CURRENT SITUATION AND LONG-TERM CONSEQUENCES OF COVID-19 INFECTION
More infoAs with the rapid increase of the number of patients who have recovered from COVID-19 globally, there needs to be a major shift of the focus from rapid pathogen detection, treatment and prevention to the promotion of better recovery. Notwithstanding the scarcity of our understandings, recent studies have unraveled a plethora of pulmonary and systemic consequences which require medical attention. These consequences remained as of the end of follow-up which ranged from 1 month to 1 year. Here, we review the consequences of COVID-19 in terms of the residual symptoms, radiological and functional manifestations, and identify the potential risk factors that contribute to a prolonged recovery. We also summarize the benefits of clinical interventions (particularly the pulmonary rehabilitation program), and address several undetermined concerns and key future research directions.
Como consecuencia del rápido aumento del número de pacientes que se han recuperado de la COVID-19 en todo el mundo, es necesario cambiar el enfoque de la detección rápida del patógeno, el tratamiento y la prevención para promover una mejor recuperación. A pesar de la escasez de nuestros conocimientos, estudios recientes han desvelado una plétora de consecuencias pulmonares y sistémicas que requieren atención médica. Estas consecuencias se mantienen al final del seguimiento, que oscila entre 1 mes y 1 año. Aquí se hace una revisión de las consecuencias de la COVID-19 en términos de síntomas residuales y manifestaciones radiológicas y funcionales y se identifican los posibles factores de riesgo que contribuyen a una recuperación demorada. También se resumen los beneficios de las intervenciones clínicas (en particular el programa de rehabilitación pulmonar) y se abordan varias preocupaciones no resueltas y direcciones clave de investigación futura.
Coronavirus disease of 2019 (COVID-19) has strained the medical infrastructure, personnel and resources worldwide. The burden continues to increase amid the emergence of new variants and the progressively waning immunity after global vaccine roll-out. According to the latest statistics, the total number of laboratory-confirmed cases of COVID-19 has exceeded 0.4 billion globally,1 despite the dramatically improved rate of survival.
Although a certain number of patients ultimately developed severe or critical illness, symptoms gradually resolved among most patients at convalescence.2–4 The number of patients who recover from COVID-19 will continue to increase considerably in the next foreseeable future. This constitutes a pressing need for caring a massive population which has not been fully characterized previously. Not all patients achieved complete remission of symptoms, radiologic findings and pulmonary function despite a prolonged convalescence phase.2,3,5–11 The consequences contributed to the ongoing suffering from physiologic dysfunction and the impaired quality-of-life which markedly dampened the overall well-being. Given the overwhelming tasks of disease prevention and control, global research efforts have characterized the clinical presentations at disease onset or during hospital admission, to identify the prognostic factors and disease phenotypes, to design and validate rapid diagnostic tests, and to verify the effects of therapeutic interventions and vaccines. Recent studies have documented the manifestations of consequences which ranged from respiratory to systemic (including psychological) disorders,12 with the follow-up duration ranging from 3 months to 1 year.2–5,10,11,13–17 In light of the variable study designs and research focus, there remains a considerable knowledge gap in the understanding of the magnitude and heterogeneity of the consequences of COVID-19, particularly in the era when the medical focus has been shifted from the diagnosis and treatment to the rehabilitation.
Exploring the spectrum of consequences among patients recovered from COVID-19 might help characterize the residual disease burden and identify the major predictors of adverse outcomes, which have important implications to the early diagnosis and accelerate the development of novel interventions. Here, we review the major manifestations of short- and long-term consequences, highlighting the clinical characteristics and predictors of having any consequences. We also summarize the latest clinical interventions and elaborate the important future research directions.
