Organizing pneumonia is a nonspecific pathologic pattern of response to lung damage. It can be cryptogenic, or secondary to different medical processes, such as infections, connective tissue diseases, drug toxicity and radiotherapy, among others. The characteristic radiological pattern consists of the presence of patchy and bilateral pulmonary consolidations of peribronchial and subpleural distribution. These lesions respond to treatment with corticoids, migrate with or without treatment, and tend to recur when treatment is decreased or withdrawn.1
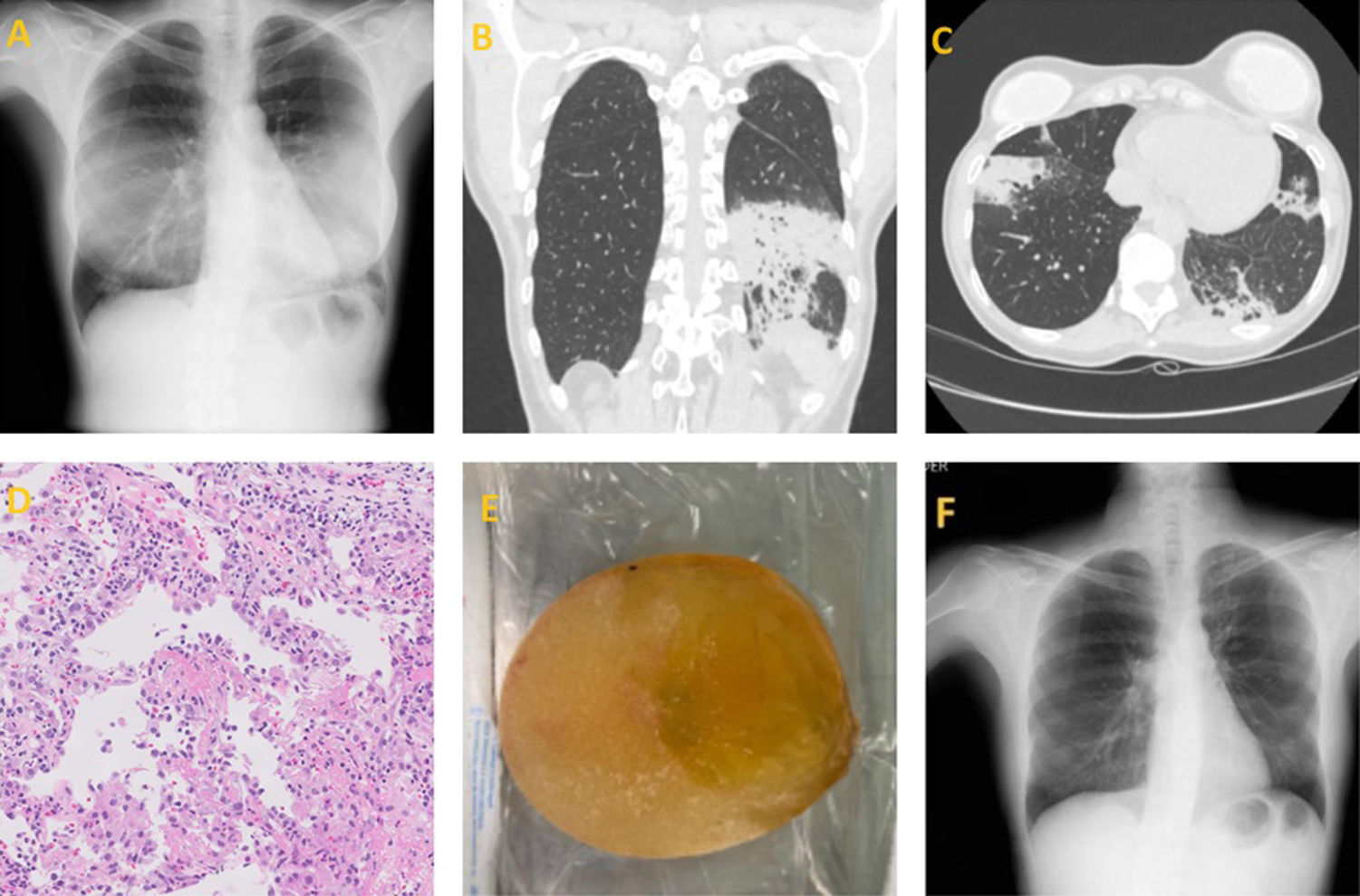
We present the case of a 50-year-old woman with a breast augmentation surgery (prothesis 270cc de Mcghan) in 1998 followed by a rupture of the left breast implant diagnosed in 2022. Three months after the patient suffered from cough and fever. In the chest X-ray (Fig. 1A) lung opacities were seen in both bases. The patient was treated with amoxicillin (500mg/8h). Days after the cough worsened and she presented weight loss (4kg) and chest pain. A chest computed tomography (CT) was performed (Fig. 1B), revealing many infiltrates with air bronchogram suggesting pneumonia in the left lower lobe. Treatment with levofloxacin (500mg/24h) and prednisone (0.5mg/kg/24h) was initiated with clinical improvement.
