Serum autoantibodies support the diagnosis of interstitial lung disease (ILD) related to systemic autoimmune diseases (SAD-ILD). Nevertheless, their presence in the bronchoalveolar lavage (BAL) has not been explored.
ObjectivesTo demonstrate the presence of autoantibodies in the BAL of ILD patients at onset of clinical evaluation, its relation with serum autoantibodies and to analyze clinical features of patients with autoantibodies in BAL.
MethodsAutoantibodies against extractable nuclear antigens (ENAs) were analyzed by immunoblot in the BAL of 155 patient with suspected diagnosis of ILD and 10 controls.
ResultsSeven ENAs were detected in the BAL of 19 patients (Anti-Ro52, Anti-Ro60, CENP-B, Anti-La, Jo-1, Sm/RNP and Anti-SL70). The most frequent ENA was anti-Ro52 (13 patients; 68,4% of positives ones). Seven patients presented more than one ENAs. Fourteen were diagnosed of SAD-ILD, 3 of interstitial pneumonia with autoimmune features, one of non-specific idiopathic pneumonia and other of silicosis. In 10 cases (52%) IgA autoantibodies were also detected. The autoantibodies observed in BAL were also detected in the serum of 17 patients (90%). There were no significant clinical differences with the patients with SAD-ILD or interstitial pneumonia with autoimmune features with patients with negative BAL.
ConclusionThe study of ENAs in BAL is feasible and can be a useful tool in the ILD initial algorithm, specifically sustaining the suspected diagnosis of SAD-ILD.
Los autoanticuerpos séricos apoyan el diagnóstico de sospecha en la enfermedad intersticial difusa (EPID) asociada a enfermedades autoinmunes sistémicas (EPID-EAS). Su presencia en el lavado broncoalveolar (LBA) no ha sido estudiada.
ObjetivosDemostrar la presencia de autoanticuerpos en el LBA de pacientes con EPID de inicio, compararlos con los resultados del suero y analizar los aspectos clínicos de los pacientes con autoanticuerpos en el LBA.
MétodosSe analizaron autoanticuerpos contra antígenos extraíbles del núcleo (ENA) mediante inmunoblot en el LBA de 155 pacientes con sospecha diagnóstica de EPID y 10 controles.
ResultadosSe detectaron 7 especificidades ENA en el LBA de 19 pacientes (anti-Ro52, anti-Ro60, CENP-B, anti-La, Jo-1, Sm/RNP y anti-SL70), siendo el anti-Ro52 el más frecuente (13 pacientes; 68,4% de los positivos). Siete pacientes presentaron más de una especificidad. Catorce fueron diagnosticados de EPID-EAS, 3 de neumonía intersticial con rasgos autoinmunes, uno de neumonía intersticial no específica idiopática y otro de silicosis. En 10 casos (52%) se detectaron autoanticuerpos de clase IgA en el LBA. Los autoanticuerpos detectados en LBA también se hallaron en el suero de 17 pacientes (90%). No hubo diferencias clínicas significativas entre los pacientes con autoanticuerpos en LBA con respecto a aquellos con EPID-EAS o neumonía intersticial con rasgos autoinmunes con LBA negativo.
ConclusiónEl estudio de ENA en LBA es factible y puede ser una herramienta útil en el algoritmo inicial en la EPID, concretamente, para apoyar el diagnóstico de sospecha de la EPID-EAS.
The heterogeneity of the different entities that constitute diffuse interstitial lung disease (ILD) means that a sequential and systematized study with a multidisciplinary diagnostic approach1–3 is required to achieve a definitive diagnosis. Several factors should be considered in the initial presentation of a patient with diffuse ILD, as these entities may share similar clinical, functional or radiological characteristics.2 In fact, in previously undiagnosed ILD, an underlying cause, such as systemic autoimmune diseases (SAD) must always be ruled out.
For this reason, the application of tools that add useful information to the ILD diagnostic algorithm is essential. Autoantibodies are primary biomarkers in the diagnosis of SAD-ILD.4 These antibodies may support a suspected diagnosis and, in some cases, help predict disease progression.5 Autoantibodies are usually studied in serum, but they can also be isolated in other biological matrices and appear to synthesize in situ in multiple tissues.6,7 Starting from this premise and taking into account the description of germinal centers in the pulmonary epithelium of patients with SAD,8 we hypothesized that autoantibodies might also be detected in bronchoalveolar lavage (BAL) in patients with ILD. The main objective of the study was to identify autoantibodies to extractable nuclear antigens (ENA) in the BAL of patients with suspected ILD. A secondary objective was to associate the presence of these autoantibodies in BAL and serum with patients’ clinical, radiological and respiratory parameters.
