COVID-19 pneumonia can result in acute hypoxemic respiratory failure (hARF) with the need of positive end-expiratory pressure (PEEP).1 The application of a continuous positive airway pressure (CPAP) through a helmet improves oxygenation and reduces the risk of intubation compared to oxygen therapy in patients with severe hARF due to pneumonia.2,3 The identification of the best PEEP for each individual COVID-19 patient undergoing helmet CPAP treatment is helpful to optimize lung recruitment and avoid complications related to over-distention.4 We designed a feasibility study to evaluate the physiological effect in terms of lung recruitability of the application of different PEEP values in patients with hARF due to COVID-19 pneumonia undergoing helmet CPAP.
Consecutive adults (≥18 years) with hARF and with an indication for helmet CPAP treatment were enrolled in a pilot, feasibility study conducted at the COVID-19 high-dependency unit (HDU) of the Policlinico Hospital in Milan, Italy, between 19th March and 16th April 2020. Indications for helmet CPAP treatment included all the following: a diagnosis of pneumonia as the only cause of hARF and a partial arterial pressure of oxygen to fraction of inspired oxygen (PaO2:FiO2) ratio <300. The presence of other causes of hARF were excluded through clinical evaluations. Patients with at least one of the following criteria were excluded: need for immediate intubation, Glasgow Coma Scale <15, respiratory acidosis, and systolic blood pressure (SBP) <90mmHg despite fluid resuscitation and/or use of vasopressors, swallowing disturbance with increasing risk of aspiration pneumonia, and inability to protect the airway. The Ethical Committees of the Policlinico hospital approved the study (No. 345_2020). All the patients underwent a standardized and non-invasive lung recruitability test (LRT) through the evaluation of vitals and blood gas analysis parameters every 30minutes at different PEEP values (0–baseline-, 2.5, 5, 7.5, 10, 12.5, and 15cmH2O). Flow in the CPAP circuit was at least 80L/min for each value of PEEP and FiO2 delivered did not change during the trial. The primary endpoint was the rate of success and partial success of the LRT. Successful LTR was defined as the occurrence of all