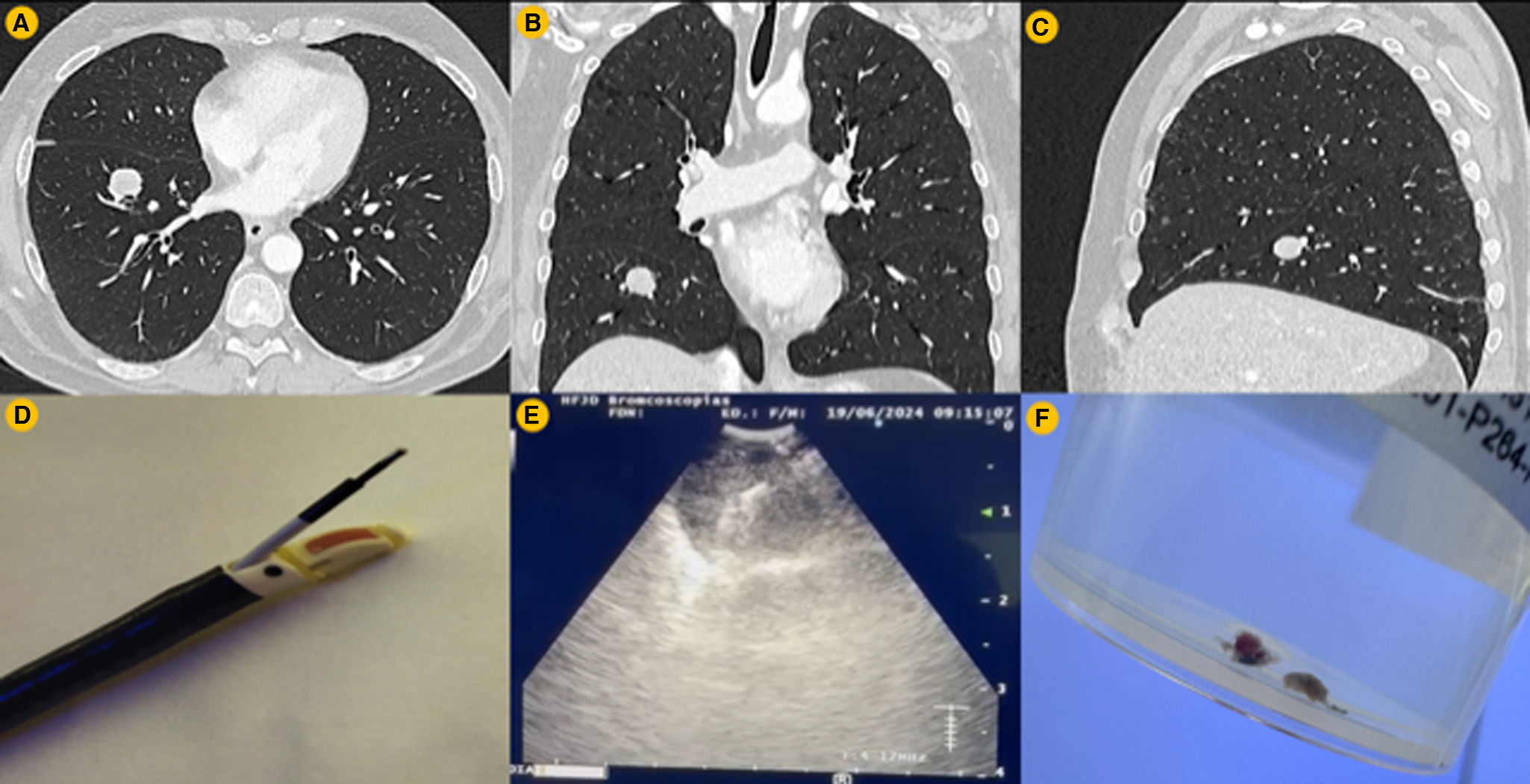
In the field of lung cancer research, there is an increasing need to enhance the quality and quantity of material submitted for pathological analysis, given the exponential rise in therapeutic targets over recent years. Linear endobronchial ultrasound (EBUS) has become a pivotal diagnostic and staging tool in lung cancer, facilitating access to pulmonary tumour lesions and mediastinal lymph nodes. The extensive use of this technique has enabled operators to attain a high level of proficiency, thereby paving the way for the development of new indications and diagnostic tools, such as miniforceps and cryoprobes. This document presents a novel technique utilising linear EBUS for the investigation of a peripheral pulmonary nodule. The procedure was conducted in the context of diagnosing a pulmonary nodule measuring 19mm in diameter in the right lower lobe (RLL). A solitary pulmonary nodule was identified in a smoker with a 40-pack-year history, measuring 19mm in the right lower lobe (RLL), with a maximum standardised uptake value (SUVmax) of 2.3 on positron emission tomography-computed tomography (PET-CT). A diagnostic bronchoscopy and linear EBUS were scheduled. In order to select the case and apply this new technique, we followed the criteria published by Gasparini et al.1 (2023), who conducted a retrospective review of cases of peripheral pulmonary nodules (PPN) addressed with linear EBUS. The selection criteria for diagnosing pulmonary nodules (PPNs) with a diameter of ≤3cm typically included the following: The pulmonary nodule must be located in the lower lobes, in close proximity to the wall of at least a subsegmental bronchus, or in a more distal position. Evidence from computed tomography (CT) scans indicated the presence of an airway with a diameter of at least 3mm in close proximity to the nodule, with a maximum diameter of 1cm. No endobronchial abnormalities were identified on flexible bronchoscopy. The EBUS-TBNA was conducted under moderate sedation (midazolam 1.5mg/kg+fentanyl 50 mcg+propofol 120mg) subsequent to the instillation of topical anaesthesia (lidocaine 1%). Throughout the procedure, the patient's oxygen saturation, electrocardiogram, and transcutaneous carbon dioxide levels were monitored continuously. Subsequently, a routine exploratory bronchoscopy was performed to rule out endobronchial lesions, after which linear EBUS (Olympus BF-UC-190F) was initiated. The 19mm nodule was successfully visualised sonographically in the anterior subsegment of the RLL segment VIII (Fig. 1a–c). The sample collection commenced with the utilisation of a 21G Olympus EBUS needle (NA201SX-4021), which facilitated the acquisition of three high-quality passes with cell block. The fine-needle pass through the bronchial wall facilitated the subsequent introduction of biopsy tools, thereby ensuring the acquisition of a high-quality histological structure. The innovative aspect of this procedure involved the utilisation of the Olympus GuideSheath Kit II (K-401) catheter, which is compatible with an EBUS channel measuring less than 2mm (Fig. 1d). From an endoscopic perspective, the catheter's distal tip, which was coloured black, was visible, and a rubber stopper was used to secure the proximal end at the point of entry to the working channel. A 1.1mm ERBE cryoprobe was passed through the catheter and identified upon exiting the distal end. The transbronchial access to the nodule, previously created with the EBUS needle, permitted the cryotherapy probe to be passed into the tumour (Fig. 1e). The cryobiopsy procedure was conducted in a step-by-step manner, as follows:
