Disease stage is the most important prognostic factor in lung cancer, and optimal staging is important to determine the best therapeutic option. FDG-PET/CT has demonstrated its value in the treatment of early stage non-small cell lung cancer (NSCLC), but there is still insufficient data to define its role in other stages.
HypothesisInformation provided by FDG-PET/CT has an impact on the therapeutic management of patients with NSCLC.
MethodsA retrospective review was made of patients who underwent FDG-PET/CT between January 2008 and December 2010 for the diagnosis of NSCLC. Clinical stage before and after FDG-PET/CT and information about any change in therapeutic decision due to information provided by FDG-PET/CT were collected. Using pathologic evaluation as the gold standard, sensitivity, specificity, and positive and negative predictive values for CT and FDG-PET/CT were calculated.
ResultsOf the 522 patients diagnosed of NSCLC, FDG-PET/CT was performed in 246 patients (47.1%). In 85 cases (34.6%) FDG-PET/CT led to stage migration. Treatment was modified in 60 patients (24.4% of all FDG-PET/CT performed), avoiding a futile thoracotomy in 13 cases (5.2%), and allowing treatment with curative intent in 26 (10.5%). Of 90 patients (36.5%) evaluated as stage iii by CT staging, FDG-PET/CT modified the therapeutic approach in 36 (40%). For 133 cases (54%) with pathological assessment of the mediastinal lymph nodes, sensitivity, specificity, positive predictive value and negative predictive value were 0.57, 0.64, 0.48 and 0.72 for CT, and 0.68, 0.86, 0.75 and 0.81 for FDG-PET/CT.
DiscussionOur data support previous reports that FDG-PET/CT is essential in the staging process not only for patients with potentially operable NSCLC, but also for stage iii patients, as demonstrated by our data.
El estadio de la enfermedad es el factor pronóstico más importante en el cáncer de pulmón, y una estadificación óptima es importante para determinar la mejor opción terapéutica. La FDG-PET/TC ha resultado útil en el estadio inicial del cáncer de pulmón no microcítico (CPNM) pero los datos existentes continúan siendo insuficientes para definir su papel en otros estadios.
HipótesisLa información aportada por la FDG-PET/TC tiene repercusiones en el enfoque terapéutico adoptado ante los pacientes con CPNM.
MétodosSe realizó una revisión retrospectiva de los pacientes a los que se practicó una FDG-PET/TC en el proceso diagnóstico del CPNM entre enero de 2008 y diciembre de 2010. Se obtuvieron datos relativos al estadio clínico antes y después de la FDG-PET/TC así como información sobre el cambio de la decisión terapéutica como consecuencia de la información aportada por la FDG-PET/TC. Utilizando como patrón de referencia el examen anatomopatológico, se calcularon los valores de sensibilidad, especificidad y valor predictivo positivo y negativo de la TC y la FDG-PET/TC.
ResultadosDe los 522 pacientes con un diagnóstico de CPNM, en 246 (47,1%) se realizó una FDG-PET/TC. En 85 casos (34,6%) la FDG-PET/TC comportó una migración del estadio. El abordaje terapéutico se modificó en 60 pacientes (24,4% del total de las FDG-PET/TC realizadas), y ello permitió evitar una toracotomía fútil en 13 casos (5,2%), e hizo posible un tratamiento con intención curativa en 26 (10,5%). De los 90 pacientes (36,5%) clasificados en el estadio iii mediante la estadificación de TC, la FDG-PET/TC modificó el abordaje terapéutico en 36 (40%). En los 133 casos (54%) con una evaluación anatomopatológica de los ganglios linfáticos mediastínicos, la sensibilidad, la especificidad, el valor predictivo positivo y el valor predictivo negativo fueron de 0,57; 0,64; 0,48 y 0,72 con la TC; y de 0,68; 0,86; 0,75 y 0,81 con la FDG-PET/TC.
