria for malignancy are the presence of lymphadenopathies whose smallest diameter is larger than 1cm. According to different studies in the literature, the precision of CT for identifying pathologic mediastinal lymphadenopathies is variable (57%–95%).4–6 This precision varies according to the lymph node stations, the highest being for the right paratracheal region and the lowest for the subcarinal region.7 Up to 15% of patients who do not have lymphadenopathies greater than 1cm on CT present mediastinal metastasis in surgery.8–10
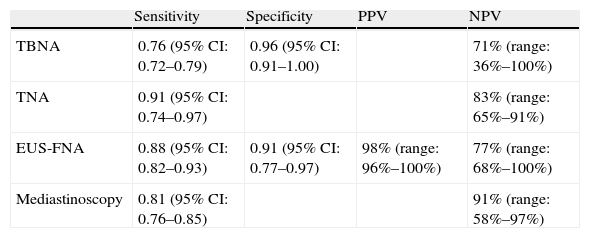
Given that the presence or absence of metastatic mediastinal lymphadenopathies influences decision-making regarding tumor resectability, CT cannot be used alone for mediastinal staging and histologic confirmation of the lymphadenopathies is imperative. Endoscopic ultrasound-guided fine needle aspiration (EUS-FNA) biopsy has been demonstrated to be highly precise for the evaluation of the lymph node stations located in the mid and posterior mediastinum (regions 4L, 5, 7, 8, and 9, and occasionally 2 and 4R), as well as in the left suprarenal gland, while the peribronchial and hilar regions and the anterior mediastinum are not accessible by EUS-FNA, but are instead accessible by needle aspiration (NA) guided by endobronchial ultrasound (EBUS).11,12 In a systematic review and a meta-analysis of 18 studies that included 1201 patients published by Micames et al.,13 EUS-FNA reached a sensitivity of 83% and a specificity of 97% for mediastinal staging (N2/N3) in patients with lung cancer. The main advantage of EUS-FNA over other non-invasive methods is its capacity to obtain histologic confirmation of the presence of disease. Compared with minimally invasive surgical techniques like mediastinoscopy, EUS-FNA (like NA guided by EBUS) only requires conscious sedation, can be done routinely in an outpatient setting and has been demonstrated to be more cost-efficient.14–17 The effectiveness of the different techniques used for staging NSCLC is detailed in Table 1.
Performance of the Different Invasive Techniques in Patients With NSCLC; Review of the Scientific Evidence Published.
| Sensitivity | Specificity | PPV | NPV | |
| TBNA | 0.76 (95% CI: 0.72–0.79) | 0.96 (95% CI: 0.91–1.00) | 71% (range: 36%–100%) | |
| TNA | 0.91 (95% CI: 0.74–0.97) | 83% (range: 65%–91%) | ||
| EUS-FNA | 0.88 (95% CI: 0.82–0.93) | 0.91 (95% CI: 0.77–0.97) | 98% (range: 96%–100%) | 77% (range: 68%–100%) |
| Mediastinoscopy | 0.81 (95% CI: 0.76–0.85) | 91% (range: 58%–97%) |
TNA: transthoracic needle aspiration; TBNA: transbronchial needle aspiration; EUS-FNA: endoscopic ultrasound-guided fine-needle aspiration; NPV: negative predictive value; PPV: positive predictive value.
There are two clinical situations in patients with NSCLC that pose a special diagnostic problem in which EUS-FNA is a good alternative and offers a new perspective: the evaluation of patients with negative mediastinum on CT, and re-staging after neoadjuvant treatment.
