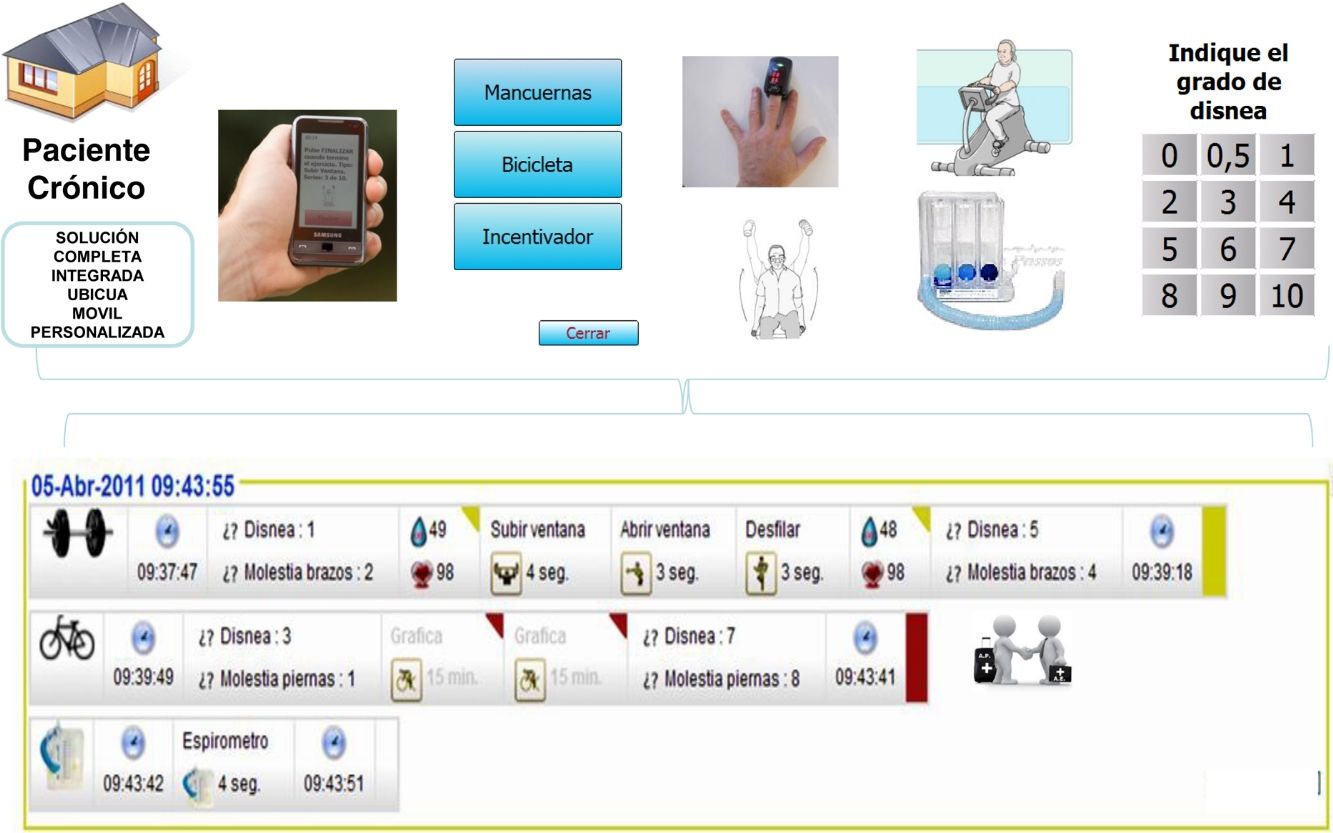
There is uncertainty regarding efficacy of telehealth-based approaches in COPD patients for sustaining benefits achieved with intensive pulmonary rehabilitation (PR).
Research questionTo determine whether a maintenance pulmonary telerehabilitation (TelePR) programme, after intensive initial PR, is superior to usual care in sustaining over time benefits achieved by intensive PR.
Study design and methodsA multicentre open-label pragmatic parallel-group randomized clinical trial was conducted. Two groups were created at completion of an 8-week intensive outpatient hospital PR programme. Intervention group (IG) patients were given appropriate training equipment and instructed to perform three weekly training sessions and send performance data through an app to a web-based platform. Patients in the control group (CG) were advised to exercise regularly (usual care).