(A) Bilateral lung opacities in both bases are seen in this posteroanterior chest X-ray. (B) Coronal cut of a CT scan showing an infiltrate with air bronchogram suggesting pneumonia in the left lower lobe. (C) Axial cut of a CT scan showing new bilateral patchy consolidations, mainly subpleural, and partial resolution of the ones seen in the previous study. (D) Haematoxylin–eosin staining shows fragments of lung parenchyma with of alveolar septa by acute and chronic polymorphous inflammatory infiltrate, alveolar spaces partially lined by pneumocytes with reactive atypia, predominantly occupied by fibrin and some Masson bodies. (E) Rupture of the breast implant. (F) Resolution of the consolidations in subsequent studies.
After 3 months the CT scan was repeated (Fig. 1C), showing new bilateral patchy consolidations, mainly subpleural, and partial resolution of those seen in the previous study. A breast implant replacement was planned, but the patient began again with cough and fever, showing new right-lung infiltrates in the chest X-ray. The study was completed with a blood test (with negative autoantibodies) and a bronchoscopy. The bronchoalveolar lavage revealed an increased proportion of lymphocytes, mostly T-cells, and an inverted CD4/CD8 ratio. The transbronchial biopsy was described as pathologic pulmonary parenchyma affected with a pattern suggesting acute/fibrinous pneumonia with discrete signs of organization (Fig. 1D). Adding up all the previous results, the diagnosis of organizing pneumonia related to an autoimmune/inflammatory syndrome induced by adjuvants (ASIA) arose in the context of breast implant rupture.
The patient initiated a longer and descending corticosteroid treatment (prednisone, 0.5mg/kg/24h) for six months, with improvement of the radiological consolidations. During this period, both breast implants were surgically removed (showing the rupture of both prothesis) with total bilateral capsulectomy (Fig. 1E). The anatomical pathology studies showed that the capsule had a large cell foreign body-type lymph node reaction to a material that was consistent with breast implant material. Finally, the patient improved clinically and with subsequent disappearance of the pulmonary infiltrates (Fig. 1F).
The autoimmune/inflammatory syndrome induced by adjuvants (ASIA) is a severe and rare condition. Patients tend to have a genetic predisposition, characterized by inappropriate immune response to adjuvants such as silicone (used in breast implants), with a higher incidence of reactions when ruptured.2 After revising the scientific literature, we have not found any clinical cases that describe an association between ASIA and organizing pneumonia after breast implant.
The wide variety of symptoms (arthralgias, myalgias, fatigue and even pulmonary or systemic affections) makes the diagnosis more difficult, and therefore, the early approach to the disease is affected. The diagnosis of ASIA syndrome is based on the diagnostic criteria proposed by Shoenfeld,3,4 and subsequently by Alijotas-Reig.5 The main issue is their lack of specificity.6 Our patient met the criteria in both scales. Treating this syndrome requires taking away the trigger of the immune response and a correct immunosuppression.
The association between ASIA syndrome and organizing pneumonia is rare. Understanding this syndrome and making an accurate diagnosis is crucial to develop new effective treatment and diagnostic strategies.
FundingThe authors declare that they have not received any fees or funding for the development of the clinical case presented.
Authors’ contributionsAll authors have made substantial contributions in each of the following aspects: conception, clinical case design and data acquisition, drafting of the article and critical revision of the intellectual content, and final approval of the presented version.
Informed consentInformed consent was obtained from the patient's relative for publication of the clinical data and images present in this manuscript.
Conflicts of interestFV-Á has attended or participated in activities organized or financed by the pharmaceutical companies Almiral, AstraZeneca, Bial, BoehringerIngelheim, Chiesi, GlaxoSmithKline, Esteve, Ferrer, Menarini, Novartis, Mundipharma, Orion, Pfizer, Teva and Zambon.
The rest of the authors declare no conflict of interest.