Clinical characteristics at acute phaseAirborne droplets or virus-laden materials were the dominant sources of infection.18 After acquisition of virus, common symptoms which consisted of fever, cough, fatigue, dyspnea, sore throat and headache typically occurred after the median incubation period of 4 days.19 However,>50% of patients remained afebrile on hospital admissions and developed fever thereafter. Gastrointestinal symptoms, such as loss of appetite, nausea and vomiting, were less common as compared with those in patients with avian influenza.20 Neurological manifestations (e.g., myalgia, encephalopathy, dysgeusia and anosmia) were specific to aid in the diagnosis although appearing less common.21 Few patients developed autoimmune disorders, including haemolytic anemia,22 immune thrombocytopenia,23 Guillain-Barre syndrome,24 and Kawasaki disease-like syndrome in children.25
On laboratory examination, the most common manifestations consisted of lymphopenia, elevated C-reactive protein and elevated liver enzymes.19 These manifestations have important clinical implications for prognosis. Lymphopenia (seen in ∼85% of patients), has been associated with the risk of progression to critical illness and death.19 Several radiologic manifestations on chest X-ray or computed tomography (CT), such as ground-glass opacity, pulmonary infiltration, and consolidation usually developed at later stages of progression.26 The radiologic abnormality, albeit rapidly evolving during progression,26 could be readily detected at early stages among most patients and continued to persist.19 Lung function findings regarding airflow limitation and lung volumes were usually unremarkable.27 By contrast, diffusing capacity impairment and reduced exercise tolerance were more specific to reflect functional impairment of COVID-19.28