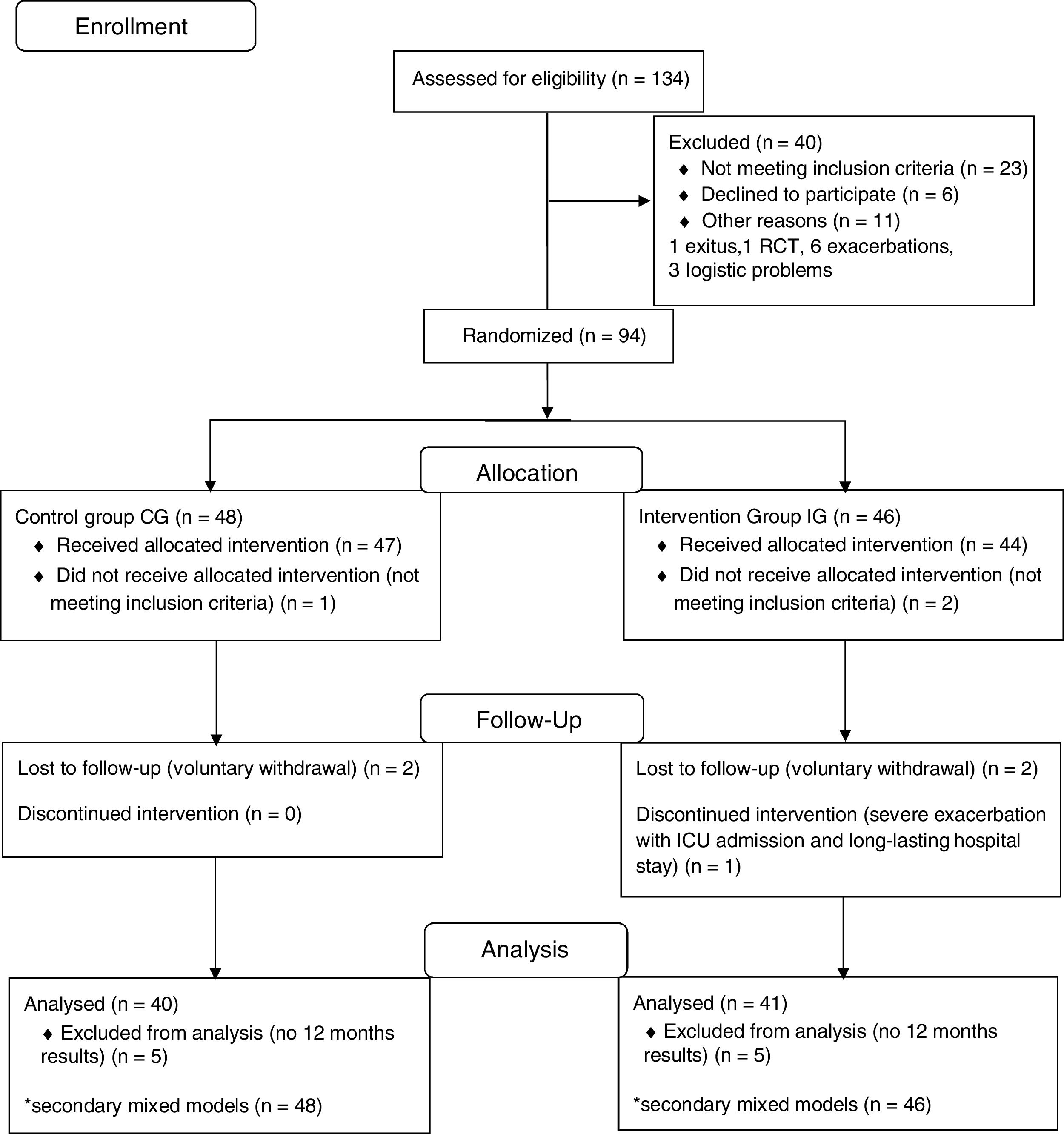
ResultsNinety-four patients (46 IG, 48 CG) were randomized. The analysis of covariance showed non-significant improvements in 6-min walk distance [19.9m (95% CI −4.1/+43.8)] and Chronic Respiratory Disease Questionnaire – Emotion score [0.4 points (0–0.8)] in the IG. Secondary linear mixed models showed improvements in the IG in Short Form-36 mental component summary [9.7, (4.0–15.4)] and Chronic Respiratory Disease Questionnaire – Emotion [0.5, (0.2–0.9)] scores, but there was no association between compliance and outcomes. Acute exacerbations were associated with a marginally significant decrease in 6-minute walk distance of 15.8m (−32.3/0.8) in linear models.
ConclusionsThe TelePR maintenance strategy was both feasible and safe but failed to show superiority over usual care, despite improvements in some HRQoL domains. Acute exacerbations may have an important negative influence on long-term physical function.
