Pleural involvement in sarcoidosis is uncommon and appears in several forms. To document the incidence and characteristics of pleural effusion in sarcoidosis patients, a review of the cases diagnosed in our center between January 2001 and December 2012 was carried out. One hundred and ninety-five patients with sarcoidosis were identified; three (two men and one woman) presented with unilateral pleural effusion (1.5%): one in the right side and two in the left. Two were in stage II and one was in stage IV. The pleural fluid of the two patients who underwent thoracocentesis was predominantly lymphocytic. One of these patients presented chylothorax and the other had high CA-125 levels. In general, these effusions are lymphocyte-rich, paucicellular, serous exudates (sometimes chylothorax) and contain proportionally higher levels of protein than LDH. Most cases are treated with corticosteroids, although it may resolve spontaneously.
La afectación pleural en la sarcoidosis es baja y se puede manifestar de diversas formas. Con el objetivo de documentar la frecuencia y las características del derrame pleural en los pacientes con sarcoidosis, revisamos los casos diagnosticados en nuestro centro entre enero de 2001 y diciembre de 2012. De los 195 pacientes con sarcoidosis identificados, 3 (2 hombres y una mujer) tenían un derrame pleural unilateral (1,5%): uno derecho y 2 izquierdo; 2 en estadio ii y uno en estadio iv. El derrame de los 2 que se puncionaron era un exudado de predominio linfocítico. Uno de ellos correspondía a un quilotórax y el otro presentaba valores elevados de CA-125. Estos derrames suelen ser exudados serosos (a veces quilotórax), paucicelulares, de predominio linfocítico, con unas proteínas proporcionalmente más elevadas que la LDH. Pueden evolucionar favorablemente de forma espontánea, si bien en la mayoría de los casos se tratan con corticoides.
Sarcoidosis is a systematic granulomatous disease of unknown etiology and variable clinical manifestation and course. Although 90% of the patients have chest involvement, any organ can be affected.1 Pleural involvement occurs in between 0.7% and 10% of the cases,2 and can appear as pleural thickening or nodules, pneumothorax or pleural effusion (PE).3 Several mechanisms are involved in the development of PE in sarcoidosis, causing the pleural fluid (PF) to present different biochemical characteristics.3 To document the incidence and characteristics of PE in patients with sarcoidosis in our setting, we retrospectively reviewed the anatomical pathology records of cases diagnosed in our hospital between January 2001 and December 2012. Patients over the age of 18 years with sarcoidosis confirmed by biopsy were included, and any with an alternative cause for granulomatous disease were excluded. Clinical and radiological records were studied to determine the presence of PE, and age, sex, radiological staging, tuberculin test results and biopsied organs leading to diagnosis were recorded.
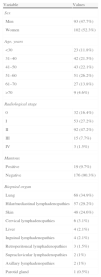
Clinical ObservationsA diagnosis of sarcoidosis was obtained in 195 patients, of whom 3 had unilateral PE. Demographic data are shown in Table 1 and PF characteristics in Table 2.
Demographic Data, Radiological Staging and Clinical Findings of Patients With Sarcoidosis (n=195).
| Variable | Values |
| Sex | |
| Men | 93 (47.7%) |
| Women | 102 (52.3%) |
| Age, years | |
| <30 | 23 (11.8%) |
| 31–40 | 42 (21.5%) |
| 41–50 | 43 (22.1%) |
| 51–60 | 51 (26.2%) |
| 61–70 | 27 (13.8%) |
| >70 | 9 (4.6%) |
| Radiological stage | |
| 0 | 32 (16.4%) |
| I | 53 (27.2%) |
| II | 92 (47.2%) |
| III | 15 (7.7%) |
| IV | 3 (1.5%) |
| Mantoux | |
| Positive | 19 (9.7%) |
| Negative | 176 (90.3%) |
| Biopsied organ | |
| Lung | 68 (34.9%) |
| Hilar/mediastinal lymphadenopathies | 57 (29.2%) |
| Skin | 48 (24.6%) |
| Cervical lymphadenopathies | 6 (3.1%) |
| Liver | 4 (2.1%) |
| Inguinal lymphadenopathies | 4 (2.1%) |
| Retroperitoneal lymphadenopathies | 3 (1.5%) |
| Supraclavicular lymphadenopathies | 2 (1%) |
| Axillary lymphadenopathies | 2 (1%) |
| Parotid gland | 1 (0.5%) |
Characteristics of Pleural Fluid in Patients With Sarcoidosis.