The path of disease progressionAlthough approximately 85% of patients developed mild diseases,19 the risk of progression to critical illness (e.g., severe pneumonia, acute respiratory distress syndrome, sepsis, multisystem organ failure) or death among the remaining patients could not be underestimated. Older age, greater symptom burdens, severe hypoxemia,29 and comorbidities (e.g., hypertension, diabetes, heart disease, chronic obstructive pulmonary disease) have been associated with a worse outcome.30 Furthermore, certain chest radiologic31 and laboratory parameters (e.g., decreased CD3+, CD4+, and CD8+T cell count, higher neutrophil-to-lymphocyte and neutrophil-to-CD8+T cell ratio) might provide prognostic implications.32 Prevention of progression to critical illness or death has been the core mission of the clinical management, because the severity of illness correlated negatively with the likelihood of, and the time needed for, clinical recovery.33
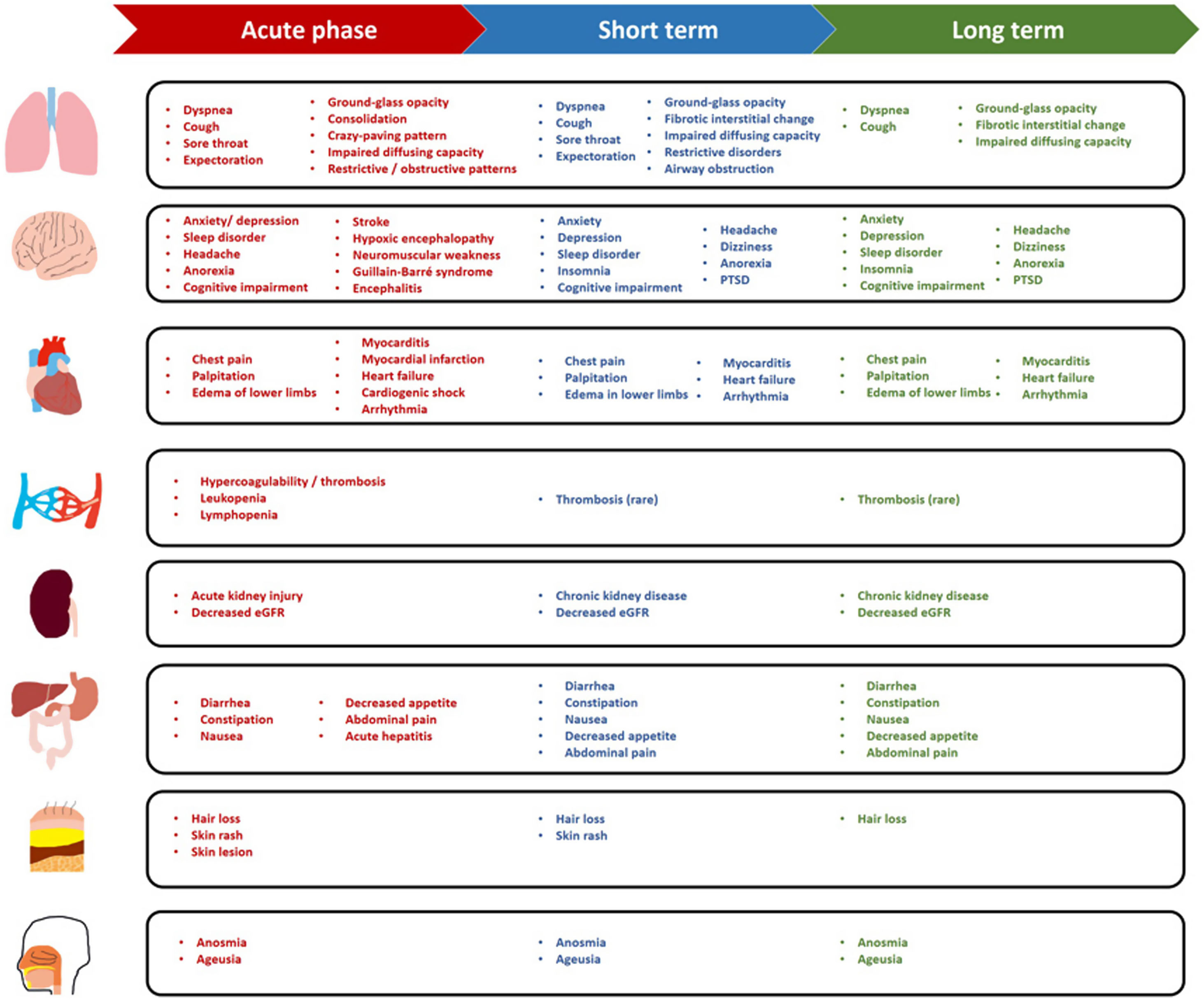
Short- and long-term consequencesAlthough the symptoms or signs of COVID-19 ultimately resolved among the majority of patients, some suffered from persistent or new-onset symptoms and conditions which have been described as “long COVID”.34 The definition is continuously evolving, although a new consensus definition for “post COVID-19 condition” has recently been proposed.35 A commonly accepted concept refers long COVID to the symptoms that continue or develop after acute COVID-19 infection which cannot be explained by an alternative diagnosis.36 However, this could be seen in many COVID-19 survivors>6 months after recovery.37 Thus, we summarize the short- (4 to 12 weeks after disease onset) and long-term (12 weeks to 1 year or longer) consequences of COVID-19 according to the ongoing symptomatic and post COVID-19 syndrome stages below, as proposed by the UK National Institute for Health and Care Excellence guideline.36
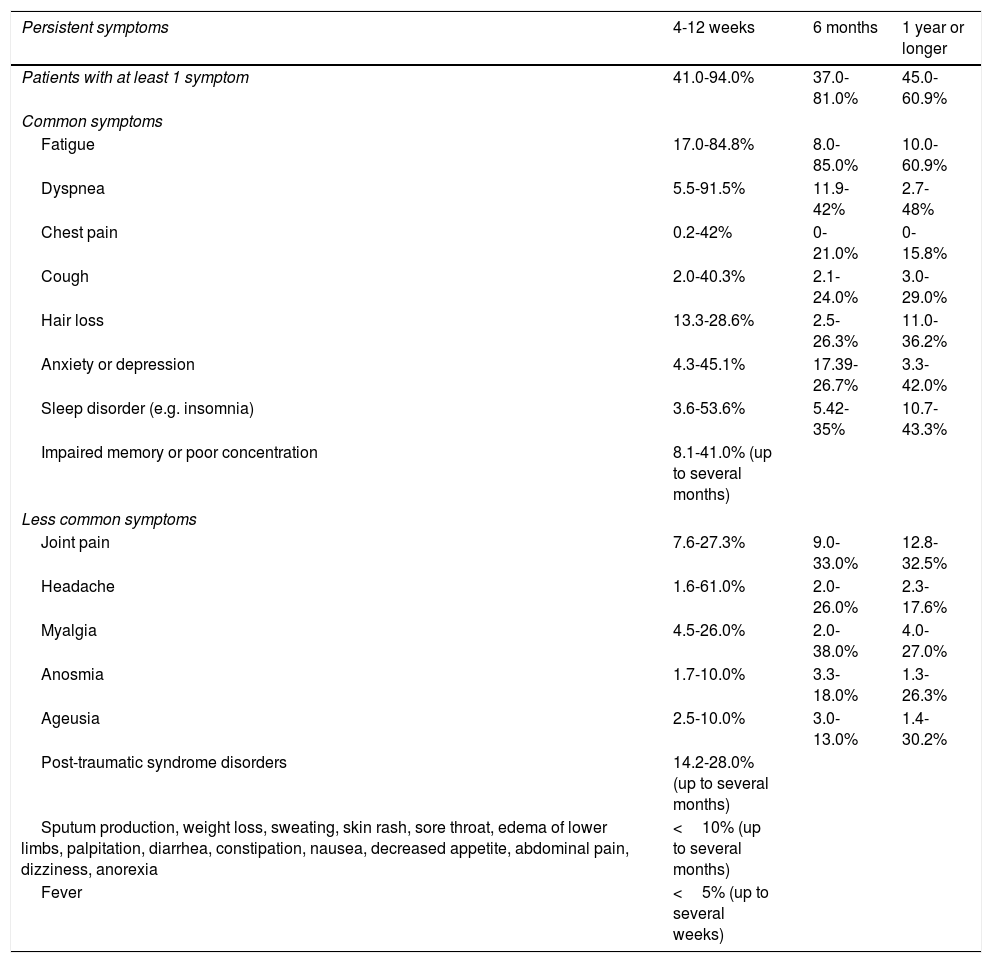
Short-term consequencesSymptoms and signs: At 4-12 weeks after the initial symptom onset, a considerable proportion of patients reported residual symptoms such as cough (2%-40.3%), dyspnea (5.5%-91.5%), chest pain (0.2%-42%), sputum production (<10%), cognitive impairment (17%-28.3%), and fatigue (17%-84.8%), while fever might