ClinicalTrials.gov identifierNCT03247933.
Existe incertidumbre con respecto a la eficacia de los enfoques basados en telesalud en pacientes con enfermedad pulmonar obstructiva crónica (EPOC) para mantener los beneficios logrados con la rehabilitación pulmonar (RP) intensiva.
Pregunta de investigaciónDeterminar si un programa de telerrehabilitación pulmonar de mantenimiento (TeleRP), después de una RP inicial intensiva, es superior a la atención habitual para mantener en el tiempo los beneficios logrados por la RP intensiva.
Diseño del estudio y métodosSe realizó un ensayo clínico aleatorizado, pragmático, abierto, multicéntrico, de grupos paralelos. Se crearon 2 grupos al finalizar un programa de RP intensiva en régimen ambulatorio de 8 semanas de duración. A los pacientes del grupo de intervención (GI) se les proporcionó el equipo de entrenamiento apropiado y se les instruyó para realizar 3 sesiones de entrenamiento semanales y enviar los datos de rendimiento a través de una aplicación a una plataforma web. Se aconsejó a los pacientes del grupo de control (GC) que hicieran ejercicio regularmente (cuidado habitual).
ResultadosSe aleatorizaron 94 pacientes (46 GI, 48 GC). El análisis de covarianza mostró mejoras no significativas en la distancia en la prueba de marcha de 6min (19,9m [IC 95%: −4,1/+43,8]) y el cuestionario de enfermedad respiratoria crónica-factor emocional (0,4 puntos [0-0,8]) en el GI. Los modelos lineales mixtos secundarios mostraron mejoras en el GI en las puntuaciones de la sección mental del SF-36 (9,7 [4,0-15,4]) y el cuestionario de enfermedad respiratoria crónica-factor emocional (0,5 puntos [0,2-0,9]), pero no se demostró asociación entre el cumplimiento y los resultados. Las exacerbaciones agudas se asociaron con una disminución marginalmente significativa en la distancia en la prueba de la marcha de 6min de 15,8m (−32,3/0,8) en los modelos lineales.
ConclusionesLa estrategia de mantenimiento TeleRP fue viable y segura, pero no se demostró superioridad frente al cuidado habitual, a pesar de las mejoras en algunos aspectos en las valoraciones de la calidad de vida en relación con la salud. Las exacerbaciones agudas podrían tener una influencia negativa importante en la función física a largo plazo.
Identificador ClinicalTrials.govNCT03247933.
Chronic obstructive pulmonary disease (COPD) is the fourth leading cause of death in the world and more than 3 million people died of COPD in 2012. COPD accounts for 56% of costs attributable to respiratory diseases.1
There is a high level of evidence that intensive pulmonary rehabilitation (PR) programmes have benefits in patients with COPD. Some studies suggest that the benefits of PR programmes last no more than 1 year with a maintenance technique and the actual effectiveness and most appropriate way of delivering maintenance programmes are still a matter of debate.2,3
Telehealth has the potential to improve access to PR and support long-term exercise maintenance strategies provided remotely to people in their homes.4–6 A few studies have explored the feasibility and impact of using telerehabilitation approaches to deliver COPD maintenance programmes.7,8
If pulmonary rehabilitation using telehealth technology (TelePR) were to be shown to be capable of maintaining benefits achieved by PR in the intensive period, then a TelePR programme should become an alternative to conventional maintenance programmes. The objective of this study was, therefore, to determine whether a maintenance TelePR carried out after an intensive initial PR programme was superior to usual care in sustaining, over time, the benefits achieved by the intensive PR.
MethodsDesign, Participants and Eligibility CriteriaThis was a multicentre open-label pragmatic parallel-group randomized clinical trial involving 13 hospitals and was conducted between 2014 and 2017. Patient selection criteria were having a diagnosis of moderate-to-severe COPD (a Body mass index, Airflow obstruction, dyspnoea and Exercise capacity [BODE] index score of 3–7) according to international guidelines9 and having been clinically stable for the previous 4 weeks (see the online supplementary material).
The study was carried out in accordance with the principles of the Declaration of Helsinki and started after receiving authorization from the Spanish Agency for Medicines and Health Products and the approval of the Basque Country Ethics Committee, as well as the local Ethics Committee and Director at each institution. All patients gave written informed consent. ClinicalTrials.gov Identifier: NCT03247933.
