Rothia mucilaginosa is a gram-positive coccus that occurs as part of the normal flora of the oropharynx and upper respiratory tract. Lower respiratory tract infections caused by this organism are rare and usually occur in immunocompromised patients. This is the case of an immunocompetent 47-year-old woman with right upper lobe pneumonia in which R. mucilaginosa was isolated in sputum and bronchial aspirate. Infections caused by this agent in the last four years in our hospital were reviewed. The most common predisposing factor was COPD with bronchiectasis. R. mucilaginosa was identified as the causative agent for pneumonia in only two cases, of which one was our case and the other was a patient with lung cancer.
Rothia mucilaginosa es un coco grampositivo que forma parte de la flora normal de la orofaringe y del tracto respiratorio superior. Las infecciones del tracto respiratorio inferior por este germen son infrecuentes y se presentan habitualmente en pacientes inmunocomprometidos. Presentamos una mujer de 47años inmunocompetente con neumonía en lóbulo superior derecho en la que se aísla R. mucilaginosa en esputo y en broncoaspirado. Además se revisan las infecciones por este germen en los últimos cuatroaños en nuestro hospital. En dicha revisión el factor predisponente más frecuente ha sido la EPOC con bronquiectasias, y en solo dos casos se ha identificado el germen como agente causante de neumonía, entre los que se encuentra nuestro caso y el de un paciente con una neoplasia pulmonar.
Rothia mucilaginosa is a Gram-positive coagulase-negative coccus, known until 2000 as Stomatococcus mucilaginosus. It can be difficult to identify, and confusion with Micrococcus, Streptococcus or Staphylococcus may lead to underdiagnosis. The genus Rothia belongs to the Micrococcaceae family and at least four species have been recognized: Rothia dentocariosa, mucilaginosa, nasimurium and amarae. The first two species form part of the oropharyngeal microflora, and have been described as the causative agents of several diseases in humans. It was first described as a source of infection in humans in a case of endocarditis published in 1978.1 In recent years it has been isolated primarily in immunocompromised patients and more rarely in immunocompetent subjects. Cases of bacteremia, endocarditis, central nervous system infections, urinary infections, osteomyelitis, peritonitis, and, very occasionally, lower respiratory tract infections have been reported.2 We describe here a case of pneumonia due to R. mucilaginosa, initially presenting as hemoptysis, in an immunocompetent patient, and we review the experience in our hospital over the last four years.
Clinical ReportA 47-year-old woman, non-smoker, was admitted to our hospital with a 15-day history of right pleuritic chest pain and scant hemoptysis. She had previously received treatment with levofloxacin without improvement.
The patient did not report any significant disease history, except for mild intermittent asthma not requiring inhaled corticosteroids.
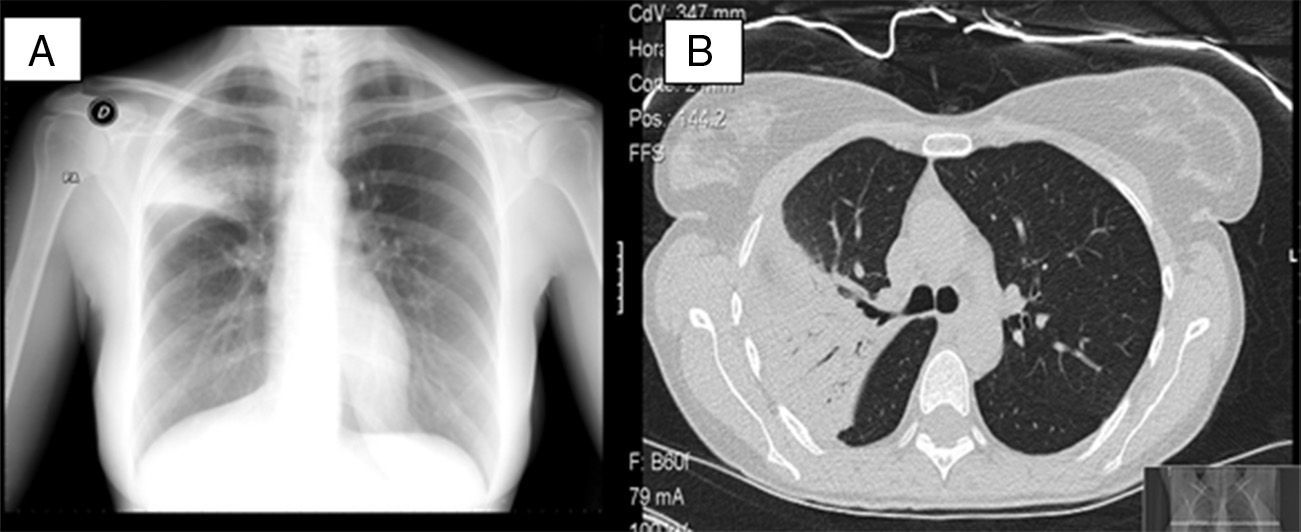
Chest X-ray (Fig. 1A) revealed alveolar infiltrate in the right upper lobe (RUL) with loss of volume. Chest CT (Fig. 1B) showed parenchymal consolidation with ground-glass opacities. R. mucilaginosa was isolated on two occasions from sputum cultures (Table 1). Fiberoptic bronchoscopy was performed, revealing transparent secretions with bloody traces originating in the RUL. R. mucilaginosa was also isolated from the bronchial aspirate (>105CFU/ml). Results from blood culture, Legionella/Pneumococcus antigens in urine, Mantoux, HIV serology, immunoglobulins, and sweat test were all normal. She was treated with amoxicillin-clavulanate for 14 days with full radiological resolution. The patient also underwent spirometric testing which was normal.