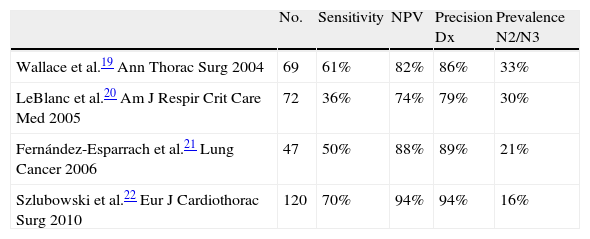
EUS-FNA in Lung Cancer in Patients With Normal Mediastinum on CTThe management of patients with NSCLC without signs of mediastinal affectation on CT is still open for discussion. EUS can detect mediastinal nodules of up to 2–3mm.18 Therefore, EUS is able to detect adenopathies smaller than 1cm considered irrelevant by CT and even obtain a sample from them to either confirm or rule out the affectation of the lymph nodes. Four prospective studies have evaluated EUS-FNA in patients with NSCLC without pathological lymph nodes on CT, showing that EUS-FNA is able to detect advanced disease (mediastinal metastasis, suprarenal metastasis, tumor invasion of the mediastinum) in approximately 25% of the patients whose CT showed only non-advanced disease19–22 (Table 2). Wallace et al.19 published a study in which EUS-FNA identified N2–N3 lymph node metastasis in 14 out of 67 patients (21%). There were also 9 false negatives due to either a sampling error (n=5) or to the presence of metastasis in pre- and paratracheal lymph nodes that were not visualized by EUS (n=4). In another study, LeBlanc et al.20 identified N3 metastasis in 5 out of 67 patients (7%) by means of EUS-FNA. EUS avoided surgery in 9 patients (13%), not only due to the N3 affectation (n=5) but also by detecting celiac metastatic lymphadenopathies (n=2), tumor invasion (n=1) and synchronic esophageal cancer (n=1). In another 9 patients, EUS had an impact in management by detecting suprarenal benign lesions (n=8) and a hepatic lesion (n=1). In total, the findings of EUS changed the patient management in 25% of cases. In the surgical staging of the remaining 62 patients in whom mediastinal disease was not diagnosed, lymph node metastasis was found in 17 more patients.
Diagnostic Performance of EUS-FNA in N2/N3 Patients With Normal CT.
| No. | Sensitivity | NPV | Precision Dx | Prevalence N2/N3 | |
| Wallace et al.19 Ann Thorac Surg 2004 | 69 | 61% | 82% | 86% | 33% |
| LeBlanc et al.20 Am J Respir Crit Care Med 2005 | 72 | 36% | 74% | 79% | 30% |
| Fernández-Esparrach et al.21 Lung Cancer 2006 | 47 | 50% | 88% | 89% | 21% |
| Szlubowski et al.22 Eur J Cardiothorac Surg 2010 | 120 | 70% | 94% | 94% | 16% |
The prevalence of mediastinal metastasis in these two studies (35% and 36%, respectively) is higher than that of other surgical series, which suggests a bias in favor of the EUS either due to the inclusion of patients with lymphadenopathies that are “borderline” in size or because more samples were taken than normal (normally 4 or 5 aspirations per lesion in the study by Wallace et al.). In the study by Fernández-Esparrach et al.,21 EUS-FNA demonstrated metastasis in N2–3 lymphadenopathies in 5 out of 47 patients (11%). There were 5 false negatives that were due to micrometastasis (n=1), the lack of need to perform FNA due to benign morphological appearance (n=3) or to the presence of metastasis in a lymphadenopathy that was not visible by EUS (n=1). In this study, the EUS-FNA was done when one or more of the classic ultrasound criteria of malignancy by EUS were present (hypoechogenicity, round shape, well-defined edges, size>5mm).23,24 Consequently, three metastatic lymphadenopathies (in two patients) visualized by EUS were not considered tributaries of EUS-FNA.
Therefore, in the mediastinal staging of NSCLC, the use of classic criteria for malignancy should be reconsidered.25 The reasons for the low sensitivity of these criteria have not been researched specifically. However, given that it is common to find inflammatory mediastinal lymphadenopathies in healthy patients, one might think that the infiltration of the lymphadenopathies by metastatic cells would not necessarily change the morphologic characteristics of the preexisting lymphadenopathies. In this way, any mediastinal lymphadenopathy would be a candidate for being aspirated in patients with NSCLC. Finally, it is well known that mediastinal lymphadenopathies can contain foci of micrometastasis in patients with NSCLC. Therefore, a more exhaustive analysis of all the visible mediastinal lymph nodes and the addition of NA guided by EBUS for exploring the regions that are not accessible by EUS would improve the diagnostic performance of EUS-FNA in these patients.22
Mediastinal Re-Staging After Neoadjuvant TherapyMediastinal re-staging in patients with NSCLC after induction of chemotherapy is a controversial point. CT and MRI have limited value due to the intrinsic inability of these techniques to differentiate between tumors and necrosis, and therefore their precision is low.26 On the other hand, PET is not a good predictor for the response to chemotherapy, either by the primary tumor or by the lymphadenopathies.27–29 The results of a recent systematic review advise against the use of PET for mediastinal lymph node re-staging as its effectiveness is not very good (sensitivity 63.8% [95% CI: 53.3%–73.7%] and specificity 85.3% [95% CI: 80.4%–89.4%]). As for the response of the primary tumor, the results are very variable, and the sensitivity, specificity, positive predictive value (PPV) and negative predictive value (NPV) were 80%–100%, 0%–100%; 42.9%–100% and 66.7%–100%, respectively.29
Recent studies have demonstrated that NSCLC patients with metastases in ipsilateral lymphadenopathies that were understaged with induction of chemotherapy had significantly higher survivals after surgical resection compared with those patients with persistent N2 disease.30–33 The three-year survival was 59% in the patients of the first group and 0% among the patients with persistent N2 disease. Therefore, a precise evaluation of the response to neoadjuvant therapy is necessary to decide the therapeutic strategy depending on prognosis.