RandomizationRecruited patients (recruitment visit, −1) attended an initial 8-week outpatient-based PR programme conducted in each hospital rehabilitation department. Following completion of this programme, patients were randomly allocated (baseline visit, 0) to either the TelePR programme [intervention group (IG)] or the standard, usual care programme [control group (CG)]. Allocation was stratified by centre and based on randomly permuted blocks of variable size with a 1:1 allocation ratio. Clinical epidemiology staff kept the centralized allocation lists concealed (see online supplementary material).
InterventionsIntensive PR (8 Weeks±4 Days) Visit (−1)The hospital-based outpatient programme consisted of three training sessions a week which included 30min of weight lifting and 30min of leg cycle ergometry along with four educational sessions which included chest physiotherapy training (see online supplementary material).
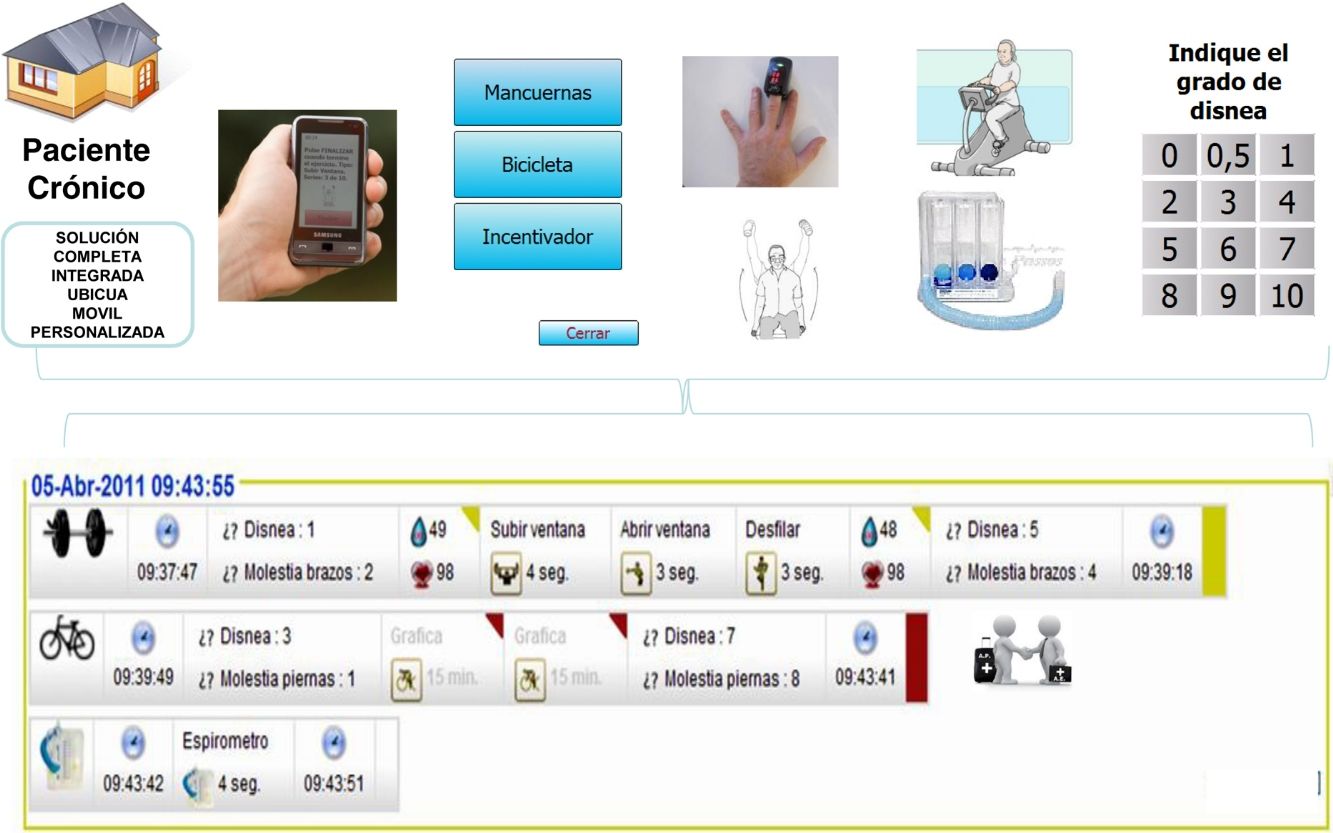
Maintenance ProgrammesPatients in the IG were asked to continue at home with a similar training schedule to that completed in the hospital. They were provided with, for the 12-month follow-up period, a telerehabilitation kit (mobile phone, pulse oximeter, dumbbells and exercise bicycle) along with a user guide with detailed instructions on the use of the mobile device to deliver data collected to the web-based platform after completion of each scheduled session (see online supplementary material).