DiscusiónNuestros datos respaldan los de trabajos previos que indican que la FDG-PET/TC es esencial en el proceso de estadificación, no solo en los pacientes con un CPNM potencialmente operable, sino también en los pacientes en estadio iii, tal como ponen de manifiesto nuestros datos.
Lung cancer is a major worldwide cause of death, with more than 1.3 million deaths per year. Non-small-cell lung cancer (NSCLC) accounts for over 80% of all primary lung tumors.1 Despite efforts to achieve early diagnosis and the advances made in the treatment of these patients, overall survival remains low at about 15% in 5-year survival.2 These poor results are due to the high percentage of patients with advanced disease at diagnosis, and the low survival rate of patients with limited, potentially curable disease, possibly reflecting the inability to detect advanced disease at initial staging.3 The disease stage is considered the most important factor in lung cancer prognosis, and optimal staging is important for determining the best therapeutic approach.
A computed tomography (CT) scan with contrast is usually the first step in the clinical staging of NSCLC. A CT scan provides excellent information for the assessment of primary tumors,4 but has limited value for the assessment of metastatic lymph nodes, since normal size lymph nodes are systematically considered benign.5 Moreover, the area covered in the CT scan for initial staging of NSCLC is usually the chest and upper abdomen, limiting the detection of distant metastases to these areas.
During the last decade, positron emission tomography with [18F] fluoro-2-deoxy-d-glucose (FDG-PET/CT) has been increasingly used for NSCLC staging. The detection rate of metastases in mediastinal lymph nodes of FDG-PET/CT is higher than that of CT, and FDG-PET/CT detects occult distant metastases in approximately 10% of patients.6 FDG-PET/CT improves the diagnostic accuracy of NSCLC staging,7 and reduces the number of futile thoracotomies.8 FDG-PET/CT has also been shown to modify or influence treatment decisions in patients with NSCLC.9 International guidelines recommend the use of FDG-PET/CT in all stages except in cases with obvious metastatic disease.2,10
FDG-PET/CT has been shown to be a valuable diagnostic instrument for patients with initial stage NSCLC,5–9 but available data are still insufficient to define the role of FDG-PET/CT in other stages.
Our aim was to investigate the impact of obtaining an FDG-PET/CT scan at the time of diagnosis on treatment decisions, according to approved clinical practice guidelines.11
MethodsWe conducted a retrospective screening of all patients diagnosed with NSCLC at our institution between January 2008 and December 2010. Our center is a reference teaching hospital in its area of influence. Patients included in the study were identified with the use of a prospectively collected institutional cancer registry database.
Diagnostic StudyAll patients with suspected lung cancer were initially evaluated by a pulmonologist, who decided the diagnostic examinations.
The workup included a conventional staging based on review of the medical history, a physical examination, the results of a chest and upper abdomen CT scan, bronchoscopy and/or CT-guided biopsy. As established in the international guidelines,2,10,11 a FDG-PET/CT scan was performed in all patients considered candidates for curative treatment (without obvious metastatic disease on CT and without comorbidities contraindicating radical treatment). The FDG-PET/CT scans were performed with a full body PET-CT scanner (General Electric Discovery ST: 47 planes, 3.27mm slice thickness, 128×128 voxels, 1.95mm×1.95mm). CT acquisition parameters were 140kV, 80mA, slice 0.8/2.5mm. Reconstruction was performed using a filtered backprojection algorithm, with FWHM ramp filter of 5.45mm. Patients fasted for at least 6h before the scan and serum glucose concentration was less than 140mg/dl in all patients. PET scanning was started 60min after intravenous administration of 4.62MBq (0.125mCi)/kg of FDG.
All cases were then evaluated by a multidisciplinary team of pulmonologists, thoracic radiologists, nuclear medicine specialists, pathologists, thoracic surgeons, radiation oncologists and clinical oncologists, in order to make therapeutic decisions.