the following: (1) A decrease of the alveolar-arterial gradient (A-aO2) of at least 20% compared to baseline; (2) Equal or reduced respiratory rate (RR) compared to baseline; (3) Absence of hemodynamic instability; (4) Equal or increase SpO2 values compared to baseline; (5) Absence of patient's discomfort. Partial success of LTR was defined by all the criteria mentioned above but decrease of A-aO2 less than 20%. Failure of LRT was defined as the occurrence of at least one of the following before reaching success criteria: (1) An increase in RR compared to baseline; (2) An increase of A-aO2 compared to baseline; (3) hemodynamic instability; (4) SpO2 <90%; (5) Respiratory distress; and (6) Patient's discomfort. No specific computations were carried out.
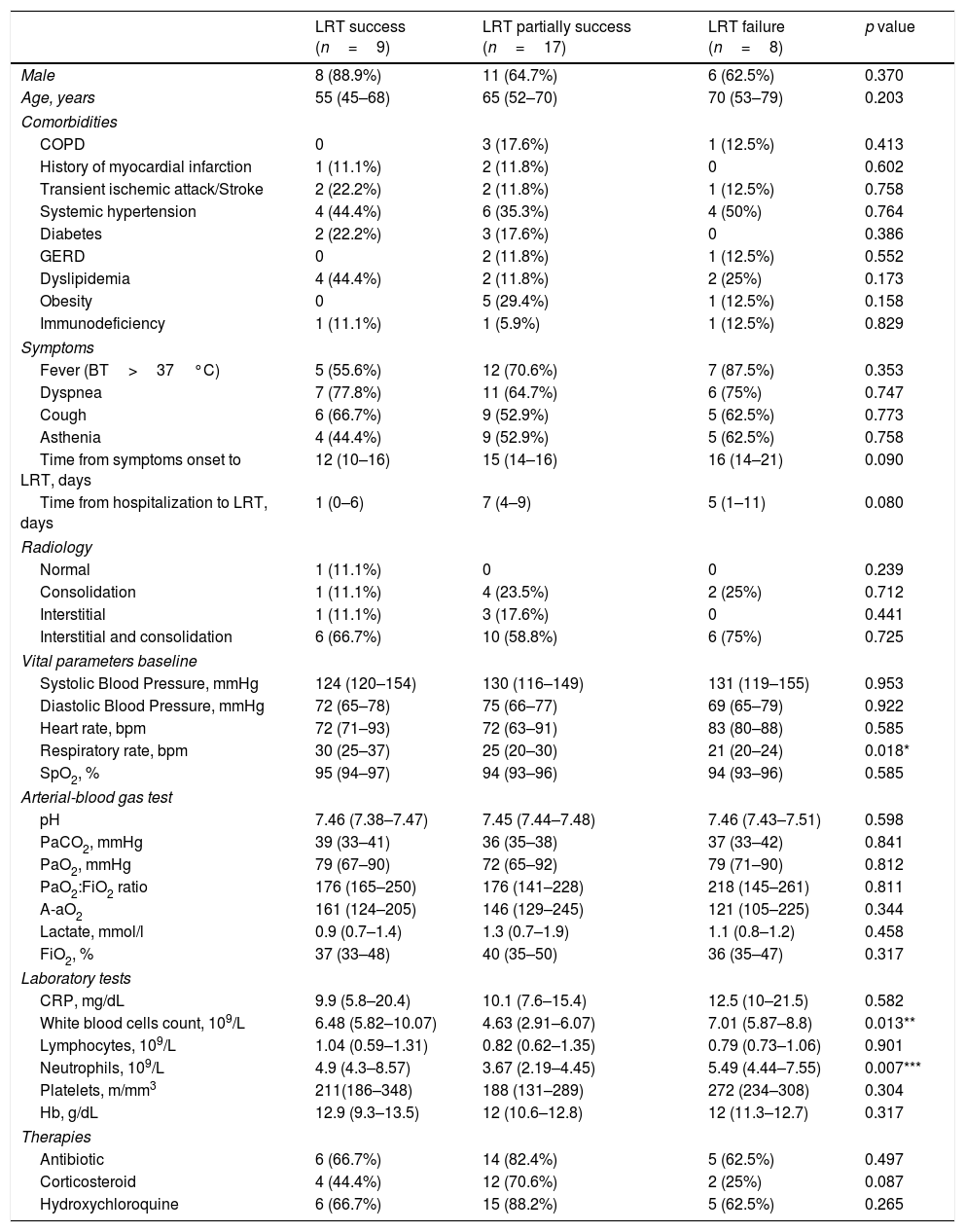
During the study period 87 patients were treated in COVID-19 HDU. 61 (71%) patients were treated with helmet CPAP, 15 (17%) patients with venturi mask oxygenation and 11 (13%) patients with high flow nasal cannulae oxygenation. Among the 34 patients [71% male; median (IQR) age: 63 (50-72) years] enrolled, the most prevalent comorbidities were systemic hypertension (42%), dyslipidemia (24%) and obesity (18%). All patients had hARF due to COVID-19 pneumonia and underwent helmet CPAP with a median (IQR) PaO2:FiO2 ratio of 177 (144–242) and A-aO2 of 146 (121–234). LRT was successful in 9 (26.5%) patients: 1 at 5cmH2O, 1 at 7.5cmH2O, 5 at 10cmH2O, and 1 at 12.5cmH2O. Partial success of LRT occurred in 17 (50%) patients: 6 at 5cmH2O, 4 at 7.5cmH2O, 4 at 10cmH2O, 1 at 12.5cmH2O and 2 at 15cmH2O. LTR failed in 8 (23.5%) patients because of the occurrence of hemodynamic instability (n=5, 14.7%), respiratory distress (n=1, 2.9%), and increase in RR (n=2, 5.9%). Baseline characteristics of the three groups are summarized in Table 1. There was a statistical significant difference in RR among the 3 groups (p=0.018), in particular patients with a successful LTR showed higher baseline RR compared to LRT failure patients (30 [25–37] versus 21 [20–24] breaths per minute, p=0.004). In patients with a successful LTR the median (IQR) decrease of A-aO2 was 26% (22-35%), while in patients with a partial success LTR the median decrease of A-aO2 was 10% (8–13%).