persist for a couple of weeks without secondary infection (Table 1).37–40 Over 60% of patients reported amelioration of olfactory and/or gustatory dysfunction within the first 2 months (with the percentage decreasing to 40% and 20%, respectively), followed by a slower rate of recovery thereafter.41 Psychological disorders (e.g., post-traumatic stress disorder [PTSD]) occurred in 24.5% of survivors at 1 month after discharge and in 22.4% of patients at 3 month follow-up.42 Insomnia, anxiety and depression could still be identified at 3 months.42
Proportion of patients with persistent symptoms at the short and long terms.
| Persistent symptoms | 4-12 weeks | 6 months | 1 year or longer |
|---|---|---|---|
| Patients with at least 1 symptom | 41.0-94.0% | 37.0-81.0% | 45.0-60.9% |
| Common symptoms | |||
| Fatigue | 17.0-84.8% | 8.0-85.0% | 10.0-60.9% |
| Dyspnea | 5.5-91.5% | 11.9-42% | 2.7-48% |
| Chest pain | 0.2-42% | 0-21.0% | 0-15.8% |
| Cough | 2.0-40.3% | 2.1-24.0% | 3.0-29.0% |
| Hair loss | 13.3-28.6% | 2.5-26.3% | 11.0-36.2% |
| Anxiety or depression | 4.3-45.1% | 17.39-26.7% | 3.3-42.0% |
| Sleep disorder (e.g. insomnia) | 3.6-53.6% | 5.42-35% | 10.7-43.3% |
| Impaired memory or poor concentration | 8.1-41.0% (up to several months) | ||
| Less common symptoms | |||
| Joint pain | 7.6-27.3% | 9.0-33.0% | 12.8-32.5% |
| Headache | 1.6-61.0% | 2.0-26.0% | 2.3-17.6% |
| Myalgia | 4.5-26.0% | 2.0-38.0% | 4.0-27.0% |
| Anosmia | 1.7-10.0% | 3.3-18.0% | 1.3-26.3% |
| Ageusia | 2.5-10.0% | 3.0-13.0% | 1.4-30.2% |
| Post-traumatic syndrome disorders | 14.2-28.0% (up to several months) | ||
| Sputum production, weight loss, sweating, skin rash, sore throat, edema of lower limbs, palpitation, diarrhea, constipation, nausea, decreased appetite, abdominal pain, dizziness, anorexia | <10% (up to several months) | ||
| Fever | <5% (up to several weeks) | ||
Abnormalities reported within 4-12 weeks53,75,99-104.
Abnormalities reported at 6 months2,3,63,70,74,83,100,105-107.
Abnormalities reported at 12 months or longer 2,46,61,62,64–67,72,108.