Mediastinal invasion was suspected on CT scan when enlarged lymph nodes with a short axis of more than 10mm were observed. All PET-CT scans were interpreted by visual and semiquantitative analysis. In visual analysis, PET-CT was considered positive if lesion FDG uptake was greater than mediastinal background uptake. Semiquantitative analysis was based on the standardized uptake value (SUV) in a region of interest. SUVs above 2.5 in the lesion area were considered positive. When no correlation between SUV and visual analysis was found, the clinical context of the patient was taken into account to reach a final decision regarding any positive or negative lesion.
If a mediastinal involvement was suspected on CT or FDG-PET/CT, a biopsy of mediastinal lymph nodes was obtained by endobronchial ultrasound-guided transbronchial needle aspiration (EBUS-TBNA). If EBUS-TBNA was found negative or not evaluable (without representation of lymphoid cells after at least three needle passes),12 a mediastinoscopy was performed. Patients with confirmed negative mediastinal lymph nodes were treated with surgery, which included a systematic dissection of the ipsilateral hilar and mediastinal lymph nodes.
Demographic, clinical and pathological data of each patient were obtained from the medical history records and included in a database. The clinical stage before and after obtention of FDG-PET/CT scan was defined according to the seventh edition of the cancer staging manual of the American Joint Committee on Cancer.13 Details on changes in treatment decisions as a result of information provided by FDG-PET/CT were also collected.
Clinical assessments of disease in mediastinal lymph nodes obtained by using CT and FDG-PET/CT were classified into binary classes of true-positive, false-positive, true-negative and false-negative (FN) using pathologic evaluation as the reference standard. Sensitivity, specificity and positive and negative predictive values for CT and FDG-PET/CT were calculated. In patients with available mediastinal evaluation by EBUS-TBNA or systematic lymph node dissection in surgery, surgical evaluation was considered the reference standard. Patients who had received chemotherapy or radiotherapy prior to FDG-PET/CT or pathologic evaluation were excluded from the analysis. Statistical analyses were performed using a specialized software (SPSS for Windows v.19).
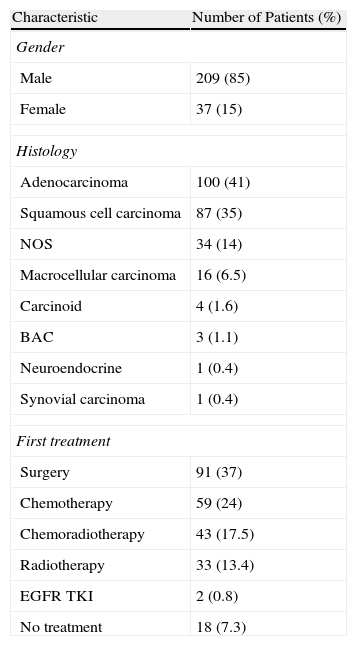
ResultsPatient Characteristics and TreatmentBetween January 2008 and December 2010, 522 patients were diagnosed with NSCLC in our hospital, and FDG-PET/CT was performed in 246 (47.1%). Patient characteristics are summarized in Table 1. The vast majority were men and the predominant histology was adenocarcinoma. Surgery was the most common initial treatment, followed by palliative chemotherapy, concurrent chemoradiotherapy, and radiotherapy alone.
Demographic Characteristics.
| Characteristic | Number of Patients (%) |
| Gender | |
| Male | 209 (85) |
| Female | 37 (15) |
| Histology | |
| Adenocarcinoma | 100 (41) |
| Squamous cell carcinoma | 87 (35) |
| NOS | 34 (14) |
| Macrocellular carcinoma | 16 (6.5) |
| Carcinoid | 4 (1.6) |
| BAC | 3 (1.1) |
| Neuroendocrine | 1 (0.4) |
| Synovial carcinoma | 1 (0.4) |
| First treatment | |
| Surgery | 91 (37) |
| Chemotherapy | 59 (24) |
| Chemoradiotherapy | 43 (17.5) |
| Radiotherapy | 33 (13.4) |
| EGFR TKI | 2 (0.8) |
| No treatment | 18 (7.3) |
CBA: bronchoalveolar carcinoma; EGFR TKI: epidermal growth factor receptor tyrosine kinase inhibitors; NOS: not otherwise specified.