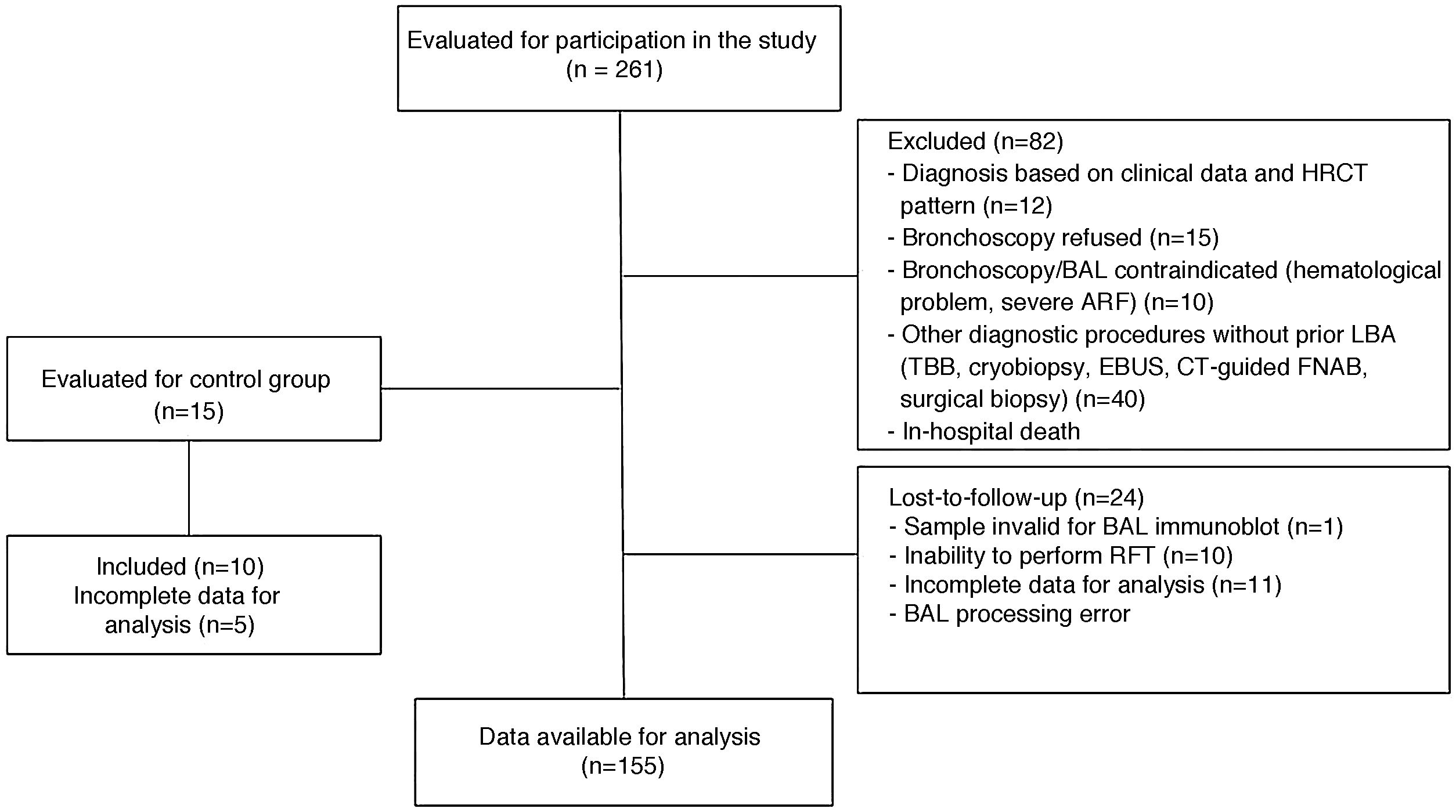
Materials and methodsPatient inclusion and BAL sample collectionThis was a prospective observational longitudinal study. The inclusion criteria were as follows: patients ≤ 80 years with initial suspected ILD seen in the ILD clinic or admitted for ILD to 2 university hospitals between the years 2014 and 2019. Patients with a diagnosis of SAD and no previous evidence of ILD were also assessed for inclusion. Control BAL samples were collected from patients who required fiberoptic bronchoscopy examination for minor hemoptysis who showed no evidence of parenchymal disease on high-resolution computed tomography. Fig. 1 is a flowchart summarizing the inclusion and exclusion of study patients.
Clinical, radiological and pathological parameters were reviewed by the multidisciplinary ILD committee of the participating centers. Diagnoses of ILD and interstitial pneumonia with autoimmune features (IPAF) were established according to ATS/ERS consensus guidelines.9–12 The diagnosis of SAD was established according to ACR/EULAR criteria.13–17
The following variables were included: demographic data, smoking history, personal, family and occupational history, environmental exposure, and clinical and radiological functional characteristics.
Forced spirometry, lung volume measurement, diffusing capacity of the lung for carbon monoxide, and 6-min walk tests were performed according to international recommendations, using the reference values published by Roca et al.18–21 The Composite Physiologic Index was calculated using the formula described by Wells et al.22
BAL was obtained by flexible fiberoptic bronchoscopy, using a standard, previously described technique.23 Briefly, the tip of the flexible bronchoscope was introduced into the segmental/subsegmental bronchus of the parenchymal zone with the greatest involvement, usually the middle lobe or the lingula. Three 50 mL aliquots of serum at body temperature for best patient tolerance were collected. The third aliquot was used to study autoantibodies. Centrifuged specimens were stored at −20 °C in 1.5 mL aliquots until use.
The study was approved by the ethics committee of each hospital (Registry No. PI-20-209 and 2018501) and all study patients signed informed consent. Patient rights were protected according to the terms of the Declaration of Helsinki.
Determination of autoantibodies in serumAntinuclear antibodies (ANA) were determined by indirect immunofluorescence (IIF) on HEp-2 cells (INOVA Diagnostics Inc., Barcelona, Spain). ENAs were studied by CLIA, QUANTA Flash® ENA 7 (Jo-1, Scl-70, SSB, Ro-52, SSA, Sm and Sm/RNP) and QUANTA Flash® Centromere (CENP-B) (INOVA Diagnostics Inc., Barcelona, Spain). Positive sera were verified by Euroline ANA Profile immunoblot (Euroimmun, Lübeck, Germany).
