The high prevalence of epithelial lung tumors means that mesenchymal lung tumors are rarities seen very occasionally in routine clinical practice. We report the case of a patient referred to the respiratory medicine clinic for the study of recurrent self-limiting episodes of bloody expectoration.
This was a female patient, 56 years of age, originally from Venezuela, who presented due to repeated self-limiting episodes of expectoration of blood while coughing, not affecting hemodynamic parameters, blood gases, or blood counts. Four years previously in her home country, she had been diagnosed on CT with a prevertebral solid tumor, determined on thoracotomy to be thoracic hemangioma, that was treated by radiation therapy (total dose unknown). Other surgical history included left inguinal herniorrhaphy, hysterectomy, and double adnexectomy. Physical examination was unremarkable, and both complete blood count and biochemistry results were within normal ranges. Chest X-ray showed the presence of a round subcarinal lesion. CT confirmed the presence of a mass enhanced by administration of contrast medium, measuring about 8.5×4.5cm in diameter, located immediately behind the right pulmonary artery and accompanied by paraaortic and retroperitoneal lymphadenopathies.
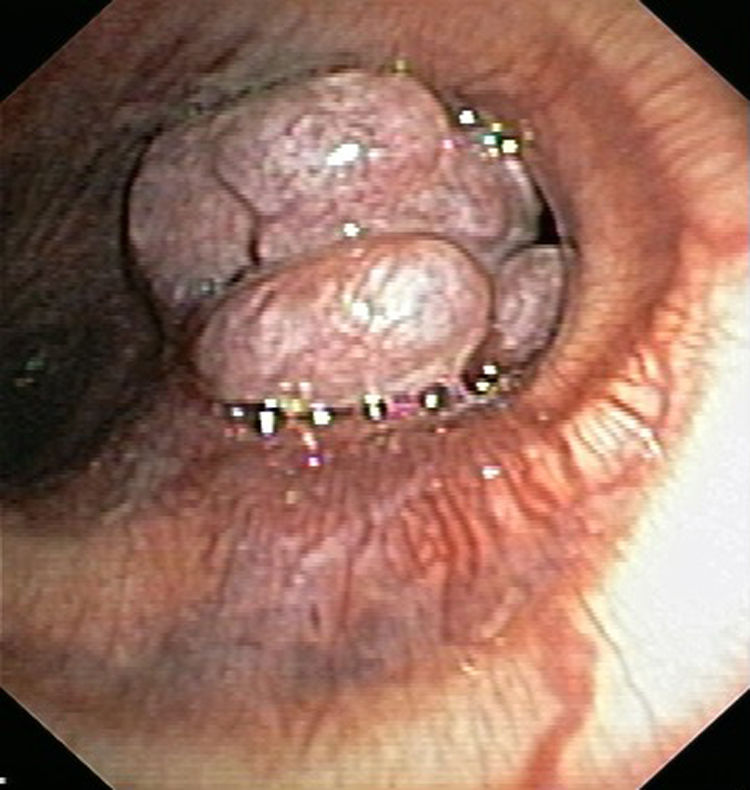
Fiberoptic bronchoscopy (Fig. 1) showed a purplish mass with a polylobulated surface located in the main carina, almost entirely occluding the entrance to the left main bronchus, but that did not prevent passage of the bronchoscope. The appearance of the endobronchial lesion and the patient's history of an intrathoracic vascular lesion made the collection of endoscopic biopsies inadvisable. Some days later, rigid bronchoscopy was performed under general anesthesia, and biopsies were collected, complicated by severe bleeding controlled with diathermy, followed by arteriography with embolization of the branches feeding the right bronchial artery. Biopsy samples showed the presence of numerous endothelized vessels with fibrous walls and a dense proliferation of cells with elongated nuclei arranged in bundles with variable spatial arrangement with no necrosis or mitosis, that were positive for markers CD34 and CD31, Ki-67 in less than 5%, and negative for epithelial and muscle markers; these findings were consistent with pulmonary hemangiopericytoma.
Between 2% and 6% of all primary mediastinal tumors are of mesenchymal origin. Of these, hemangiopericytoma is a rarity: it originates in the Zimmermann pericytes, which form part of the outer layer surrounding the endothelium of the capillaries and is now classified as perivascular tumor. It represents less than 2% of all soft tissue sarcomas,1 and approximately 1% of all tumors of vascular origin.2 The main sites tend to be the muscle tissue of the limbs, subcutaneous tissue, and the retroperitoneum. The chest (usually mediastinal) as the primary site of hemangiopericytoma is extremely rare, as attested by a review of literature which retrieved very few cases.3 Published cases of pulmonary involvement mostly began as solitary pulmonary nodules, and to our knowledge, this is the first case with endobronchial expression evidenced by bronchoscopy.
This type of tumor usually begins with a wide variety of symptoms, which most notably include hemoptysis.4 Image testing, especially CT with and without contrast and magnetic resonance imaging (MRI),1,2 helps to visualize the lesion and point to its vascular origin, although these tests are not diagnostic in themselves.
Pericytomas tend to behave unpredictably.5,6 In the case of our patient, after the initial diagnosis of angioma, the lesion grew slowly without showing any clinical signs or symptoms that would suggest malignancy. Findings that suggest a more aggressive behavior are: size greater than 10cm (with a 66% probability of metastasis), the existence of more than 3 mitoses per field, necrosis, pleural invasion, and vascular invasion.7 In our case, despite the size of the lesion (8.5×4.5cm), the absence of necrosis or mitosis on the pathology study suggested a lack of aggressive behavior up to the time of diagnosis. It is clear however that, since it was a large central endobronchial vascular tumor, the risk of potentially fatal local complications was high.
Treatment of tumors of this type is based primarily on surgical resection of the lesion, if possible.8 Prior embolization of the feeder arterial branches is always advisable. Postoperative radiation therapy also plays an important role in lesions of this type, while chemotherapy appears to have no clear benefit and is reserved for selected cases and always administered with palliative intent.
In our case, the site of the lesion and the treatments received earlier ruled out surgical resection, so we opted for tumor embolization, implantation of a silicone Dumon Y stent, and external radiation therapy. This prosthesis had to be removed 6 months later due to intractable cough, after which successive bronchial dilation procedures were performed using rigid bronchoscopy, during the last of which massive bronchial bleeding occurred due to rupture of the tumor mass that resulted in the death of the patient.
Please cite this article as: Hernández Pérez JM, Pérez Negrín L, López Charry CV. Hemangiopericitoma pulmonar primario. Arch Bronconeumol. 2019;55:593–594.