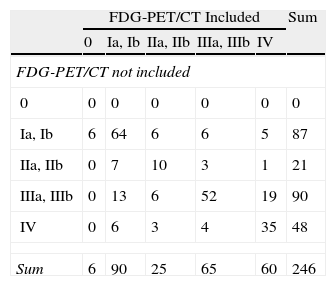
With conventional staging by CT, 87 patients (35.5%) were classified in stage I, 21 (8.5%) in stage II, 90 (36.5%) in stage III and 48 (19.5%) in stage IV. Staging after FDG-PET/CT scan was as follows: six patients (2.4%) with no FDG uptake were classified in stage 0, 90 (36.5%) in stage I, 25 (10.1%) in stage II, 65 (26.5%) in stage III and 60 (24.5%) in stage IV.
FDG-PET/CT did not modify staging in 65.4% of cases. Forty of the 85 patients (34.6%) with stage migration were classified in a more severe stage and 45 in a less severe stage according to FDG-PET/CT data. FDG-PET/CT detected metastatic disease unidentified in conventional staging in 25 patients (10.1%). Of these metastases, four were histologically confirmed. In cases without histological confirmation, the clinical progression of these lesions during follow-up confirmed their malignant nature. FDG-PET/CT also identified concomitant malignancies in other organs (pancreas, rectum and pharynx) in three cases, all histologically confirmed. A detailed presentation of conventional CT and FDG-PET/CT staging is shown in Table 2.
Stage Migration.
| FDG-PET/CT Included | Sum | |||||
| 0 | Ia, Ib | IIa, IIb | IIIa, IIIb | IV | ||
| FDG-PET/CT not included | ||||||
| 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Ia, Ib | 6 | 64 | 6 | 6 | 5 | 87 |
| IIa, IIb | 0 | 7 | 10 | 3 | 1 | 21 |
| IIIa, IIIb | 0 | 13 | 6 | 52 | 19 | 90 |
| IV | 0 | 6 | 3 | 4 | 35 | 48 |
| Sum | 6 | 90 | 25 | 65 | 60 | 246 |
FDG-PET/CT: positron emission tomography with [18F] fluoro-2-deoxy-d-glucose/computed tomography.
Cases in which the clinical stage was not modified are indicated in gray. The table elements located above the gray line indicate patients in whom FDG-PET/CT indicated a higher stage. Elements located below the gray line correspond to patients in whom stage was considered lower after FDG-PET/CT.
Given the high negative predictive value of FDG-PET/CT in mediastinum evaluation, in those cases classified as N0 by FDG-PET/CT, a tissue sample of mediastinal lymph nodes was not systematically obtained to confirm N0 before surgery. In 13 of the 45 patients in whom stage was reduced after FDG-PET/CT examination, histological confirmation of mediastinum was not available because surgical intervention was not carried out. The reasons were poor respiratory function in eight cases, rapid clinical deterioration in two, treatment rejection by two patients, and diagnosis of simultaneous pancreatic cancer in one case. In the other 32 cases, histological confirmation of mediastinal lymph nodes was obtained by systematic dissection of ipsilateral hilar and mediastinal lymph nodes during surgery.
Changes in the Therapeutic Approach Based on the Results of FDG-PET/CTIn 60 (24.4% of all FDG-PET/CT performed) of the 85 patients in whom FDG-PET/CT results modified the clinical stage, stage migration involved a modification of the therapeutic approach. This therapeutic change followed an increase in severity stage in 31 cases (12.6%). FDG-PET/CT indicated a more severe stage in 13 cases that had been classified as stage I or II by CT, and initially candidates for radical surgery. The new classification was stage III, with mediastinal involvement in nine cases (candidates for radical chemoradiotherapy), and stage IV in 4 cases (candidates for palliative chemotherapy). The remaining 18 cases were initially classified stage III by CT, theoretically candidates for radical chemoradiotherapy, and FDG-PET/CT indicated stage IV. As a result of these stage modifications, the use of FDG-PET/CT prevented futile thoracotomy in 13 cases (5.2%) and futile chemoradiotherapy treatment in 18 (7.3%).
