Although the etiology and pathogenesis of autoimmune diseases is often unknown, the interaction of various environmental factors in genetically predisposed individuals causes an abnormal immune response.1 Thus, exposure to silica dust, in addition to causing respiratory diseases, is considered a risk factor for the development of certain autoimmune diseases: rheumatoid arthritis, scleroderma, systemic lupus erythematosus, and vasculitis.2 Although some isolated cases of the association between Sjögren's syndrome (SS) and the risk of silicosis have been published, there are few references on the subject in the medical literature (Pubmed, keywords, Sjögren's syndrome and silicosis). For this reason, we believe that this case is of interest.
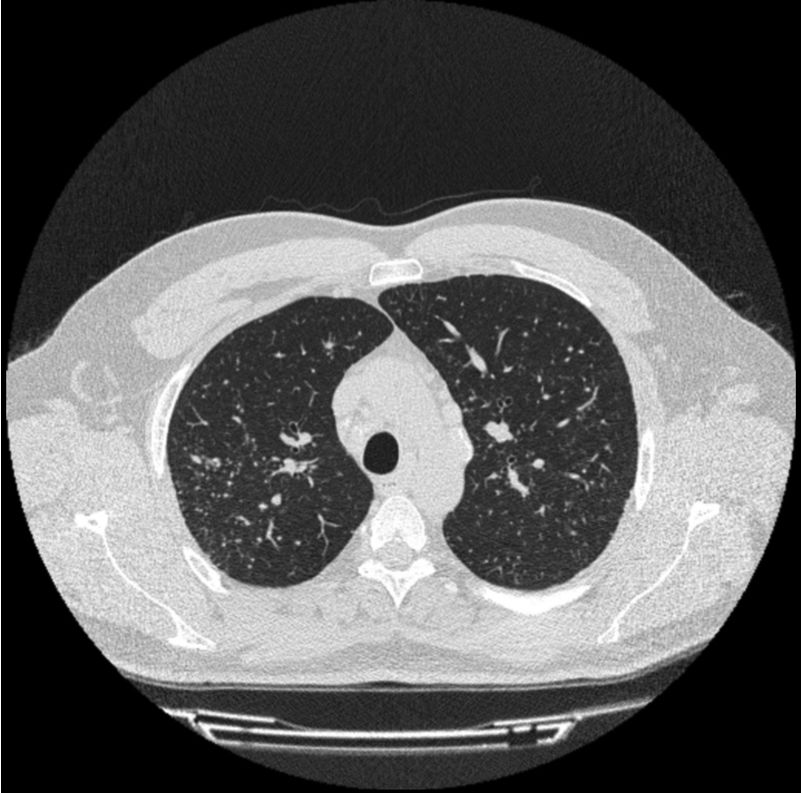
Our patient was a 36-year-old man, employed for the last 20 years in the cutting of ornamental rock (continuous occupational exposure to quartz, granite, marble, etc.), and an active smoker of 5 pack-years. He consulted with a 1-year history of episodes of arthritis in the elbows, wrists and proximal interphalangeal joints, with morning stiffness and limited hand flexion, which improved throughout the day. This was accompanied a 1-year history of dyspnea on moderate exertion and dry mouth and eyes. Physical examination revealed inflammation and swelling of the proximal interphalangeal joint, wrist and left elbow. Pulmonary auscultation was normal. Clinical laboratory tests showed raised acute phase reactants (ESR 35mm and CRP 25mg/l) and strongly positive anti-Ro and anti-La. Other parameters (ANA, rheumatoid factor, and anti-citrullinated protein antibodies) were negative. Schirmer's test was positive (3mm) and salivary scintigraphy showed moderate-to-severe hypofunction of the parotid glands and submaxillary glands. Chest computed tomography (CT) (Fig. 1) revealed the presence of mediastinal and hilar lymphadenopathies and a diffuse micronodular pattern in the upper fields. Respiratory function tests (spirometry, plethysmography, diffusion, and walk test) were normal. The patient was given a diagnosis of silicosis and SS, and, in view of the intensity of his arthritis, treatment began with tapering doses of prednisone and hydroxychloroquine, with subsequent improvement.
SS is an autoimmune disease characterized by dysfunction of the exocrine glands and sometimes non-glandular organs due to lymphocyte invasion. Both genetic factors and other environmental factors contribute to the pathogenesis of this entity.
It has been known since the 1950s that silica exposure is associated with the development of rheumatoid arthritis and scleroderma.3 Since then, multiple studies have linked silica exposure with autoimmune diseases. Rosemann et al.4 observed a prevalence of rheumatoid arthritis of 5.2% in a study of 463 patients with silicosis. Another US study5determined the association of silica exposure with systemic lupus erythematosus; and a recent meta-analysis links it with the risk of ANCA-associated vasculitis.6
In spite of the foregoing studies, the relationship between these diseases and silicosis has not been fully characterized. Inhalation of silica dust causes the activation and apoptosis of alveolar macrophages, with the resulting release of antigens. These antigens activate macrophages and dendritic cells that migrate to local lymph nodes, where they stimulate B and T cells, thus perpetuating an autoimmune response.7
Very few cases of SS associated with exposure to silica have been reported.8,9 The symptoms that cause glandular involvement are less marked than those caused by other organs, possibly contributing to underdiagnosis. Moreover, the majority of workers exposed to silica are men, so SS is rarely suspected, since this disease occurs predominantly in women. It is not clear, however, if it is the exposure to crystalline silica which contributes to the development of autoimmune diseases or if, inversely, the presence of these diseases confers an increased susceptibility to silica dust.10
Another aspect to bear in mind is that the prevalence of lung disease in SS, defined as the presence of symptoms or functional changes, ranges from 9% to 22%, or close to 50% if subclinical disease is taken into consideration.11 The spectrum of pulmonary manifestations of primary SS covers three groups: airway abnormalities (bronchiolitis, bronchial hyperreactivity, and bronchiectasis), interstitial lung disease (non-specific interstitial pneumonia, usual interstitial pneumonia, chronic lymphocytic interstitial pneumonitis, and organizing pneumonitis), and another miscellaneous group (pulmonary lymphoma, amyloidosis, pulmonary hypertension, and pulmonary thromboembolism).12 Our case showed the classic manifestation of simple silicosis: diffuse bilateral nodular pattern, predominantly in both upper lobes.
We believe that occupational exposure to silica must be taken into account in the evaluation of patients with autoimmune diseases and vice versa. Patients exposed to silica dust should be actively screened for signs and symptoms of autoimmune diseases, including SS.
Please cite this article as: Asenjo MM, Guerra JM, Pérez CI, Paula JM. Silicosis y síndrome de Sjögren. Arch Bronconeumol. 2019;55:537–538.