Mediastinoscopy is still a valid tool in the re-staging of locally advanced NSCLC after neoadjuvant treatment, with a diagnostic precision of 85%.26,34–37 Mediastinoscopy provides histological evidence of the response to treatment and selects those patients who will benefit from a later thoracotomy. However, the frequency of incomplete procedures is high (40%) due to the technical difficulties secondary to the presence of mediastinal fibrosis.38
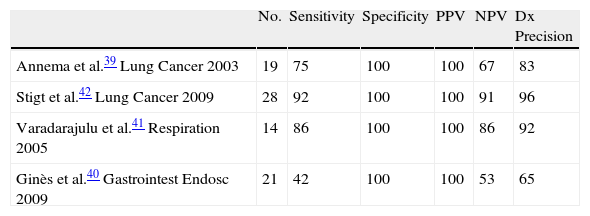
Annema et al.39 published the first case study with EUS-FNA for mediastinal re-staging in 19 patients with N2 disease who had been treated with induction of chemotherapy and obtained a sensitivity, NPV and diagnostic precision of 75%, 67%, and 83%, respectively. There were three false negatives (sample errors in two cases and a paraesophageal tumor not detected by EUS in one case). These results are lower than those obtained with EUS-FNA in the patients who have not been treated with chemotherapy, but they are very similar to those published by other authors39–42 (Table 3). In the study by Ginès et al.,40 the sensitivity and NPV values are the lowest published to date and could be explained by the location of some lymphadenopathies in stations that are more difficult to visualize by EUS, as is the case of station 4R.
Other authors have compared CT-PET with EUS-FNA. In the study by Stigt et al.42 in 28 patients, EUS-FNA had an NPV and a diagnostic precision of 92%. The agreement between the findings of the re-staging by EUS-FNA and the metabolic response of the metastatic lymphadenopathies occurred in 17 out of 27 patients. However, and due to the fact that the histologic confirmation is higher and therefore necessary, EUS-FNA is the first-choice procedure for mediastinal re-staging.
The lymphadenopathies of the right paratracheal region cannot be correctly visualized by EUS. This part of the mediastinum could be explored by EBUS.43 A recent study of 124 patients treated with induction of chemotherapy for stage IIIA44 reached a sensitivity of 76% for NA guided by EBUS. However, due to the low NPV (20%), the authors suggest that the negative findings should be confirmed by means of surgical staging prior to thoracotomy.
Some of the reasons of this low NPV of NA guided by EBUS are the following:
- 1.
After chemotherapy, the lymphadenopathies that initially contain tumors present necrosis and fibrosis and are more difficult to biopsy, while the quantity of cell material for analysis is less.
- 2.
The malignant cells can be found focally inside the lymphadenopathy in the interior of dense areas of extracellular matrix.
- 3.
The presence of necrosis within the aspirated sample often makes the pathological evaluation more difficult.
- 4.
There was no in situ evaluation by the cytologist, which possibly would have been able to increase the sensitivity for the detection of malignancy.
It is important to remember the different territories that are usually accessible by EUS and EBUS. EBUS enables access to the paratracheal (stations 2, 3p, and 4), subcarinal (station 7), hilar (station 10) and interlobar (station 11) regions. By means of EUS, we can visualize the regions of the posterior-inferior mediastinum (stations 4L, 5, 7, 8, 9, and occasionally 2 and 4R).11,12 Given the fact that these techniques are complementary, the combination of both could improve their precision. Furthermore, it has been demonstrated that the sequential performance of EBUS and EUS is well tolerated by the patients when done under conscious sedation.45
In conclusion, EUS-FNA should be considered in the mediastinal staging of patients with NSCLC with and without pathologic adenopathies by CT and in the mediastinal re-staging after neoadjuvant treatment.
Conflict of InterestsThe authors declare having no conflict of interests.
Please cite this article as: Fernández-Esparrach G, et al. Nuevas aplicaciones de la ultrasonografía endoscópica (USE) en el cáncer de pulmón: evaluación de pacientes con mediastino negativo por TC y reestadificación después de tratamiento neoadyuvante. Arch Bronconeumol. 2011;47:410–4.