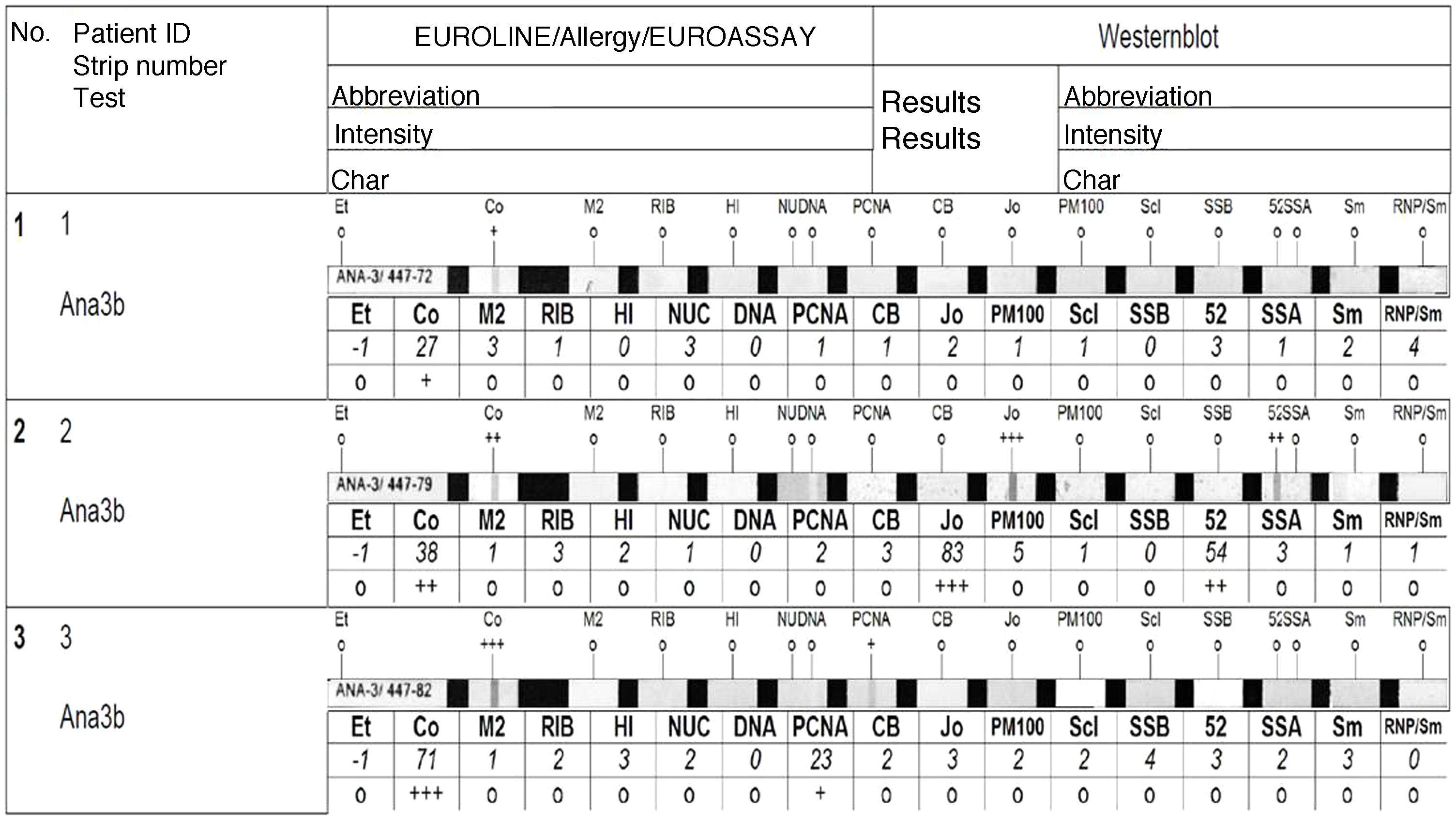
Determination of autoantibodies in BALENAs were studied using the Euroline ANA Profile immunoblot in a EUROBlotMaster automatizer (both from Euroimmun, Lübeck, Germany). The results were scanned and interpreted using the EUROLineScan system software (Euroimmun, Lübeck, Germany), which provides semi-quantitative results. This method can be used for simultaneous detection of the following antibodies: AMA-2, ribosomal-p, histones, nucleosome, dsADN, PCNA, CENP-B, Jo-1, PM100, Scl-70, SSB, Ro-52, SSA, Sm and Sm/RNP. Two modifications were applied to the supplier’s protocol:
- 1)
The optimal dilution was established by serial dilutions. Some samples maintained positivity at a dilution equivalent to that of serum (1/100); however, less intense positivities may be missed at dilutions greater than 1/10, so we decided that samples should be processed undiluted.
- 2)
IgA and IgG conjugates were used in parallel.
To avoid categorizing staining artifacts as positives, we decided that only the bands classified by the analysis software as medium (++) or high (+++) intensity would be considered positive (Fig. 2).
Positive samples were confirmed by IIF on HEP-2 cells with a dilution of 1/1 (INOVA Diagnostics Inc., Barcelona, Spain). ANA patterns were reported according to ICAP24 nomenclature.
Statistical analysisA descriptive analysis was conducted. The results of the quantitative variables were expressed as mean ± standard deviation or median with interquartile range. Percentages and absolute frequencies were used for qualitative variables. In the case of quantitative or qualitative ordinal variables, a test of normal distribution (Kolmogorov–Smirnov test) was applied first. When variables were normally distributed, a comparison of means (Student’s t for independent samples) was performed. When the distribution was not normal, the non-parametric equivalent (Mann–Whitney U for independent samples) was used. In the case of qualitative variables, contingency tables and Pearson’s chi-square statistic were used. The dependent variable was the presence or absence of ENA in BAL. The remaining parameters were the independent variables. A p-value < 0.05 was considered statistically significant. Analyses were performed using the SPSS® 14.0 statistical package (IBM SPSS Statistics, Armonk, NY, USA).
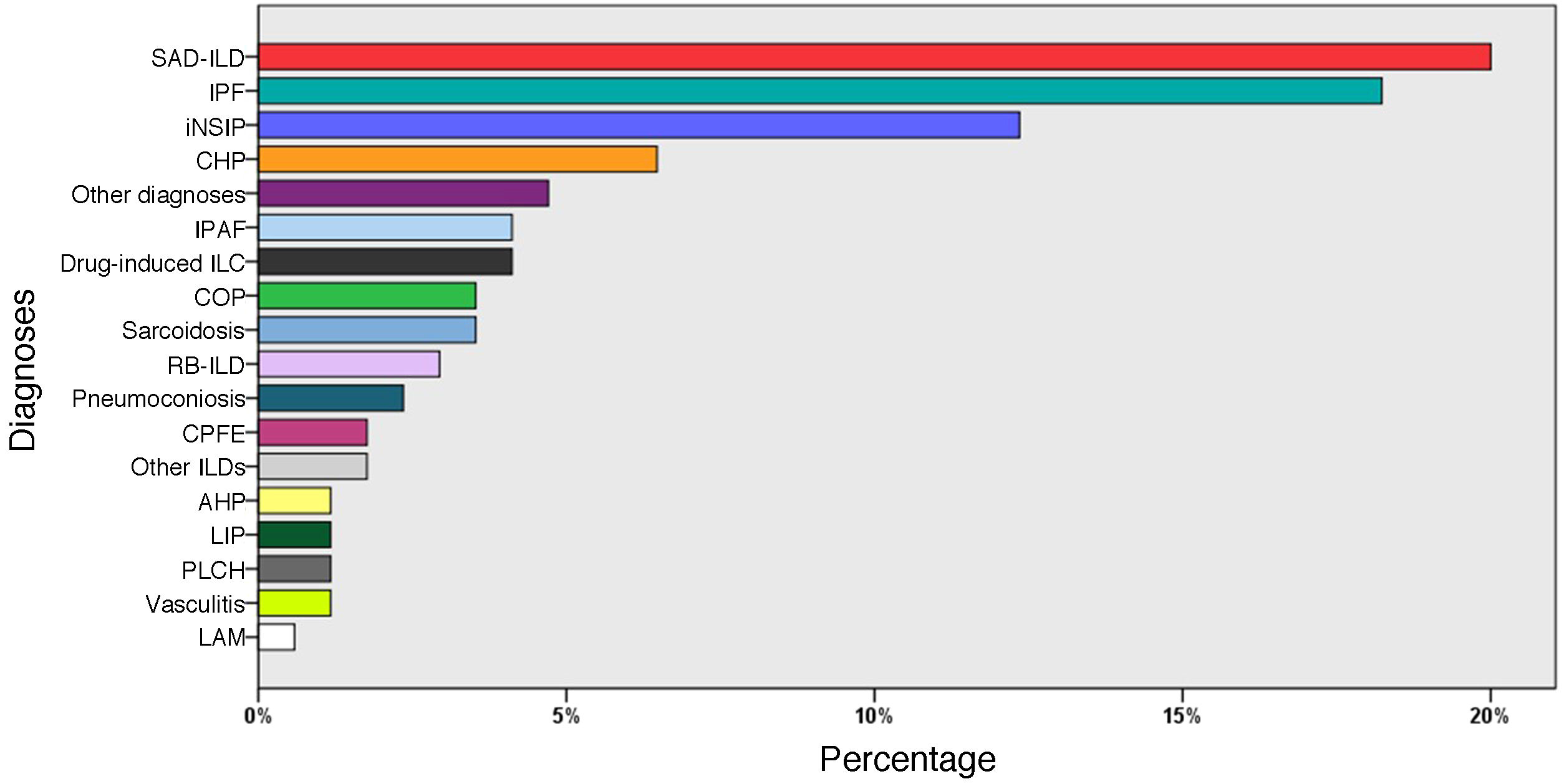
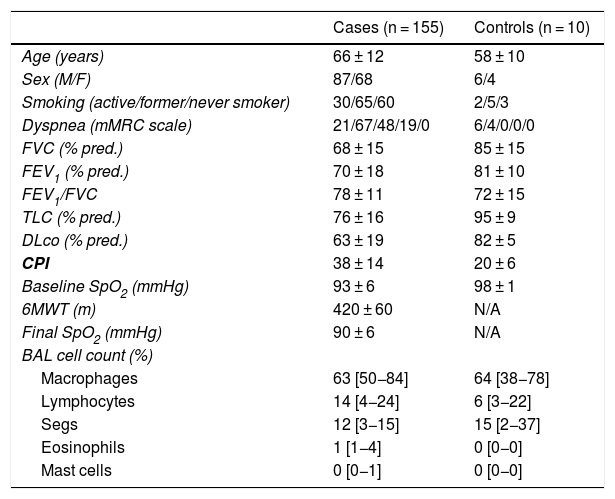
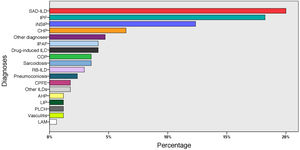