- 1.
The 1.1mm ERBE cryoprobe was then inserted through the catheter, which was lodged in the EBUS working channel.
- 2.
The lesion was then visualised, and the cryoprobe was advanced into the tumour at the same location as the initial needle puncture.
- 3.
A four-second freezing period was initiated, after which the bronchoscope, catheter, and cryoprobe were removed in a single block, leaving the cryoprobe's tip visible within the central airway. At this juncture, the cryobiopsy was visualised through the bronchoscope.
- 4.
The distal end of the cryoprobe was adjusted in conjunction with the catheter, resulting in the probe being enveloped within the catheter's distal end.
- 5.
The cryoprobe, still encased within the catheter, was reactivated by depressing the foot pedal in order to maintain the biopsy's adherence. Subsequently, the catheter and cryoprobe were extracted as a single unit from the EBUS.
- 6.
The biopsy was then retrieved by extending the cryoprobe beyond the catheter once the latter had been removed from the bronchoscope. The procedure was straightforward to perform and, most importantly, safe for the patient, yielding high-quality samples from within the tumour (Fig. 1f). Furthermore, this technique could be employed in the context of cryobiopsies of mediastinal lesions, particularly in instances where there is a risk of excessive bleeding. Furthermore, this approach enhances the quality of the procedure by circumventing the necessity for repeated intubations and extubations during spontaneous ventilation. This study was conducted in accordance with the Declaration of Helsinki and received approval from the FJD Ethics Committee (CERM: 172/2023). The data were anonymised, and informed consent was not obtained. The pathological analysis of the cryobiopsy samples from the peripheral pulmonary nodule revealed a carcinoid tumour to be the most likely diagnosis. The diagnosis of peripheral nodules represents a significant challenge in the context of lung cancer. In light of the advent of new therapies, the ability to obtain larger2 and structurally preserved3 samples has become of paramount importance. The utilisation of real-time sample collection with ultrasound guidance, as facilitated by linear EBUS, has the potential to enhance the diagnostic yield while simultaneously improving patient safety by circumventing biopsies in high-risk areas. The diagnostic yield for mediastinal staging with linear EBUS is over 92%,4 in comparison to 72%5 with radial EBUS, as evidenced by meta-analyses of published studies. In contrast to other cryobiopsies utilising linear EBUS for mediastinal lesions,6 this technique obviates the necessity for the bronchoscope to be removed in order to obtain the sample. This precludes the potential for friction with the airway during specimen retrieval and permits continuous monitoring of the airway, which is of paramount importance in peripheral nodule7 biopsies due to the risk of bleeding. This approach is also applicable in mediastinal studies8 where bleeding risk is a concern.
No artificial intelligence was used in the preparation of this manuscript.
FundingNone declared.
Conflicts of InterestNone declared.