Baseline characteristics of the study population (data are presented as n [%] or medians [IQR: 25th–75th interquartile range]).
| LRT success (n=9) | LRT partially success (n=17) | LRT failure (n=8) | p value | |
|---|---|---|---|---|
| Male | 8 (88.9%) | 11 (64.7%) | 6 (62.5%) | 0.370 |
| Age, years | 55 (45–68) | 65 (52–70) | 70 (53–79) | 0.203 |
| Comorbidities | ||||
| COPD | 0 | 3 (17.6%) | 1 (12.5%) | 0.413 |
| History of myocardial infarction | 1 (11.1%) | 2 (11.8%) | 0 | 0.602 |
| Transient ischemic attack/Stroke | 2 (22.2%) | 2 (11.8%) | 1 (12.5%) | 0.758 |
| Systemic hypertension | 4 (44.4%) | 6 (35.3%) | 4 (50%) | 0.764 |
| Diabetes | 2 (22.2%) | 3 (17.6%) | 0 | 0.386 |
| GERD | 0 | 2 (11.8%) | 1 (12.5%) | 0.552 |
| Dyslipidemia | 4 (44.4%) | 2 (11.8%) | 2 (25%) | 0.173 |
| Obesity | 0 | 5 (29.4%) | 1 (12.5%) | 0.158 |
| Immunodeficiency | 1 (11.1%) | 1 (5.9%) | 1 (12.5%) | 0.829 |
| Symptoms | ||||
| Fever (BT>37°C) | 5 (55.6%) | 12 (70.6%) | 7 (87.5%) | 0.353 |
| Dyspnea | 7 (77.8%) | 11 (64.7%) | 6 (75%) | 0.747 |
| Cough | 6 (66.7%) | 9 (52.9%) | 5 (62.5%) | 0.773 |
| Asthenia | 4 (44.4%) | 9 (52.9%) | 5 (62.5%) | 0.758 |
| Time from symptoms onset to LRT, days | 12 (10–16) | 15 (14–16) | 16 (14–21) | 0.090 |
| Time from hospitalization to LRT, days | 1 (0–6) | 7 (4–9) | 5 (1–11) | 0.080 |
| Radiology | ||||
| Normal | 1 (11.1%) | 0 | 0 | 0.239 |
| Consolidation | 1 (11.1%) | 4 (23.5%) | 2 (25%) | 0.712 |
| Interstitial | 1 (11.1%) | 3 (17.6%) | 0 | 0.441 |
| Interstitial and consolidation | 6 (66.7%) | 10 (58.8%) | 6 (75%) | 0.725 |
| Vital parameters baseline | ||||
| Systolic Blood Pressure, mmHg | 124 (120–154) | 130 (116–149) | 131 (119–155) | 0.953 |
| Diastolic Blood Pressure, mmHg | 72 (65–78) | 75 (66–77) | 69 (65–79) | 0.922 |
| Heart rate, bpm | 72 (71–93) | 72 (63–91) | 83 (80–88) | 0.585 |
| Respiratory rate, bpm | 30 (25–37) | 25 (20–30) | 21 (20–24) | 0.018* |
| SpO2, % | 95 (94–97) | 94 (93–96) | 94 (93–96) | 0.585 |
| Arterial-blood gas test | ||||
| pH | 7.46 (7.38–7.47) | 7.45 (7.44–7.48) | 7.46 (7.43–7.51) | 0.598 |
| PaCO2, mmHg | 39 (33–41) | 36 (35–38) | 37 (33–42) | 0.841 |
| PaO2, mmHg | 79 (67–90) | 72 (65–92) | 79 (71–90) | 0.812 |
| PaO2:FiO2 ratio | 176 (165–250) | 176 (141–228) | 218 (145–261) | 0.811 |
| A-aO2 | 161 (124–205) | 146 (129–245) | 121 (105–225) | 0.344 |
| Lactate, mmol/l | 0.9 (0.7–1.4) | 1.3 (0.7–1.9) | 1.1 (0.8–1.2) | 0.458 |
| FiO2, % | 37 (33–48) | 40 (35–50) | 36 (35–47) | 0.317 |
| Laboratory tests | ||||
| CRP, mg/dL | 9.9 (5.8–20.4) | 10.1 (7.6–15.4) | 12.5 (10–21.5) | 0.582 |
| White blood cells count, 109/L | 6.48 (5.82–10.07) | 4.63 (2.91–6.07) | 7.01 (5.87–8.8) | 0.013** |
| Lymphocytes, 109/L | 1.04 (0.59–1.31) | 0.82 (0.62–1.35) | 0.79 (0.73–1.06) | 0.901 |
| Neutrophils, 109/L | 4.9 (4.3–8.57) | 3.67 (2.19–4.45) | 5.49 (4.44–7.55) | 0.007*** |
| Platelets, m/mm3 | 211(186–348) | 188 (131–289) | 272 (234–308) | 0.304 |
| Hb, g/dL | 12.9 (9.3–13.5) | 12 (10.6–12.8) | 12 (11.3–12.7) | 0.317 |
| Therapies | ||||
| Antibiotic | 6 (66.7%) | 14 (82.4%) | 5 (62.5%) | 0.497 |
| Corticosteroid | 4 (44.4%) | 12 (70.6%) | 2 (25%) | 0.087 |
| Hydroxychloroquine | 6 (66.7%) | 15 (88.2%) | 5 (62.5%) | 0.265 |
Abbreviations: COPD: chronic obstructive pulmonary disease, GERD: gastro-esophageal reflux disease, BT: body temperature, LRT: lung recruitment test, PaCO2: partial pressure of arterial carbon dioxide, PaO2: partial pressure of arterial oxygen, FiO2: fractional inspired oxygen, A-aO2: alveolar–arterial gradient of oxygen, CRP: C-reactive protein, Hb: hemoglobin.