ResultsPatient characteristicsIn total, 155 patients were included, of whom 87 were male (56.1%), with a mean age of 66 ± 12 years, and 10 controls with a mean age of 58 ± 10 years, of whom 6 were male (60%). Clinical and functional data, and BAL cell count data for these patients are summarized in Table 1. The most frequent diagnoses were: SAD-ILD (36 patients; 23%), idiopathic pulmonary fibrosis (31; 20%), and idiopathic non-specific interstitial pneumonia (20; 13.5%) (Fig. 3).
Demographic and functional data and cell count in bronchoalveolar lavage of study subjects and controls.
| Cases (n = 155) | Controls (n = 10) | |
|---|---|---|
| Age (years) | 66 ± 12 | 58 ± 10 |
| Sex (M/F) | 87/68 | 6/4 |
| Smoking (active/former/never smoker) | 30/65/60 | 2/5/3 |
| Dyspnea (mMRC scale) | 21/67/48/19/0 | 6/4/0/0/0 |
| FVC (% pred.) | 68 ± 15 | 85 ± 15 |
| FEV1 (% pred.) | 70 ± 18 | 81 ± 10 |
| FEV1/FVC | 78 ± 11 | 72 ± 15 |
| TLC (% pred.) | 76 ± 16 | 95 ± 9 |
| DLco (% pred.) | 63 ± 19 | 82 ± 5 |
| CPI | 38 ± 14 | 20 ± 6 |
| Baseline SpO2 (mmHg) | 93 ± 6 | 98 ± 1 |
| 6MWT (m) | 420 ± 60 | N/A |
| Final SpO2 (mmHg) | 90 ± 6 | N/A |
| BAL cell count (%) | ||
| Macrophages | 63 [50−84] | 64 [38−78] |
| Lymphocytes | 14 [4−24] | 6 [3−22] |
| Segs | 12 [3−15] | 15 [2−37] |
| Eosinophils | 1 [1−4] | 0 [0−0] |
| Mast cells | 0 [0−1] | 0 [0−0] |
% pred.: percentage of predicted value; 6MWT: 6-min walk test; Baseline SpO2: arterial oxygen saturation at the start of 6MWT; CPI: Composite Physiologic Index; DLco: diffusing capacity of the lung for carbon monoxide; F: female; FEV1/FVC: forced expiratory volume in 1 s/forced vital capacity expressed as an absolute percentage; FEV1: forced expiratory volume in 1 s; Final SpO2: arterial oxygen saturation at the end of 6MWT; FVC: forced vital capacity; M: male; mMRC: modified Medical Research Council dyspnea scale; N/A: not applicable; TLC: total lung capacity.
Results of the quantitative variables were expressed as mean ± standard deviation or median (interquartile range).
Diagnoses of all patients (n = 155).
AHP: acute hypersensitivity pneumonitis; CHP: chronic hypersensitivity pneumonitis; COP: cryptogenic organizing pneumonia; CPFE: combination of pulmonary fibrosis and emphysema; ILD: interstitial lung disease; iNSIP: idiopathic non-specific interstitial pneumonia; IPAF: interstitial pneumonia with autoimmune features; IPF: idiopathic pulmonary fibrosis; LAM: lymphangioleiomyomatosis; LIP: lymphocytic interstitial pneumonia; PLCH: pulmonary Langerhans cell histiocytosis; RB-ILD: respiratory bronchiolitis associated with ILD; SAD-ILD: systemic autoimmune disease-associated ILD.