Patients in the CG received usual care, that is, they were advised to exercise regularly (at least walking for 1 hour a day) and provided with general educational material, following current clinical practice at participating sites.
Follow-up VisitsAfter the randomization visit, follow-up appointments were arranged for both groups at 3, 9 and 12 months. At each follow-up visit, the general health status of the patient was assessed, medical treatment was revised and optimized, educational reinforcement was provided, 6-minute walk test (6MWT) and spirometry tests were carried out and HRQoL measures were taken. Lastly, BODE scores were calculated at each follow-up visit for the IG and at baseline and 12-month visits for the CG.
Outcomes MeasuresEfficacy and Safety MeasuresExercise tolerance (the ability to sustain exercise) was assessed using distance covered in the 6MWT.10,11 Health-related quality of life (HRQoL) was measured using a generic instrument (SF-36)12 and the Chronic Respiratory Disease Questionnaire (CRQ).13,14 The assessment of HRQoL was based on their component dimensions: the physical and mental component summaries (PCS and MCS, respectively) for the SF-36 and dyspnoea, fatigue, emotion and mastery subscale scores (CRQ-D, -F, -E and -M, respectively) for the CRQ. The BODE index, a multidimensional grading system, was also used15,16 (see online supplementary material).
Patients were instructed to contact healthcare teams in the event of any potential adverse events (e.g. cardiovascular episodes, strains or other types of injuries, etc.) and specific questions were asked at each visit. Clinical records were also used to keep track of any exacerbation episodes, regardless of whether they led to emergency department or hospital admission or were managed by general practitioners or pulmonologists on an outpatient basis. Exacerbations were defined as episodes of increased cough, purulent mucus production and worsening of symptoms that required administration of antibiotics and/or corticosteroids (see online supplementary material).
Compliance and AdherenceA patient in the IG arm was classified as a non-complier if he/she did not perform scheduled exercises for at least 8 consecutive weeks, unless there were clear clinical reasons not to. Patients were considered non-adherent if they did not attend any 2 follow-up appointments and there was no clear clinical justification for not attending. At each visit, patients were advised to continue doing exercise and attending follow-up appointments even if they decided not to comply with the assigned maintenance exercises, supporting the intention-to-treat principle (see online supplementary material).
Statistical AnalysisContinuous variables are described using either the mean and standard deviation or the median and range, depending on the data distribution. Categorical variables are described using frequencies and percentages. The main efficacy analysis used analysis of covariance (ANCOVA) techniques to test for differences between groups in outcome variables at the 12-month visit, adjusted for baseline values. Least squares mean differences and 95% confidence intervals were calculated. Secondly, additional analyses were conducted using linear mixed models, adjusted for baseline values and including time-group interaction terms. Although P-values lower than .05 were a priori considered statistically significant in two-sided tests, the threshold was adjusted for multiple comparisons using the Holm method whenever a P-value lower than .05 was found in a main effect test (group variable or time effect). Missing values were not imputed. All patients were analyzed in the group to which they were randomized. Stata 15.1 for Windows was used for all the analyses (StataCorp. 2017.Stata Statistical Software: Release 15. College Station, TX: StataCorp LLC).
Sample SizeThe sample size calculation was based on the effect of the intervention on exercise tolerance (6MWT distance) at 12 months as primary outcome: assuming superiority of the IG, we calculated that 70 patients (35 per group) would be required for a two-sided statistical comparison with a 5% type I error rate, to detect a clinically relevant difference of 35m between arms with a standard deviation of 50m and statistical power of 80%. The initial plan was to recruit 84 patients, allowing for a 20% dropout rate. This was subsequently altered, given recruitment pace and logistical capabilities. An extra 10 patients were recruited increasing the statistical power to 90%, while keeping all the other statistical considerations unchanged.