Footnotes:
Although well-tolerated, only a small proportion of LRTs conducted in COVID-19 patients on helmet CPAP leads to a complete success, while a decrease of the A-aO2 lead to a partially success in about half of patients. Furthermore, the median level of PEEP was highly variable among patients that fully or partially success. The A-aO2 gradient was adopted as endpoint because of the COVID-19 pneumonia-related ARF. A-aO2 gradient can better assess gas exchange dysfunction in comparison with PaO2:FiO2 ratio being some of the patients hypocapnic. Notably, 14.7% of the tests failed because of hemodynamic instability. Physicians should be aware of strict monitoring of blood pressure and cava collapse at ultrasound evaluation during LRT, especially in case higher levels of PEEP are adopted. This study has several limitations. First, it was designed as a purely physiologic study, without assessment of the potential impact of tests on clinical outcomes. Further randomized, control trials are needed to evaluate the efficacy LTR on clinically meaningful outcomes such as intubation and mortality. Second, someone could speculate that a 30-minute length of duration of each PEEP application is relatively short to assess improvement in oxygenation as a consequence of a stable re-opening of atelectatic lung. Furthermore, the relatively low success rate might be mainly related to the complex pathophysiology of respiratory failure in COVID19 patients, where diffuse alveolar damage and diffuse endothelial damage leading to pulmonary intravascular coagulopathy with evidence of disseminated micro-thrombosis.
In conclusion, the identification of a positive physiological response to the application of PEEP during helmet CPAP treatment along with optimal levels of PEEP is of paramount importance among patients with hARF due to pneumonia in general and especially due to COVID-19. Our feasibility study paves the way for future clinical studies looking at a possible impact of LRT during helmet CPAP on clinically meaningful outcomes of COVID-19 patients.
FundingThis research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Conflict of interestProf. Aliberti reports grants and personal fees from Bayer Healthcare, grants and personal fees from Aradigm Corporation, grants and personal fees from Grifols, personal fees from Astra Zeneca, personal fees from Basilea, personal fees from Zambon, personal fees from Novartis, personal fees from Raptor, grants and personal fees from Chiesi, personal fees from Actavis UK Ltd, personal fees from Horizon, grants and personal fees from INSMED, outside the submitted work. Prof. Blasi reports grants and personal fees from astrazeneca, grants from bayer, grants and personal fees from chiesi, grants and personal fees from gsk, personal fees from guidotti, personal fees from grifols, grants and personal fees from insmed, personal fees from menarini, personal fees from mundipharma, personal fees from novartis, grants and personal fees from pfizer, personal fees from zambon, outside the submitted work.
We would like to acknowledge the support of all the respiratory fellows and consultants, nurses and respiratory physiotherapists of the COVID-19 high-dependency unit of the Policlinico Hospital in Milan.