Chest radiologic findings: Chest radiologic abnormalities could be identified in>75% of patients at initial presentation19 and could persist at convalescence. Ground-glass opacification (GGO) was highly prevalent at discharge (99.5%) but was only detected among 37.3% of patients at 3 months. However, consolidation and crazy-paving pattern might have resolved at hospital discharge. At 1 month after discharge, GGO (42.4%, 37.5%) and reticular pattern (22.0%, 47.5%) were most common in patients with mild and severe COVID-19. These figures decreased to 29.7% and 28.3% for GGO and 13.0% and 36.7% for reticular patterns at 3 months.43 By contrast, parenchymal bands, thickening of adjacent pleura and reticular lesions persisted in 34.4%, 13.9%, and 14.8% of patients at 3 months.44
Lung function abnormality: Although occurring in a small proportion of patients during acute phase, pulmonary function remained abnormal in the early post-acute phase especially in patients with severe COVID-19.45,46 Reduced diffusing capacity, seen in 10-20% of patients at presentation, could substantially improve at 3 month in patients with severe COVID-19.46–48
Systemic complications: COVID-19 conferred notable adverse impacts on multiple organ systems, whose function was progressively normalized in the short term. Cardiovascular complications included myocarditis, myocardial infarction, heart failure, cardiogenic shock and arrhythmia.49 COVID-19 appeared to be associated with persistent cardiac injury after recovery, particularly subclinical myocardial injury in the short term and diastolic dysfunction in the long term.50 Despite the high incidence at acute phase,19 acute kidney injury could be generally resolved within 3 weeks without renal replacement therapy among patients with non-severe COVID-19;51,52 new-onset chronic kidney disease was unusual but could relapse after discharge.3,53 Acute liver injury or hepatitis occurred in few patients only and resolved during the acute phase among patients with non-critical illness, with minor impacts on long-term outcomes.30,54,55 Thyroid function might be impaired at acute phase in approximately 13.5% of patients and could be normalized within 2 months.56 Hypercoagulability (e.g., venous and arterial thromboses) was common at acute phase, especially in patients with severe or critical illness,57 but the incidence of venous thromboembolism fell to below 5% in the post-acute phase.38
Short-term mortality: Although most patients with COVID-19 had asymptomatic or mild-to-moderate illnesses, findings from the initial outbreak indicated the fatality rate of approximately 1.4% in mainland China.19 Findings from Spain have also lent support to these earlier observations. In a multicenter Spanish cohort of 12,126 patients, the mortality rate was 12.5% for non-hospitalized patients, 29.8% in hospitalized patients and 38.8% in patients admitted to the intensive care unit.58 The key predictors of short-term mortality consisted of the age over 50, obesity, cardiac diseases, fever, dyspnea, lung infiltration, lymphopenia, D-dimer levels above 1000 ng/mL, and requirement of mechanical ventilation.58 When taking into account the impact of the underlying diseases, in particular the chronic respiratory diseases, another multicenter Spanish cohort study of 5847 hospitalized patients with COVID-19 revealed a significantly higher 30-day mortality rate in patients with prior lung diseases than those without (29.5% vs. 17.9%).59 The study also revealed a more impressive 30-day mortality rate (approximately 40%) in patients with pre-existing COPD.59 Both studies, however, concurred that a greater age and higher D-dimer levels were associated with a significantly elevated risk of short-term death.58,59 Patients aged greater than 60 years were at a particularly high risk of death at the short term, with the overall in-hospital mortality rate of 23.5% in a retrospective single-center study in Spain.60 Among these elderly patients, those with malignancy, chronic liver diseases, obesity and diabetes had an increased mortality risk despite the adjustment with the age.62 These studies have provided the scientific rationale for rapid identification of patients at risk of death and the implementation of more intensive therapeutic management.