FDG-PET/CT results modified the therapeutic approach in 29 patients (11.7%) in whom stage level was reduced, which allowed curative treatment in 26 (10.5%). Seventeen patients (6.9%) were treated with surgery including a systematic dissection of the ipsilateral hilar and mediastinal lymph nodes (13 lobectomies, 3 bilobectomies and one segmentectomy). Six patients (2.4%) received radical radiotherapy (surgery was ruled out due to comorbidity), and three (1.2%) radical chemoradiotherapy (two cases classified stage III by FDG-PET/CT and one treated with induction chemoradiotherapy). Curative treatment was discarded in the remaining three patients due to severe comorbidity. Radical surgery was performed in the 17 cases in which FDG-PET/CT reduced the stage, and clinical and pathological stagings were compared. Staging remained unchanged in 10 cases, and three cases classified as stage I by FDG-PET/CT were considered to be in a more severe stage after pathological examination. Of these, two patients were diagnosed as stage II and one as stage III. Two cases clinically classified in stage II had pathologic stages I and III. One patient classified in stage I was found to be stage III (FN of FDG-PET/CT) and, finally, a case initially classified in stage III had a pathologic stage II (mediastinal invasion was assessed by mediastinoscopy before surgery due to discrepancies between CT and FDG-PET/CT results).
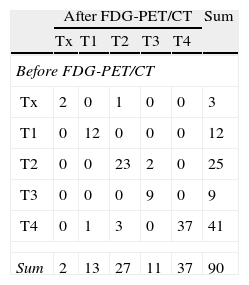
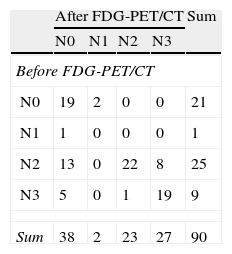
CT results initially classified most patients in our series as stage III. We decided to specifically analyze the impact of FDG-PET/CT in this subgroup of patients. FDG-PET/CT modified the therapeutic approach in 38 (42%) of the 90 patients (36.5%) classified as stage III by CT, and initially candidates for treatment with radical chemoradiotherapy (±surgery). Stage was more severe in 19 patients after FDG-PET/CT, who were therefore treated with palliative chemotherapy. The stage of the remaining 19 was milder, and they were treated with surgery. Details on the T and N classification according to FDG-PET/CT results in patients considered to be in stage III by CT are summarized in Tables 3 and 4, respectively.
T Migration in Patients Classified Stage III by CT.
| After FDG-PET/CT | Sum | |||||
| Tx | T1 | T2 | T3 | T4 | ||
| Before FDG-PET/CT | ||||||
| Tx | 2 | 0 | 1 | 0 | 0 | 3 |
| T1 | 0 | 12 | 0 | 0 | 0 | 12 |
| T2 | 0 | 0 | 23 | 2 | 0 | 25 |
| T3 | 0 | 0 | 0 | 9 | 0 | 9 |
| T4 | 0 | 1 | 3 | 0 | 37 | 41 |
| Sum | 2 | 13 | 27 | 11 | 37 | 90 |
FDG-PET/CT: positron emission tomography with [18F] fluoro-2-deoxy-d-glucose/computed tomography; CT: computed tomography.
Cases in which T was not modified are indicated in gray. The table elements located above the gray line indicate patients in whom FDG-PET/CT indicated a higher T. Elements located below the gray line correspond to patients in whom T was considered lower after FDG-PET/CT.
N Migration in Patients Classified Stage III by CT.
| After FDG-PET/CT | Sum | ||||
| N0 | N1 | N2 | N3 | ||
| Before FDG-PET/CT | |||||
| N0 | 19 | 2 | 0 | 0 | 21 |
| N1 | 1 | 0 | 0 | 0 | 1 |
| N2 | 13 | 0 | 22 | 8 | 25 |
| N3 | 5 | 0 | 1 | 19 | 9 |
| Sum | 38 | 2 | 23 | 27 | 90 |
FDG-PET/CT: positron emission tomography with [18F] fluoro-2-deoxy-d-glucose/computed tomography; CT: computed tomography.