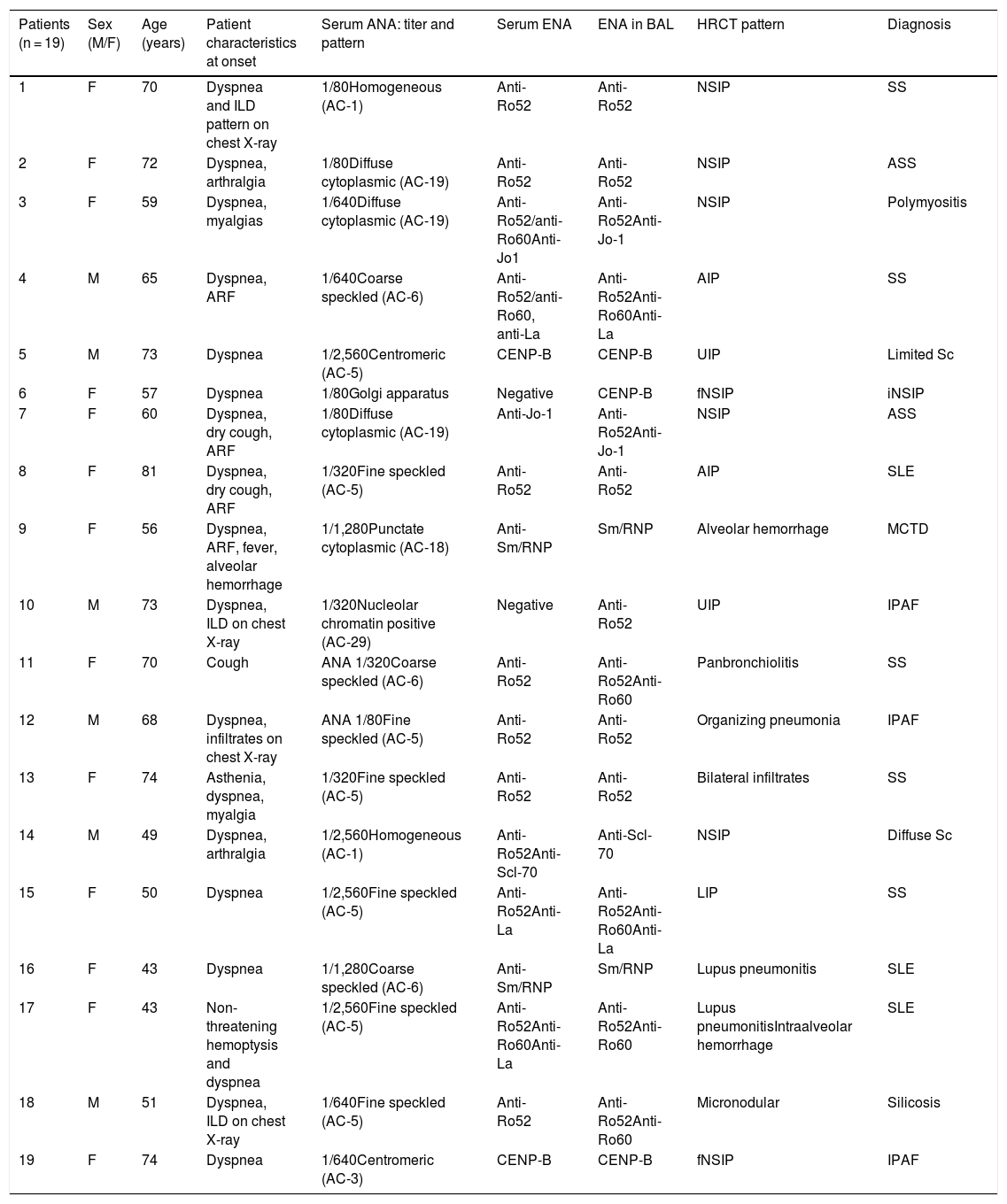
Seven different specificities (28 in total) were detected in the BAL of 19 patients, representing 39.5% of all patients with a final diagnosis of SAD-ILD or IPAF. Mean age was 60 ± 12 years and the group consisted of 14 women (73.6%) and 5 men (26.3%). Fourteen patients were diagnosed with SAD-ILD, 1 with idiopathic non-specific interstitial pneumonia, 3 with IPAF, and 1 with silicosis. Almost one third of the cases (5; 26.3%) had low baseline serum ANA titers (1/80). In the group of patients with SAD-ILD or IPAF, 10 had a radiological pattern of early-onset ILD. Four patients had a previous diagnosis of SAD, and ILD occurred as an acute complication during their disease (bronchiolitis in a Sjögren’s syndrome patient or lupus pneumonitis in patients with systemic lupus erythematosus). Table 2 summarizes the clinical characteristics of these patients at study baseline, serum and BAL autoantibodies, high-resolution computed tomography patterns, and final diagnosis. When clinical and functional data and BAL cell count were compared in SAD-ILD or IPAF patients with positive autoantibodies in BAL vs. those without, no statistically significant differences were found (Table 3). No ENA in BAL or other forms of ILD not associated with SAD were observed in the control group.
Clinical characteristics and autoantibodies in serum and in bronchoalveolar lavage of patients with positive extractable nuclear antibodies.
| Patients (n = 19) | Sex (M/F) | Age (years) | Patient characteristics at onset | Serum ANA: titer and pattern | Serum ENA | ENA in BAL | HRCT pattern | Diagnosis |
|---|---|---|---|---|---|---|---|---|
| 1 | F | 70 | Dyspnea and ILD pattern on chest X-ray | 1/80Homogeneous (AC-1) | Anti-Ro52 | Anti-Ro52 | NSIP | SS |
| 2 | F | 72 | Dyspnea, arthralgia | 1/80Diffuse cytoplasmic (AC-19) | Anti-Ro52 | Anti-Ro52 | NSIP | ASS |
| 3 | F | 59 | Dyspnea, myalgias | 1/640Diffuse cytoplasmic (AC-19) | Anti-Ro52/anti-Ro60Anti-Jo1 | Anti-Ro52Anti-Jo-1 | NSIP | Polymyositis |
| 4 | M | 65 | Dyspnea, ARF | 1/640Coarse speckled (AC-6) | Anti-Ro52/anti-Ro60, anti-La | Anti-Ro52Anti-Ro60Anti-La | AIP | SS |
| 5 | M | 73 | Dyspnea | 1/2,560Centromeric (AC-5) | CENP-B | CENP-B | UIP | Limited Sc |
| 6 | F | 57 | Dyspnea | 1/80Golgi apparatus | Negative | CENP-B | fNSIP | iNSIP |
| 7 | F | 60 | Dyspnea, dry cough, ARF | 1/80Diffuse cytoplasmic (AC-19) | Anti-Jo-1 | Anti-Ro52Anti-Jo-1 | NSIP | ASS |
| 8 | F | 81 | Dyspnea, dry cough, ARF | 1/320Fine speckled (AC-5) | Anti-Ro52 | Anti-Ro52 | AIP | SLE |
| 9 | F | 56 | Dyspnea, ARF, fever, alveolar hemorrhage | 1/1,280Punctate cytoplasmic (AC-18) | Anti-Sm/RNP | Sm/RNP | Alveolar hemorrhage | MCTD |
| 10 | M | 73 | Dyspnea, ILD on chest X-ray | 1/320Nucleolar chromatin positive (AC-29) | Negative | Anti-Ro52 | UIP | IPAF |
| 11 | F | 70 | Cough | ANA 1/320Coarse speckled (AC-6) | Anti-Ro52 | Anti-Ro52Anti-Ro60 | Panbronchiolitis | SS |
| 12 | M | 68 | Dyspnea, infiltrates on chest X-ray | ANA 1/80Fine speckled (AC-5) | Anti-Ro52 | Anti-Ro52 | Organizing pneumonia | IPAF |
| 13 | F | 74 | Asthenia, dyspnea, myalgia | 1/320Fine speckled (AC-5) | Anti-Ro52 | Anti-Ro52 | Bilateral infiltrates | SS |
| 14 | M | 49 | Dyspnea, arthralgia | 1/2,560Homogeneous (AC-1) | Anti-Ro52Anti-Scl-70 | Anti-Scl-70 | NSIP | Diffuse Sc |
