Combined pulmonary fibrosis and emphysema (CPFE) was first described by Cottin et al. in 2005 as a distinct entity characterised by upper lobe emphysema and lower lobe fibrosis on high-resolution computed tomography (HRCT) of the chest resulting in preserved lung volumes and markedly impaired carbon monoxide diffusing capacity of the lung (DLCO).1 Since then, a wide heterogeneity of radiological features of CPFE has been described and a consensual definition or a standard diagnostic algorithm has not yet been reached.2–9 From a clinical standpoint, it is sometimes a challenge to distinguish emphysema from fibrosis and whether both conditions coexist.10–11 Pulmonary function tests may be a useful adjunct tool in these uncertain cases. In view of that, this study aims to identify functional features that can help to distinguish CPFE from idiopathic pulmonary fibrosis (IPF) and/or emphysema.
We performed a retrospective analysis of patients with CPFE and IPF followed in an Interstitial Lung Diseases (ILDs) Outpatient Clinic between 2011 and 2016. A convenience sample of patients with emphysema attending a Chronic Obstructive Pulmonary Disease (COPD) Outpatient Clinic was obtained.
The diagnosis of CPFE and IPF was established by multidisciplinary discussion. The diagnostic criteria for IPF were applied according to the 2011 ATS/ERS/JRS/ALAT guidelines for diagnosis and management of IPF. CPFE was only considered in patients with emphysema involving at least 10% of the whole lung. The diagnosis of emphysema was established by visual assessment of HRCT.
Demographics, pulmonary function test results and evidence of pulmonary hypertension (PH) at diagnosis were reviewed from medical records. Only data regarding pulmonary function tests and echocardiograms performed up to 6 months within the baseline HRCT was included. PH was defined as a systolic arterial pulmonary pressure≥40mmHg plus central venous pressure on echocardiogram.
Kruskal–Wallis test was used to compare pulmonary function test results at diagnosis between groups. Statistical significance was set at p<0.05.
We identified 14 patients with CPFE, 49 with IPF (35 UIP pattern; 16 possible/inconsistent UIP, who underwent lung biopsy) and 57 with emphysema. Overall, 85% were men with a median age at diagnosis of 68.5 years (IQR 61.0–75.0) and 21% were never smokers. All CPFE patients were male and all but two were current or ex-smokers. The median age at diagnosis was 66.5 years (IQR 60.8–77.0) and 30.8% of CPFE patients had PH at diagnosis. No significant differences between patients with CPFE and patients with IPF or emphysema were found regarding gender, age at diagnosis, smoking history or prevalence of PH at diagnosis.
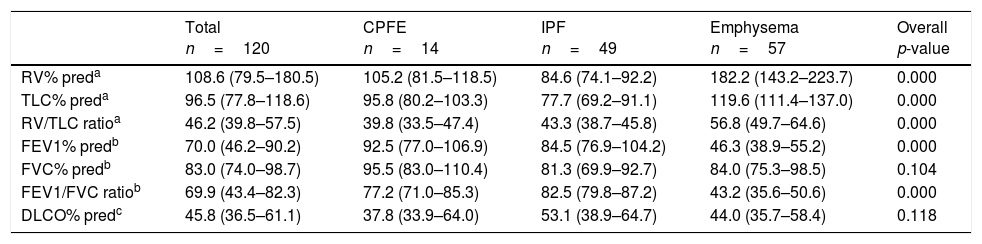
The pulmonary function test results at diagnosis are presented in Table 1. Compared to patients with emphysema, those with CPFE had lower RV% pred (p 0.005), TLC% pred (p 0.012) and RV/TLC ratio (p 0.001), and higher FEV1% (p<0.001) and FEV1/FVC ratio (p<0.001; all pairwise comparisons). FVC% pred (p 0.104) and DLCO% pred (p 0.118) did not differ between the three groups of patients. No significant differences in analysed pulmonary function test parameters were found comparing patients with CPFE and IPF.