Cases in which N was not modified are indicated in gray. The table elements located above the gray line indicate patients in whom FDG-PET/CT indicated a higher N. Elements located below the gray line correspond to patients in whom N was considered lower after FDG-PET/CT.
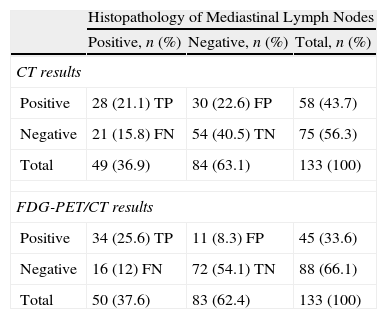
Of the 246 cases evaluated with FDG-PET/CT, pathologic evaluation of mediastinal lymph nodes was available in 133 (54%); systematic surgical dissection of the lymph nodes has been performed in 88, and EBUS-TBNA in 45. Given the high negative predictive value of FDG-PET/CT in the evaluation of mediastinal invasion in cases classified as N0, a tissue sample of mediastinal lymph nodes was not systematically obtained before surgery. In 13 of the 45 patients in whom stage was reduced after FDG-PET/CT examination, histological confirmation of mediastinum was not available because the surgical intervention was not carried out. The reasons were deterioration of respiratory function in 8 cases, rapid clinical deterioration of two patients, treatment rejection by two patients, and diagnosis of simultaneous pancreatic cancer in one case. In the other 32 cases, histological confirmation of mediastinal lymph nodes was obtained by systematic dissection of ipsilateral hilar and mediastinal lymph nodes. Mediastinal involvement was not explored in all stage IV cases because the therapeutic strategy (i.e. palliative chemotherapy) had not been modified by its results.
Using pathologic evaluation as the reference standard, sensitivity was 0.57 and 0.68 for CT and FDG-PET/CT, respectively. Specificity was 0.64 for CT and 0.86 for FDG-PET/CT. Positive predictive value was 0.48 for CT and 0.75 for FDG-PET/CT, and negative predictive value was 0.72 for CT and 0.81 for FDG-PET/CT. Statistical analysis results are summarized in Table 5.
Staging of Mediastinal Lymph Nodes.
| Histopathology of Mediastinal Lymph Nodes | |||
| Positive, n (%) | Negative, n (%) | Total, n (%) | |
| CT results | |||
| Positive | 28 (21.1) TP | 30 (22.6) FP | 58 (43.7) |
| Negative | 21 (15.8) FN | 54 (40.5) TN | 75 (56.3) |
| Total | 49 (36.9) | 84 (63.1) | 133 (100) |
| FDG-PET/CT results | |||
| Positive | 34 (25.6) TP | 11 (8.3) FP | 45 (33.6) |
| Negative | 16 (12) FN | 72 (54.1) TN | 88 (66.1) |
| Total | 50 (37.6) | 83 (62.4) | 133 (100) |
FDG-PET/CT: positron emission tomography with [18F] fluoro-2-deoxy-d-glucose/computed tomography; FN: false negative; FP: false positive; CT: computed tomography; TN: true negative; TP: true positive.
The aim of our study was to define the role of FDG-PET/CT in daily clinical practice, so we analyzed each case of lung cancer in which FDG-PET/CT was performed including all histologies. Most of these results are descriptive. The main findings are that FDG-PET/CT modified the therapeutic approach in 24.4% of cases, and prevented a futile thoracotomy in 5.2%, while making possible a treatment of curative intention in 10.5%. With regard to mediastinal lymph node evaluation, FDG-PET/CT was superior to CT in terms of sensitivity, specificity, positive predictive value and negative predictive value.