| 15 | F | 50 | Dyspnea | 1/2,560Fine speckled (AC-5) | Anti-Ro52Anti-La | Anti-Ro52Anti-Ro60Anti-La | LIP | SS |
| 16 | F | 43 | Dyspnea | 1/1,280Coarse speckled (AC-6) | Anti-Sm/RNP | Sm/RNP | Lupus pneumonitis | SLE |
| 17 | F | 43 | Non-threatening hemoptysis and dyspnea | 1/2,560Fine speckled (AC-5) | Anti-Ro52Anti-Ro60Anti-La | Anti-Ro52Anti-Ro60 | Lupus pneumonitisIntraalveolar hemorrhage | SLE |
| 18 | M | 51 | Dyspnea, ILD on chest X-ray | 1/640Fine speckled (AC-5) | Anti-Ro52 | Anti-Ro52Anti-Ro60 | Micronodular | Silicosis |
| 19 | F | 74 | Dyspnea | 1/640Centromeric (AC-3) | CENP-B | CENP-B | fNSIP | IPAF |
AIP: acute interstitial pneumonia; ANA: antinuclear antibodies; Anti-Scl-70: anti-topoisomerase antibodies; ARF: acute respiratory failure; ASS: antisynthetase syndrome; BAL: bronchoalveolar lavage; CENP-B: anti-centromere B protein antibodies; ENA: extractable nuclear antibody; F: female; fNSIP: fibrotic non-specific interstitial pneumonia; HRCT: high-resolution computed tomography; ILD: interstitial lung disease; iNSIP: idiopathic non-specific interstitial pneumonia; IPAF: interstitial pneumonia with autoimmune features; LIP: lymphocytic interstitial pneumonia; M: male; MCTD: mixed connective tissue disease; NSIP: non-specific interstitial pneumonia; Sc: scleroderma; SLE: systemic lupus erythematosus; SS: Sjögren’s syndrome; UIP: usual interstitial pneumonia.
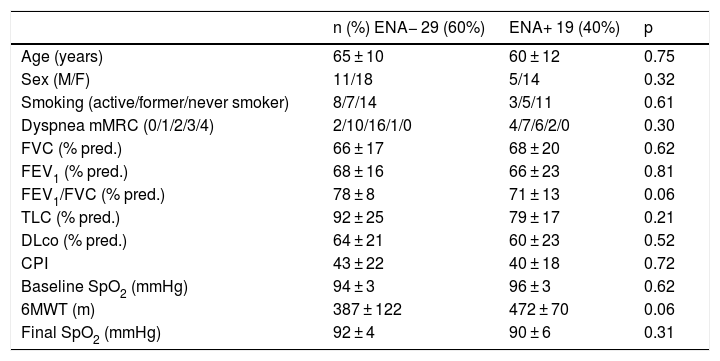
Comparison of functional variables in patients with SAD-ILD and IPAF according to the presence of extractable nuclear antibodies in bronchoalveolar lavage.
| n (%) ENA− 29 (60%) | ENA+ 19 (40%) | p | |
|---|---|---|---|
| Age (years) | 65 ± 10 | 60 ± 12 | 0.75 |
| Sex (M/F) | 11/18 | 5/14 | 0.32 |
| Smoking (active/former/never smoker) | 8/7/14 | 3/5/11 | 0.61 |
| Dyspnea mMRC (0/1/2/3/4) | 2/10/16/1/0 | 4/7/6/2/0 | 0.30 |
| FVC (% pred.) | 66 ± 17 | 68 ± 20 | 0.62 |
| FEV1 (% pred.) | 68 ± 16 | 66 ± 23 | 0.81 |
| FEV1/FVC (% pred.) | 78 ± 8 | 71 ± 13 | 0.06 |
| TLC (% pred.) | 92 ± 25 | 79 ± 17 | 0.21 |
| DLco (% pred.) | 64 ± 21 | 60 ± 23 | 0.52 |
| CPI | 43 ± 22 | 40 ± 18 | 0.72 |
| Baseline SpO2 (mmHg) | 94 ± 3 | 96 ± 3 | 0.62 |
| 6MWT (m) | 387 ± 122 | 472 ± 70 | 0.06 |
| Final SpO2 (mmHg) | 92 ± 4 | 90 ± 6 | 0.31 |
% pred.: percentage of predicted value; 6MWT: 6-min walk test; baseline SpO2: arterial oxygen saturation at the start of 6MWT; CPI: Composite Physiologic Index; DLco: diffusing capacity of the lung for carbon monoxide; F: female; FEV1/FVC: forced expiratory volume in 1 s/forced vital capacity expressed as an absolute percentage; FEV1: forced expiratory volume in 1 s; Final SpO2: arterial oxygen saturation at the end of 6MWT; FVC: forced vital capacity; IPAF: interstitial pneumonia with autoimmune features; M: male; mMRC: modified Medical Research Council dyspnea scale; SAD-ILD: systemic autoimmune disease-associated interstitial lung disease; TLC: total lung capacity.
The most frequent ENA was anti-Ro52 (13 patients; 68.4% of those positive), followed by anti-Ro60 (5; 26.3%), anti-CENP-B (3; 15.8%), anti-La, Jo-1, and Sm/RNP (2; 10.5%), and anti-Scl-70 (1; 5.3%). Seven patients (36.8%) were positive for more than one ENA. Band intensity was high in all patients, including those with low ANA titer.