Medium- to long-term consequencesSymptoms and signs: At week 12 and thereafter, symptoms or signs progressively waned but could still fluctuate over time. Some patients reported residual symptoms for up to 6 months or longer, especially those with severe COVID-19.3,61 Cough lasted for 6 months or longer in 20% of patients (Table 1),62 which might be associated with pulmonary fibrotic changes.63 Breathlessness could persist for up to 1 year.2 Chest pain or tightness was reported in approximately 15.8% of patients over 1 year.64 Systemic symptoms such as fatigue persisted for 12 months among 10-60.8% of patients,2,58,64–68 which might be associated with the accentuated systemic inflammatory response and psychological disorders.68 Persistent taste and smell disorders have been reported at 6 months (7% and 11%, respectively),3 although recovered progressively thereafter.2 Albeit less commonly reported, arthralgia and myalgia could persist for over 6 months.2,69–71 Hair loss was reported in>20% of patients at 6 months while decreased to 11% at 1 year.2 Neurological disorders (e.g. sleep disorder, headache, dizziness) persisted with minor improvement for 1 year.2,62,71 Noticeably, 27% of patients suffered from sleep disorder at 6 months and 17% still had insomnia at 1 year.2 Cognitive impairment was reported to be persistent, the magnitude of which fluctuated from 4 months to 1 year, and was more prevalent in patients with severe COVID-19.62,72–75 COVID-19 imposed a considerable burden on psychology. More than 20% of patients suffered from PTSD at 1 year or longer.76 Anxiety and/or depression was reported in approximately 23% of patients at 6 months and 26% at 12 months.2,62,76
Chest radiologic findings: During recovery, GGO and interstitial fibrotic changes remained common.2,44,53 More than 60% of patients with mild or moderate COVID-19 ultimately had resolved radiologic abnormalities. However, GGO and fibrotic changes could persist while consolidation was rare on chest CT at 1 year follow-up.2,77 Furthermore, the disease severity was associated with the magnitude of chest CT manifestations. At 3 months, pulmonary structural abnormalities were highly prevalent (up to 80.7%) in patients hospitalized in the intensive care unit due to ARDS.78 Radiologic abnormalities persisted in 20% of patients with severe COVID-19 who did not require mechanical ventilation at 12 months.79 Compared with patients who had completely resolved, a significantly higher proportion of patients diagnosed as having severe pneumonia and acute respiratory distress syndrome at the initial presentation had residual radiologic abnormality at 1 year.44
Lung function abnormality: Restrictive and obstructive patterns and impaired diffusing capacity were common in COVID-19 survivors,80 although improved considerably in non-critical survivors within 3 months.47 The improvement of diffusing capacity, however, seemed to slow down at 6 months. For up to 12 months, impaired diffusing capacity was identified in 20-30% of patients who had moderate COVID-19, whose prevalence remained constant since month 6. By contrast, impaired diffusing capacity could be found in approximately 54% of patients with critical illness at 1 year.2 In patients who had initially suffered from severe COVID-19 but did not require mechanical ventilation, impaired pulmonary function improved gradually over 1 year, with females having a higher risk of the ongoing impairment as compared with males.79 Impaired diffusing capacity could persist for a longer period, for instance, up to 18 months after discharge, among 57.92% of survivors with more severe disease.46,81 Reduced exercise capacity was another important characteristic lung function change among survivors,37 which improved gradually since month 3 in patients with severe COVID-19.79
Systemic complications: As mentioned above, most systemic complications appeared to be transient. However, the estimated glomerular filtration rate decreased in 35% of patients after 6 months, 13% of which was categorized as the new-onset reduction of glomerular filtration rate despite the normal renal function at COVID-19 onset.3 At 6 months, hyperglycemia persisted in>30% of patients who experienced new-onset hyperglycemia during the hospitalization.82 A cohort study reported new-onset diabetes in 3.3% out of 1733 patients at 6 months.3
Functional abnormality: The magnitude of improvement depended considerably on the initial disease severity. Most patients with severe illness reported a failure of recovery in physical activity at 6 months.83 45% of hospitalized survivors exhibited limitations on daily living activities at 7 months (Table 1).84 The health care workers with severe COVID-19 showed improvement in functional fitness within 1 year, and rigorous intervention might be considered for accelerating the pulmonary rehabilitation (Fig. 1).85
Medium- and long-term mortality: The adverse impact of COVID-19 on the mortality risk extended to the long terms. In a cross-sectional study conducted in France, the mortality rate was 62.5% during the intensive care unit stay and increased to 72.1% to month 6 among patients aged greater than 80 years who were receiving mechanical ventilation.86 At 1 year, patients treated with corticosteroids had a markedly higher mortality rate as compared with those not (50.0% vs. 21.0%), although the data interpretation was limited by the small sample sizes.87 A study conducted in Italy has also demonstrated a markedly higher mortality rate among hospitalized patients with COVID-19 than those without (48.4% vs. 33.9%) at 1 year.88 The 1-year mortality rate was also higher in non-hospitalized emergency room visitors with COVID-19 than those without (18.0% vs. 8.7%).88
Future research perspectiveThe continuously evolving global pandemic has elicited an unprecedented challenge for healthcare workers, policy makers and the public. Current research has outlined the clinical spectrum of the consequences, but this would surely benefit from the in-depth mechanistic insights which would unveil the biologic pathways that contribute to the ongoing disorders. Continuous profiling of the cytokine and chemokine expression levels within the lower airways (e.g., induced sputum) or peripheral blood will help understand the dynamics of residual inflammatory responses. The combination of chest imaging with high-resolution CT or functional magnetic resonance imaging with isotopes and lung function assessment (e.g., lung volume, diffusing capacity, exercise capacity) will provide clinicians an indispensable avenue for tracking the trend of changes in pulmonary functional abnormality.7–9,17,89 Further extension of the clinical observation is needed to reveal the long-term impact (e.g., 3-5 years) on symptom perception, lung function (particularly the exercise capacity), radiological characteristics, quality-of-life and psychology.10,11,16,90 This will help reveal the duration of recovery of individual symptoms which would normally take, and determine the optimal timing for evaluating the magnitude of recovery and defining complete or partial recovery.