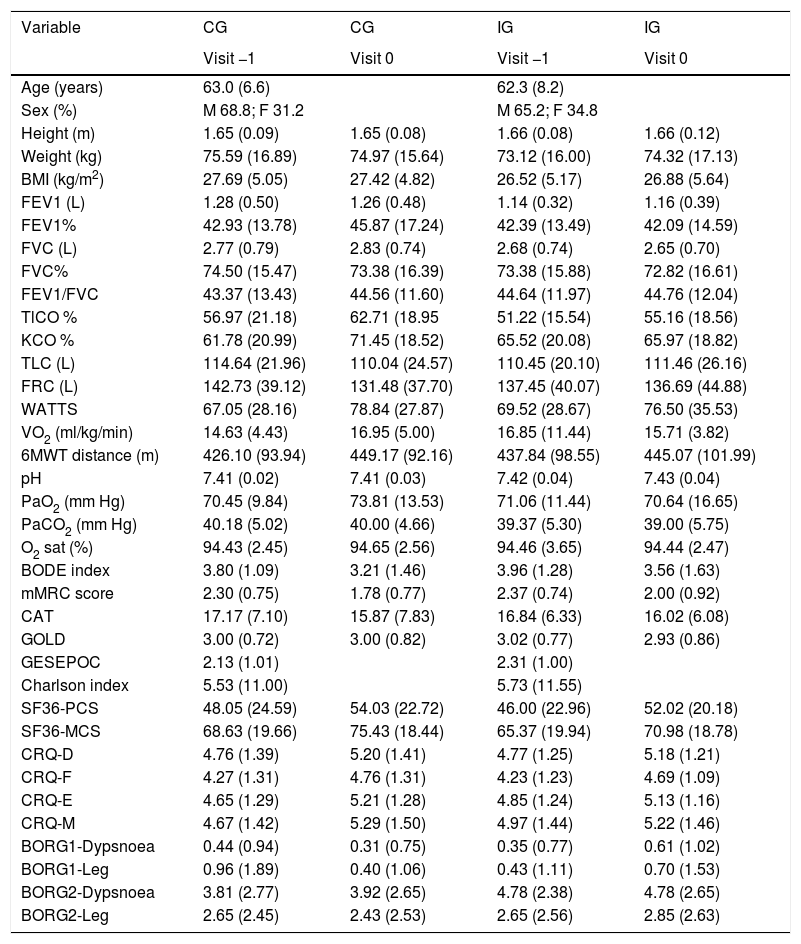
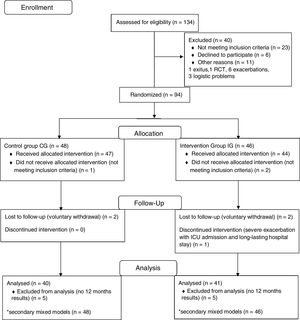
ResultsOverall, 134 patients were screened and 94 were eventually randomized into two groups: 48 to the CG and 46 to the IG (Fig. 1). Eighty-one patients (41 from the IG and 40 from the CG) with data from both baseline and 12-month visits were included in the ANCOVA. All available data from the randomized patients were used in the mixed models. Baseline characteristics for all participants are described in Table 1. No adverse events related to the home-based exercises were reported.
Characteristics of Patients at Enrolment and Randomization Visits.
| Variable | CG | CG | IG | IG |
|---|---|---|---|---|
| Visit −1 | Visit 0 | Visit −1 | Visit 0 | |
| Age (years) | 63.0 (6.6) | 62.3 (8.2) | ||
| Sex (%) | M 68.8; F 31.2 | M 65.2; F 34.8 | ||
| Height (m) | 1.65 (0.09) | 1.65 (0.08) | 1.66 (0.08) | 1.66 (0.12) |
| Weight (kg) | 75.59 (16.89) | 74.97 (15.64) | 73.12 (16.00) | 74.32 (17.13) |
| BMI (kg/m2) | 27.69 (5.05) | 27.42 (4.82) | 26.52 (5.17) | 26.88 (5.64) |
| FEV1 (L) | 1.28 (0.50) | 1.26 (0.48) | 1.14 (0.32) | 1.16 (0.39) |
| FEV1% | 42.93 (13.78) | 45.87 (17.24) | 42.39 (13.49) | 42.09 (14.59) |
| FVC (L) | 2.77 (0.79) | 2.83 (0.74) | 2.68 (0.74) | 2.65 (0.70) |
| FVC% | 74.50 (15.47) | 73.38 (16.39) | 73.38 (15.88) | 72.82 (16.61) |
| FEV1/FVC | 43.37 (13.43) | 44.56 (11.60) | 44.64 (11.97) | 44.76 (12.04) |
| TlCO % | 56.97 (21.18) | 62.71 (18.95 | 51.22 (15.54) | 55.16 (18.56) |
| KCO % | 61.78 (20.99) | 71.45 (18.52) | 65.52 (20.08) | 65.97 (18.82) |
| TLC (L) | 114.64 (21.96) | 110.04 (24.57) | 110.45 (20.10) | 111.46 (26.16) |
| FRC (L) | 142.73 (39.12) | 131.48 (37.70) | 137.45 (40.07) | 136.69 (44.88) |
| WATTS | 67.05 (28.16) | 78.84 (27.87) | 69.52 (28.67) | 76.50 (35.53) |
| VO2 (ml/kg/min) | 14.63 (4.43) | 16.95 (5.00) | 16.85 (11.44) | 15.71 (3.82) |
| 6MWT distance (m) | 426.10 (93.94) | 449.17 (92.16) | 437.84 (98.55) | 445.07 (101.99) |
| pH | 7.41 (0.02) | 7.41 (0.03) | 7.42 (0.04) | 7.43 (0.04) |
| PaO2 (mm Hg) | 70.45 (9.84) | 73.81 (13.53) | 71.06 (11.44) | 70.64 (16.65) |
| PaCO2 (mm Hg) | 40.18 (5.02) | 40.00 (4.66) | 39.37 (5.30) | 39.00 (5.75) |
| O2 sat (%) | 94.43 (2.45) | 94.65 (2.56) | 94.46 (3.65) | 94.44 (2.47) |
| BODE index | 3.80 (1.09) | 3.21 (1.46) | 3.96 (1.28) | 3.56 (1.63) |
| mMRC score | 2.30 (0.75) | 1.78 (0.77) | 2.37 (0.74) | 2.00 (0.92) |