| Variable | Case 1 | Case 2 |
| Appearance | Milky | Serous |
| Nucleated cells (×106/l) | 1360 | 7000 |
| Differential count (%) | 70% lymphocytes; 24% macrophages | 62% lymphocytes; 27% macrophages |
| pH | 7.40 | 7.46 |
| Total protein (g/dl) | 4.2 | 5.9 |
| PE/S protein ratio | 0.63 | 0.9 |
| LDH (U/l) | 244 | 303 |
| PE/S LDH | 1 | 0.92 |
| Glucose (mg/dl) | 116 | 101 |
| ADA (U/l) | 36 | 45 |
| Cholesterol (mg/dl) | 98 | 78 |
| Triglycerides (mg/dl) | 251 | |
| CA-125 (U/l) | 1.343 | |
| Chylomicrons | Yes | |
| Cultures | Negative | Negative |
ADA: adenosine deaminase; CA-125: carbohydrate antigen 125; LDH: lactate dehydrogenase; PF: pleural fluid; S: serum.
A 45-year-old man presented with right chest pain and dyspnea with signs of PE on examination. Chest computed tomography (CT) showed bilateral hilar lymphadenopathies, right PE, micronodular disease and probable massive fibrosis in the right side. Angiotensin-converting enzyme (ACE) was 76U/l and Mantoux negative. PE was chylothorax. Biopsy of right paratracheal lymphadenopathies showed non-necrotizing epithelioid granulomas. Ziehl-Nielsen staining was negative. Treatment was started with octreotide and corticosteroids. PE resolved but reappeared on reduction or suspension of the corticosteroid (prednisone at less than 10mg/day).
Case 2An 83-year-old woman presented with left pleuritic pain and dyspnea with symptoms of PE on physical examination. Chest CT revealed multiple bilateral hilar and paratracheal mediastinal lymphadenopathies (2R, 4R, 4L), moderate left PE and suspected micronodular disease. ACE was 57U/l and Mantoux negative. PE was serous with elevated CA-125. Non-specific inflammatory changes were observed in the pleural biopsy and the transbronchial biopsy showed non-necrotizing granulomas. Treatment with corticosteroids was administered, resulting in good clinical progress and resolution of PE.
Case 3A 39-year-old man presented with a clinical picture of asthenia, cough, progressive dyspnea and dysphonia. Of interest on physical examination was auscultation of bibasal crackles. Results of blood tests were normal with the exception of ACE 170U/l, and the CT scan revealed bilateral hilar lymphadenopathies, small left PE and bilateral interstitial disease. Mantoux was negative and non-necrotizing granulomatous inflammation was found on biopsy. PE was not aspirated and resolved after treatment with corticosteroids.
DiscussionPrevalence of PE in our series (1.5%) is in the normal range,2 although rates of up to 8.2% have been reported.4 Mechanisms involved in the development of PE are increased capillary permeability due to direct involvement of the pleura (exudate) or obstruction of the thoracic duct (chylothorax), as observed in our cases. More rarely, PE may be hemorrhagic (due to compression of vessels by granulomas) or transudate (superior vena cava obstruction, atelectasia due to bronchial stenosis or trapped lung).
PE in sarcoidosis affects both sexes and occurs in any age group or radiological stage. It is generally small to moderate in size, and is more common in the right side. It can present at diagnosis or at a later time, coinciding with disease exacerbation.3 In this respect, our patients were of different sexes and their age groups, radiological stages and involved sides all varied.
PE is generally a paucicellular, lymphocyte-rich exudate with a high CD4/CD8 ratio, although it may also be eosinophilic, or predominantly neutrophilic. Protein levels are proportionally higher than LDH levels.2 ADA is usually low and high CA-125 may be found, as in case 2 reported here, in women with evidence of peritoneal sarcoidosis. CA-125 levels fall after treatment with corticosteroids.5 The tuberculin test is negative and diagnosis can be derived from biopsies of both lung and pleura that show non-caseating granulomas. The latter may suggest a diagnosis of tuberculous PE. PEs may resolve spontaneously but most cases require treatment with corticosteroids.
Authors ContributionFerreiro L. Editor and author. Development and design. Data analysis and interpretation. Final approval of manuscript.
San José E. Co-author. Data analysis and interpretation. Final approval of manuscript.
González-Barcala FJ. Co-author. Data collection. Article review. Final approval of manuscript.
Suárez-Antelo J. Co-author. Data collection. Article review. Final approval of manuscript.
Toubes ME. Co-author. Data collection. Article review. Final approval of manuscript.
Valdés L. Editor and author. Development and design. Data analysis and interpretation. Final approval of manuscript.
Please cite this article as: Ferreiro L, San José E, González-Barcala FJ, Suárez-Antelo J, Toubes ME, Valdés L. Derrame pleural y sarcoidosis: una asociación poco frecuente. Arch Bronconeumol. 2014;50:554–556.