Comparison of patients with positive ENA in serum and BALSeventeen patients (90%) with autoantibodies in BAL showed the same antibodies in serum. In 3 patients (7, 11 and 15), specificities were observed in BAL that were not detected in serum. However, 1 patient presented anti-Ro52 autoantibodies in BAL with negative serum. This patient died of lung cancer months after the appearance of the autoantibodies in BAL, so seroconversion could not be studied. Nor could positive seroconversion be confirmed in a patient with CENP-B in BAL and a diagnosis of idiopathic non-specific interstitial pneumonia. In contrast, 3 patients showed fewer specificities in BAL than in serum (3, 14, and 17).
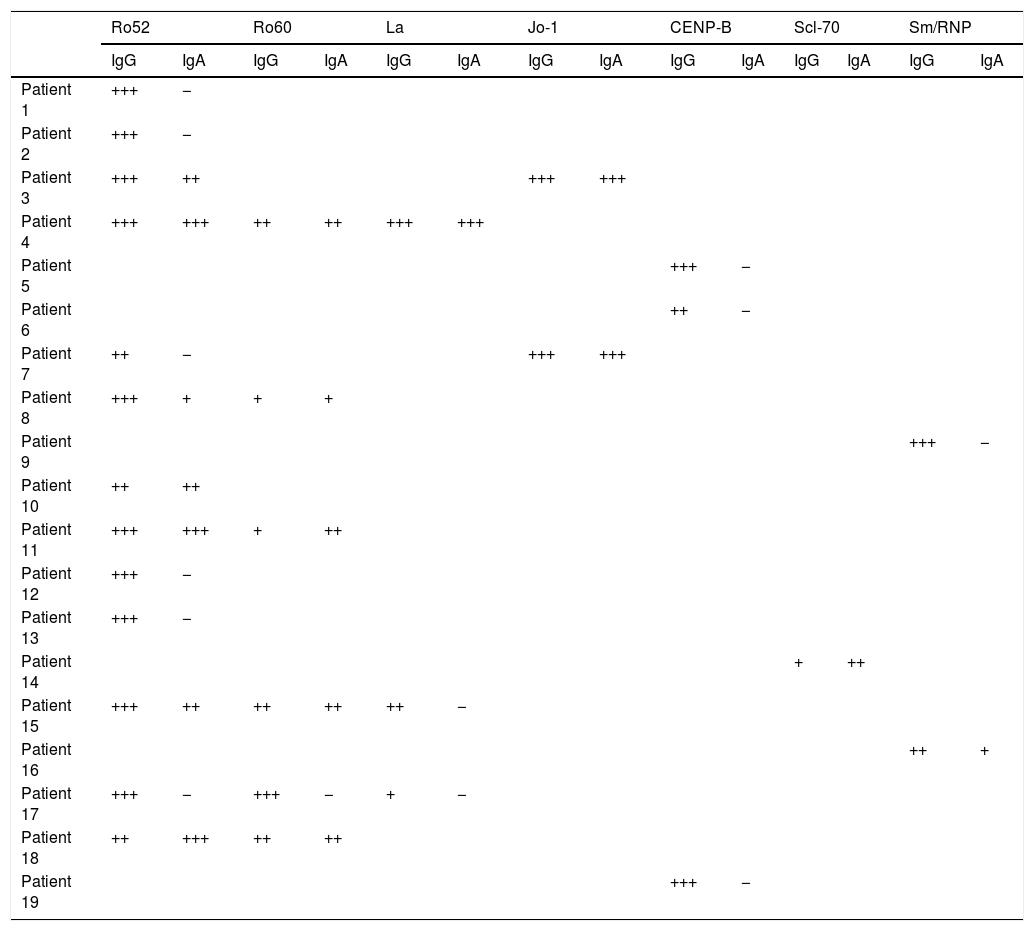
Comparison of patients with IgA and IgG class ENA in BALMore than half (10/19) of patients with IgG class antibodies in BAL had IgA class antibodies against the same specificities. Of the 28 specificities identified, 13 showed IgG antibodies only. In 2 cases, IgA intensity was higher than IgG intensity. In one of them, the intensity of the IgG band was low, so according to the cut-off point established for the study, if the IgA antibody study had not been performed this patient would have been considered negative (Table 4).
Results of immunoblot of extractable nuclear antibodies in bronchoalveolar lavage (IgG; IgA conjugates).
| Ro52 | Ro60 | La | Jo-1 | CENP-B | Scl-70 | Sm/RNP | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| IgG | IgA | IgG | IgA | IgG | IgA | IgG | IgA | IgG | IgA | IgG | IgA | IgG | IgA | |
| Patient 1 | +++ | − | ||||||||||||
| Patient 2 | +++ | − | ||||||||||||
| Patient 3 | +++ | ++ | +++ | +++ | ||||||||||
| Patient 4 | +++ | +++ | ++ | ++ | +++ | +++ | ||||||||
| Patient 5 | +++ | − | ||||||||||||
| Patient 6 | ++ | − | ||||||||||||
| Patient 7 | ++ | − | +++ | +++ | ||||||||||
| Patient 8 | +++ | + | + | + | ||||||||||
| Patient 9 | +++ | − | ||||||||||||
| Patient 10 | ++ | ++ | ||||||||||||
| Patient 11 | +++ | +++ | + | ++ | ||||||||||
| Patient 12 | +++ | − | ||||||||||||
| Patient 13 | +++ | − | ||||||||||||
| Patient 14 | + | ++ | ||||||||||||
| Patient 15 | +++ | ++ | ++ | ++ | ++ | − | ||||||||
| Patient 16 | ++ | + | ||||||||||||
| Patient 17 | +++ | − | +++ | − | + | − | ||||||||
| Patient 18 | ++ | +++ | ++ | ++ | ||||||||||
| Patient 19 | +++ | − | ||||||||||||
Crosses indicate the degree of intensity of the bands: weak (+); intermediate (++); strong (+++).
Weak intensities were interpreted as negative.
In all cases, the IIF pattern observed was consistent with that associated with the specificities obtained. Ro52 and Ro60 were not associated with any ANA pattern, so not all sera were positive.
DiscussionThis study shows the presence of autoantibodies in the BAL of patients with ILD during the diagnostic process. As far as we know, this is a new finding not previously described. Positivities were detected for 7 different ENA specificities, anti-Ro52 being the most frequent. BAL studies revealed high baseline levels of autoantibodies in all patients, regardless of the ANA titer in serum.
ENA is detected in BAL using an inexpensive, widely available technique. Our results show that these determinations, combined with the serum study, can provide additional information for screening ILD of autoimmune origin. Detection using different techniques (immunoblot, IIF) confirms that this is a reproducible strategy that could be incorporated into routine diagnostic procedures.