| CAT | 17.17 (7.10) | 15.87 (7.83) | 16.84 (6.33) | 16.02 (6.08) |
| GOLD | 3.00 (0.72) | 3.00 (0.82) | 3.02 (0.77) | 2.93 (0.86) |
| GESEPOC | 2.13 (1.01) | 2.31 (1.00) | ||
| Charlson index | 5.53 (11.00) | 5.73 (11.55) | ||
| SF36-PCS | 48.05 (24.59) | 54.03 (22.72) | 46.00 (22.96) | 52.02 (20.18) |
| SF36-MCS | 68.63 (19.66) | 75.43 (18.44) | 65.37 (19.94) | 70.98 (18.78) |
| CRQ-D | 4.76 (1.39) | 5.20 (1.41) | 4.77 (1.25) | 5.18 (1.21) |
| CRQ-F | 4.27 (1.31) | 4.76 (1.31) | 4.23 (1.23) | 4.69 (1.09) |
| CRQ-E | 4.65 (1.29) | 5.21 (1.28) | 4.85 (1.24) | 5.13 (1.16) |
| CRQ-M | 4.67 (1.42) | 5.29 (1.50) | 4.97 (1.44) | 5.22 (1.46) |
| BORG1-Dypsnoea | 0.44 (0.94) | 0.31 (0.75) | 0.35 (0.77) | 0.61 (1.02) |
| BORG1-Leg | 0.96 (1.89) | 0.40 (1.06) | 0.43 (1.11) | 0.70 (1.53) |
| BORG2-Dypsnoea | 3.81 (2.77) | 3.92 (2.65) | 4.78 (2.38) | 4.78 (2.65) |
| BORG2-Leg | 2.65 (2.45) | 2.43 (2.53) | 2.65 (2.56) | 2.85 (2.63) |
Data are presented as mean (SD), unless otherwise stated. IG: intervention group; CG: control group; Visit −1 (enrolment visit) (treatment groups not yet established); Visit 0: random allocation visit; BMI: body mass index; FEV1: forced expiratory volume in 1s; FVC: forced vital capacity; % pred: % predicted; 6MWT: 6-minute walk test; TLC: total lung capacity; FRC: functional residual capacity; TLCO%: diffusion capacity of the lung for carbon monoxide %; TLCO% (TLCO/VA)%; VO2: Oxygen uptake; PaO2: Arterial O2 pressure; PaCO2: Arterial CO2 pressure; Sat: oxygen saturation; CAT:COPD assessment Test; GOLD: Global Initiative for Chronic Obstructive Lung Disease; Gesepoc: Guía Española de la Epoc; BODE: Body mass index, Airflow obstruction, Dyspnoea and Exercise capacity; mMRC: modified Medical Research Council Dyspnoea score; SF-36: 36-Item Short Form Health Survey; PCS: physical component score; MCS: mental component score; CRQ: Chronic Respiratory Disease Questionnaire; D: dyspnoea dimension F: fatigue dimension E: emotional function dimension M: mastery dimension; BORG1-Dyspnoea: dyspnoea perceived by the patient before bicycle exercises; BORG1-Leg: leg discomfort perceived by the patient before bicycle exercises; BORG2-Dyspnoea: dyspnoea perceived by the patient immediately after bicycle exercises; BORG2-Leg: leg discomfort perceived by the patient immediately after bicycle exercises.