Comparison of pulmonary function test results at diagnosis between the three groups of patients.
| Total n=120 | CPFE n=14 | IPF n=49 | Emphysema n=57 | Overall p-value | |
|---|---|---|---|---|---|
| RV% preda | 108.6 (79.5–180.5) | 105.2 (81.5–118.5) | 84.6 (74.1–92.2) | 182.2 (143.2–223.7) | 0.000 |
| TLC% preda | 96.5 (77.8–118.6) | 95.8 (80.2–103.3) | 77.7 (69.2–91.1) | 119.6 (111.4–137.0) | 0.000 |
| RV/TLC ratioa | 46.2 (39.8–57.5) | 39.8 (33.5–47.4) | 43.3 (38.7–45.8) | 56.8 (49.7–64.6) | 0.000 |
| FEV1% predb | 70.0 (46.2–90.2) | 92.5 (77.0–106.9) | 84.5 (76.9–104.2) | 46.3 (38.9–55.2) | 0.000 |
| FVC% predb | 83.0 (74.0–98.7) | 95.5 (83.0–110.4) | 81.3 (69.9–92.7) | 84.0 (75.3–98.5) | 0.104 |
| FEV1/FVC ratiob | 69.9 (43.4–82.3) | 77.2 (71.0–85.3) | 82.5 (79.8–87.2) | 43.2 (35.6–50.6) | 0.000 |
| DLCO% predc | 45.8 (36.5–61.1) | 37.8 (33.9–64.0) | 53.1 (38.9–64.7) | 44.0 (35.7–58.4) | 0.118 |
% pred: predicted percentage. RV: residual volume. TLC: total lung capacity. FEV1: forced expiratory volume in the first second. FVC: forced vital capacity.
In our study, CPFE was mainly diagnosed in elderly male smokers, as previously described.1,3,5–9,12–14 Almost a third of CPFE patients had PH at diagnosis, and this prevalence did not significantly differ in comparison with IPF or emphysema patients. However, there is previous evidence that PH is more frequent in CPFE than IPF or emphysema patients.1,5–9,12
To the best of our knowledge, RV/TLC ratio has not been described in CPFE, or even compared between CPFE, IPF and emphysema patients. We found that CPFE patients had significantly lower RV/TLC ratio than emphysema patients (39.8 vs. 56.8). Accordingly, a normal (lower than 40) or slightly increased RV/TLC ratio could help to discriminate patients with emphysema and superimposed fibrosis from patients only with emphysema.
CPFE patients had significantly lower static lung volumes and higher FEV1% pred and FEV1/FVC ratio compared to emphysema patients, which is in accordance with previous studies.3,5
A trend towards higher lung volumes was seen comparing CPFE and IPF patients, but did not reach statistical significance. Our IPF patients had median lung volumes at diagnosis within the normal or near-normal range, which could underestimate the difference between these two groups of patients. Kohashi et al. also did not find significant differences in FEV1% pred, FVC% pred and FEV1/FVC ratio between IPF alone and IPF-emphysema.14 However, other studies have consistently shown higher lung volumes and lower FEV1/FVC ratio in CPFE versus IPF.5,13,14
Although CPFE patients presented the lowest median DLCO% pred, this functional parameter did not significantly differ between the three groups of patients. This result is not accordant with previous studies. More than 20 years ago, Wells et al. showed that patients with IPF and emphysema had lower DLCO% pred by approximately 15% as compared to patients with IPF alone,15 and subsequent studies have also suggested that DLCO% pred is significantly lower in CPFE versus IPF.3,5 It is possible that the extent of fibrosis and emphysema (i.e. stage of disease at diagnosis) and PH could have influenced our results as suggested by other studies.9,12,14
The main strengths of our research are: (i) direct comparison of static lung volumes and DLCO% pred between CPFE, IPF and emphysema patients, (ii) use of “real-life” data, and (iii) pre-specified threshold of at least 10% of emphysema to define CPFE.
This study has also some limitations: (i) its retrospective nature, (ii) the relative small number of CPFE patients analysed, (iii) the lack of a quantitative evaluation of the extent of fibrosis or emphysema and (iv) selection bias of emphysema patients from a COPD Outpatient Clinic; all emphysema patients had evidence of airflow obstruction, which is not always observed in clinical practice.
In daily clinical practice, to accurately diagnose CPFE may be difficult as it shares many clinical and radiological characteristics with other smoking-related lung diseases. This study showed that a lower RV/TLC ratio could be a potential surrogate to distinguish between CPFE and emphysema when diagnostic work-up raises doubts.