BAL analysis is widely used in the diagnostic algorithm of ILDs,2,25 and can reflect the humoral and immunobiological mechanisms that occur in the lung in a more reliable and predictable manner than peripheral blood.26 However, information on biomarkers of ILD in BAL is still limited. Most of the molecules studied are associated with the aberrant activation of epithelial cells or alveolar macrophages and, to a lesser extent, a modified immune response.8,27,28
The appearance of autoantibodies in serum can occur years before the first clinical manifestations.1,12 Identifying the organ in which autoimmunity begins could open the door to new diagnostic or therapeutic strategies. In this series, all patients were studied at the onset of pulmonary manifestations (clinical or radiological), so we propose that the appearance of autoantibodies in BAL could serve as an early marker of ILD development in patients with SAD. In contrast with idiopathic interstitial pneumonias, the study of autoantibodies in SAD-ILD is essential, since their presence is directly related to pathogenesis and tissue damage.28,29
Three patients showed autoantibodies in BAL that were undetectable in serum, while 3 others had serum autoantibodies that were not detectable in BAL. These disparities may reflect differences in local response. Autoantibodies detected in BAL could only derive directly from the lung, while those found only in serum could be synthesized in other tissues. These findings support the need for studying both matrices in parallel.
The fact that no positive results were obtained in another group of common ILDs, including idiopathic pulmonary fibrosis or hypersensitivity pneumonitis, may also indicate that the detection of autoantibodies in BAL is highly indicative of underlying SAD. In this regard, it should be noted that 1 patient with silicosis in the group with positive BAL also had positive serum. Although this result may seem contradictory, exposure to silica dust has been described as a risk factor for the development of several types of SAD.30,31 It should also be noted that autoantibodies were not detected in the control group or in patients with diseases that could be confused clinically or radiologically with onset ILD (congestive heart failure, infection in immunosuppressed patients, and others).2
When the group of patients with positive BAL was compared with those diagnosed with SAD or IPAF and negative BAL, we found no significant differences in clinical or functional parameters. Nevertheless, we believe that it might be interesting to analyze the progress of these patients to assess the role of autoantibodies in the prognosis of these diseases.
The ENA most frequently detected in the BAL of these patients was anti-Ro52. Anti-Ro52 is found in several SADs, including Sjögren’s syndrome, systemic lupus erythematosus, scleroderma, and others. Its presence in serum has been associated with a higher prevalence of ILD32 and with a worse prognosis when it coexists with myositis-specific autoantibodies.32–34 In our series, patients with positive anti-Ro52 in BAL were mostly women and presented different diagnoses and widely varying clinical and radiological manifestations, ranging from asymptomatic cases that were investigated due to a radiological pattern consistent with ILD to patients whose initial manifestations included acute respiratory failure, alveolar hemorrhage, or signs of acute interstitial pneumonia on high-resolution computed tomography. The appearance of ILD is of interest in patients with systemic lupus erythematosus, in whom the prevalence of this clinical finding is generally lower than in other SAD.13 However, when it occurs, it can present as an acute complication, as occurred in our cases. These results are in line with studies suggesting that patients with positive anti-Ro52 in serum may have a highly heterogeneous clinical phenotype.35
In the group of patients with positive BAL, autoantibodies with high specificity and clinical relevance were also detected, particularly Jo-1, SM/RNP and Scl-70. These autoantibodies are included in the classification criteria for various SADs, such as antisynthase syndrome, mixed connective tissue disease and diffuse sclerosis, respectively. These markers can also provide prognostic information.29,35,36
No standardized protocols are currently available for immunological studies in ILD screening. Many laboratories study only ANA by IIF, and if these are negative, ENA specificities are not investigated. In our study, the isolation of ENAs in both BAL and serum also supports the recommendations made by other authors to expand serum autoantibody testing to other matrices. In addition to myositis markers, anti-Ro52, that is highly prevalent and lacks a characteristic ANA pattern in serum (it may be negative), should be routinely studied in these patients.4,29,35,36
Another unexpected finding is the higher prevalence of IgG antibodies over IgA antibodies. IgA antibodies are dominant in mucous membranes and the concentration of IgG in the lung is very low. However, we have found patients who are positive for IgG antibodies and negative for IgA antibodies. This apparent incongruity could be explained by a lack of sensitivity of the polymeric IgA conjugate. It could also be because the polymeric IgA antibodies are strongly bound to the mucous layer and do not penetrate the liquid phase that is collected in the BAL. In peripheral blood, IgA isotype immunoglobulins have a half-life of a few days, so they are less prevalent.37 For this reason, it is highly unlikely that the IgA antibodies detected in our patients are a result of contamination with peripheral blood. Likewise, despite being scant, their mere presence demonstrates synthesis in situ. This would support the hypothesis posited in other studies that, in at least some patients, the lung is an organ involved in autoimmunity mechanisms.26,38 Our results, that open the door to future research, would need to be replicated in functional studies.
This study has some limitations. First, it has no validation cohort to determine the predictive value of the test. Since the primary objective of the study was to detect the presence of autoantibodies in BAL, our findings answer the research question. Second, the patients in the control group were not healthy volunteers and some had a history of smoking. Although BAL is a minimally invasive technique, it can be an unpleasant experience for some patients, and, as such, is difficult to justify in a healthy population. Finally, although the study includes patients from 2 centers, the group of patients with SAD is too small for adequate statistical analysis. However, we assume that it reflects real clinical practice in ILD units in our setting, where a high percentage of patients are referred for follow-up of a previous diagnosis of SAD.
In summary, this study shows that autoantibodies can be detected in BAL in patients with early-onset ILD using conventional techniques. The parallel analysis of autoantibodies in serum adds valuable information to the diagnostic algorithm for these diseases. This approach may be useful for the early identification of interstitial involvement in SAD patients in the different possible scenarios.
FundingThis study has been funded by the following grants: research grant from the Spanish Society of Pulmonology and Thoracic Surgery (SEPAR): project 020, EPID Futuro 2014 grant, SEPAR international research fellowship 2018 (Karina Portillo), FIS P16/0216 P16/0216 (Marian García-Nuñez), and SEPAR‐Boehringer EPID 2018 Grant for Young Researchers (Laura Rodríguez Pons).
Conflict of interestThe authors state that they have no conflict of interest.
Please cite this article as: Salvador-Corres I, Quirant-Sanchez B, Teniente-Serra A, Centeno C, Moreno A, Rodríguez-Pons L, et al. Detección de autoanticuerpos en el lavado broncoalveolar en pacientes con enfermedad pulmonar intersticial difusa. Arch Bronconeumol. 2021;57:351–358.