Few intervention strategies have been proposed to accelerate recovery, with pulmonary rehabilitation demonstrating some promising findings.91,92 Anti-inflammatory medications or biologics might have a role in antagonizing the residual inflammation, especially when taking into account the pulmonary (e.g., cough, sputum production) and systemic manifestations (e.g., fatigue). There also lacks a crystal-clear indication for the population which would benefit most from therapeutic interventions after discharge. Whether non-pharmaceutical interventions are superior to drug therapy for improving the functional well-being (e.g., exercise capacity) merits further investigations from the personalized interventions. It would be important to address the optimal duration of such interventions should they be proven effective.
Previous studies have indicated a strong immune protective effect against acquiring re-infections among the COVID-19 rehabilitees.93 The natural immunity has constituted the front line which prevents from being susceptible. However, approximately 4% of patients were susceptible to re-infections with the precise mechanisms not fully explored.93–95 Despite the formation of pan-sarbecovirus antibodies,96 it remains unclear whether COVID-19 survivors would be immune to other coronavirus. Continuous follow-up might help delineate whether re-infections were caused by the waning immunity alone, the underlying immunodeficiency, or emerging variants evading the established immunity. A minority of patients were tested continuously RNA-positive or re-positive after discharge which might not necessarily reflect prolonged viral shedding.97,98 A work-up with the government, epidemiologists and clinicians will help ascertain the true infectivity of COVID-19 survivors by gathering the information of the dynamic nucleic acid assays monitoring, enforcing symptom monitoring of patients and close contacts, and integrating multiple laboratory tests (e.g., viral culture, high-throughput sequencing). This will be important for redefining the community-based prevention measures, since no existing study has disclosed the infectivity of COVID-19 survivors who were discharged after having at least two consecutive RNA-negative laboratory test findings.
ConclusionsThe world has come across a tough period when COVID-19 resulted in an overwhelming burden. Thanks to the efforts of the medical and scientific communities, the rapid deployment of effective vaccines and accelerated development of repurposed and novel medications, most patients have either partially or completely recovered from COVID-19. Although many questions remain unanswered, we keep optimistic that the ongoing efforts will better characterize the trajectory of clinical recovery and unveil new interventions which would promote recovery. The goal of enabling a better recovery would ultimately be realized for most COVID-19 rehabilitees.
Support statementThis work was supported by Guangzhou Institute for Respiratory Health Open Project (funded by China Evergrande Group) - Project No. 2020GIRHHMS09 and 2020GIRHHMS19 (Prof. Guan) and the Open Project of the State Key Laboratory of Respiratory Disease (SKLRD-OP-201909) (Dr. Gao).
Author contributionsYang Gao, Wei-quan Liang, Yi-ran Li, Jian-xing He, and Wei-jie Guan drafted the manuscript; Wei-jie Guan, and Jian-xing He critically revised the manuscript. All authors have approved the final submission.
Conflicts of interestThe authors declared no conflict of interest with any financial organization regarding the material discussed in the manuscript.
We thank Prof. Jiang Xie (Department of Pulmonary and Critical Care Medicine, Beijing Anzhen Hospital, Capital Medical University) for his helpful comments on the writing of the review.