The usefulness of FDG-PET/CT in the staging process of NSCLC has been previously demonstrated,5–9 and this examination has become standard.2,10 In order to confirm this in our daily practice, we decided to assess the impact of the use of FDG-PET/CT, the reason for conducting this study.
Our data confirm the limitations of CT alone for the individualization of treatment decisions, given the higher sensitivity, specificity and positive and negative predictive values of FDG-PET/CT in the evaluation of mediastinal disease. We found a negative predictive value of FDG-PET/CT slightly lower than results in other published studies.14–18 This could be due to the high percentage of FN cases with FDG-PET/CT in our series (12%). Of the 16 FN cases, 13 had metastatic mediastinal lymph nodes smaller than 10mm, which is a known cause of FN results in FDG-PET/CT.19
We also confirmed the usefulness of FDG-PET/CT for the detection of non-identified distant metastases, therefore avoiding futile therapeutic interventions. In our series, FDG-PET/CT identified previously unknown distant metastases in 10.1% of cases, similarly to that reported in other series.6
Our data are consistent with previously published results regarding therapeutic modifications.9,14,20–23 In our series, the therapeutic approach was modified in almost a quarter of patients after FDG-PET/CT, which allowed for curative treatment in more than 10%.
Most published data on the clinical impact of FDG-PET/CT in NSCLC focus on differentiating operable from inoperable disease at an early stage, which has shown the usefulness of FDG-PET/CT.6–8,16,18,21 Our study also evaluated the role of FDG-PET/CT in patients with stage III NSCLC. Stage III is associated with low survival rates even after potentially curative treatment with surgery and chemoradiotherapy in select cases.24 Some authors have hypothesized that the inability of conventional staging to detect microscopic disease may be responsible for these poor results,3 suggesting that a more accurate staging could allow for better patient selection and hence better prognosis. In our study, stage III is the most represented early stage, occurring in 90 patients (36.5%). Interestingly, FDG-PET/CT modified the therapeutic approach in more than 40% of these potential candidates for radical chemoradiotherapy. These figures clearly support the use of FDG-PET/CT in this specific subgroup of patients. First, to avoid unnecessary toxicity and costs of irradiation in patients in whom the stage is more severe, and second, to increase the cure rate with an optimal surgical treatment in patients in whom stage was considered less severe. This may result in improved cure rates of these patients through a better choice of treatment.
The low frequency of use of mediastinoscopy for staging among the methods used in our study is due to the availability of EBUS-TBNA in our center.12
One of the concerns about the use of FDG-PET/CT as a standard diagnostic method in patients with lung cancer is the delay that this may bring to completion of the diagnostic workup, especially in locations where this technology is not available at the same center. FDG-PET/CT was available in our center while conducting this study, and all diagnostic techniques were completed within two weeks in most patients.
Knowing whether the inclusion of FDG-PET/CT in the diagnosis has implications for patient survival, due to improved staging, would be interesting. Unfortunately, our study was not designed for this purpose and we therefore cannot provide survival data. One limitation of this study is that we have not analyzed the specific nodal stations invaded in cases classified as N1–N3. Consequently, we cannot specify in which stations misclassifications of FDG-PET/CT occur more often.
Our data support the previous studies suggesting that FDG-PET/CT is essential in the staging process, not only in patients with potentially operable NSCLC, but also in patients with stage III NSCLC.
Author DisclosuresAll authors collaborated in the conduct of the study presented here. A. Taus developed the database and drafted the manuscript. All other authors participated in drafting the manuscript by defining the CT and PET/CT stage of each patient at the multidisciplinary committee and by reviewing the manuscript.
Conflicts of InterestThe authors declare no conflicts of interest.
Please cite this article as: Taus Á, Aguiló R, Curull V, Suárez-Piñera M, Rodríguez-Fuster A, Rodríguez de Dios N, et al. Impacto del 18F-FDG PET/TC en el abordaje terapéutico del cáncer de pulmón no microcítico. Arch Bronconeumol. 2014;50:99–104.