(A) Posteroanterior chest X-ray showing alveolar condensation and loss of volume in right upper lobe. (B) Chest computed tomography showing parenchymal consolidation and ground-glass opacities in right segment 3 and part of apical and anterior segments with air bronchogram and a fully patent bronchus.
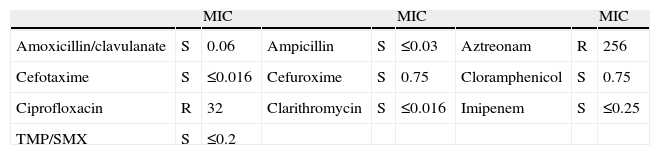
Antibiogram of the Rothia mucilaginosa Isolated in Sputum and Bronchial Aspirate.
| MIC | MIC | MIC | ||||||
| Amoxicillin/clavulanate | S | 0.06 | Ampicillin | S | ≤0.03 | Aztreonam | R | 256 |
| Cefotaxime | S | ≤0.016 | Cefuroxime | S | 0.75 | Cloramphenicol | S | 0.75 |
| Ciprofloxacin | R | 32 | Clarithromycin | S | ≤0.016 | Imipenem | S | ≤0.25 |
| TMP/SMX | S | ≤0.2 |
MIC: minimum inhibitory concentration (μg/ml); R: resistant; S: sensitive; TMP/SMX: trimethoprim-sulfamethoxazole.
The R. mucilaginosa isolates identified with mass spectrometry (MALDI-TOF) in the microbiology laboratory of our hospital between March 2009 and March 2013 were reviewed. During this period, the coccus was isolated in 22 patients from sputum (13), blood (4), pleural fluid (2), peritoneal cavity fluid (1), urine (1) and bronchial aspirate (1) samples. Sputum samples were good quality (sputum>25 polymorphonuclear leukocytes and <10 epithelial cells). None of the isolates were resistant to third-generation cephalosporins. Penicillin resistance was determined in 17% of the cases and quinolone resistance in 50%. A total of 42% of the subjects had received empirical treatment with levofloxacin, 37% with third-generation cephalosporins, and 21% with other antibiotics.
The most prevalent infections were bronchitis/exacerbations of bronchiectasis (50%), bacteremia (14%), pleural empyema (9%), pneumonia (9%), ascites (9%) and urinary infection (9%).
Two of our patients were completely healthy. Six were immunocompromised (five had active oncohematological disease and another was receiving cyclophosphamide for pulmonary fibrosis associated with rheumatoid arthritis). Finally, there were 14 non-immunocompromised patients who presented comorbidities. Of these, eight (57%) had COPD with bronchiectasis but had not received systemic steroids in the previous three months.
DiscussionWe report a case of pneumonia in which R. mucilaginosa was the single agent isolated from two good quality sputum samples and bronchial aspirate in an apparently immunocompetent patient. This microbe is rarely identified as a causative agent in lower respiratory tract infection.2 Clinical manifestations range from mild bronchitis to recurrent pneumonia and pulmonary abscess,3 and most patients have some form of immunodeficiency. The clinical significance of R. mucilaginosa in respiratory samples is difficult to determine; in the diagnosis of R. mucilaginosa pneumonia, therefore, it is advisable to obtain a culture from a bronchoscopy specimen.4,5 Vancomycin, third-generation cephalosporins and rifampicin are the treatments of choice for this disease. Sensitivity to penicillin, clindamycin and macrolides can be variable, while resistance rates to quinolones and aminoglycosides are higher.
According to the review performed in our hospital, 22 cases of infection due to this microbe were diagnosed in a 4-year period, two of which were pneumonia (the case described here and another patient with lung cancer). It is worth mentioning that a predisposing factor in 8 of our patients with R. mucilaginosa isolated in sputum was COPD with bronchiectasis. Accordingly, this coccus should be considered in patients of this type, even if there is no evidence of immunodeficiency, given the possibility of resistance to empirical antibiotics habitually used in this situation.
Conflict of InterestsThe authors state that they have no conflict of interests directly or indirectly associated with the content of this manuscript.
Please cite this article as: Baeza Martínez C, Zamora Molina L, García Sevila R, Gil Carbonell J, Ramos Rincon JM, Martín Serrano C. Neumonía por Rothia mucilaginosa en paciente inmunocompetente. Arch Bronconeumol. 2014;50:493–495.