No statistically significant differences between groups were found for any pre-specified endpoint (Table 2). After adjustment for baseline values, IG patients were able to walk on average almost 20m more in the 6MWT than controls (19.9m, 95% CI [−4.1/+43.8], P=.104) (Table 2). The CRQ-E score showed a difference in favour of the IG of 0.4 points (95% CI [0–0.8], P=.067). The analysis of the CRQ-D component showed a significant interaction between baseline score and intervention group (P=.023), with patients with lower baseline scores faring better in the CG, whereas the opposite was seen in patients with higher baseline scores.
Descriptive Data for Outcome Variables at Randomization (Visit 0) and Follow-up Visits.
| Descriptive Analysis | ANCOVA | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Outcome | Baseline | 3 Months | 9 Months | 12 Months | Difference (IG-CG) | 95% CI | P Value | ||||
| IG (n=46) | CG (n=48) | IG (n=41) | CG (n=40) | IG (n=38) | CG (n=32) | IG (n=38) | CG (n=39) | (n=77) | |||
| 6MWT m | 445.1 (102.0) | 449.2 (92.2) | 436.2 (113.3) | 447.7 (95.2) | 432.9 (117.1) | 423.9 (106.3) | 441.8 (106.3) | 423.7 (101.6) | 19.9 mts. | (−4.1/43.8) | .104 |
| Baseline | 3 Months | 9 Months | 12 Months | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| IG (n=46) | CG (n=48) | IG (n=42) | CG (n=37) | IG (n=38) | CG (n=35) | IG (n=41) | CG (n=40) | (n=81) | |||
| SF36 PCS | 52.0 (20.2) | 54.0 (22.7) | 50.1 (23.8) | 49.4 (24.2) | 45.8 (23.8) | 41.5 (22.7) | 46.1 (24.0) | 51.1 (24.6) | −2.2 | (−9.3/4.8) | .529 |
| SF36 MCS | 71.0 (18.8) | 75.4 (18.4) | 73.8 (16.0) | 63.8 (23.6) | 70.5 (18.4) | 59.4 (22.3) | 68.7 (19.6) | 67.0 (22.4) | 4.4 | (−3.4/12.2) | .262 |
| Baseline | 3 Months | 9 Months | 12 Months | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| IG (n=46) | CG (n=48) | IG (n=43) | CG (n=38) | IG (n=40) | CG (n=36) | IG (n=41) | CG (n=40) | (n=81) | |||
| CRQ-D | 5.2 (1.2) | 5.2 (1.5) | 5.2 (1.1) | 4.7 (1.5) | 4.9 (1.2) | 4.8 (1.3) | 4.8 (1.5) | 5.0 (1.3) | ** | ||
| CRQ-F | 4.7 (1.1) | 4.8 (1.3) | 4.7 (1.0) | 4.2 (1.4) | 4.5 (1.2) | 4.1 (1.2) | 4.5 (1.3) | 4.3 (1.5) | 0.3 | (−0.2/0.8) | .197 |
| CRQ-E | 5.1 (1.2) | 5.2 (1.3) | 5.3 (1.0) | 4.6 (1.4) | 5.1 (1.2) | 4.7 (1.3) | 5.1 (1.2) | 4.8 (1.3) | 0.4 | (0.0/0.8) | .067 |
| CRQ-M | 5.2 (1.5) | 5.3 (1.5) | 5.5 (1.2) | 4.9 (1.5) | 5.2 (1.5) | 5.0 (1.5) | 5.1 (1.4) | 5.0 (1.5) | 0.2 | (−0.4/0.7) | .546 |
| BODE | 3.6 (1.6) | 3.2 (1.5) | 3.5 (1.7) | – | 3.5 (1.7) | – | 3.7 (1.8) | 3.5 (1.8) | 0.0 | (−0.6/0.6) | .885 |
ANCOVA of between-group differences at 12-month visit. Data are presented as mean (SD) or mean (95% CI). ANCOVA: analysis of covariance; IG: intervention group; CG: control group; 6MWT: 6-min walk test; m.: metres; SF-36: 36-Item Short Form Health Survey; PCS: physical component score; MCS: mental component score; CRQ: Chronic Respiratory Disease Questionnaire; D: dyspnoea dimension F: fatigue dimension E: emotional function dimension M: mastery dimension; BODE: Body mass index, Airflow obstruction, dyspnoea and Exercise capacity.
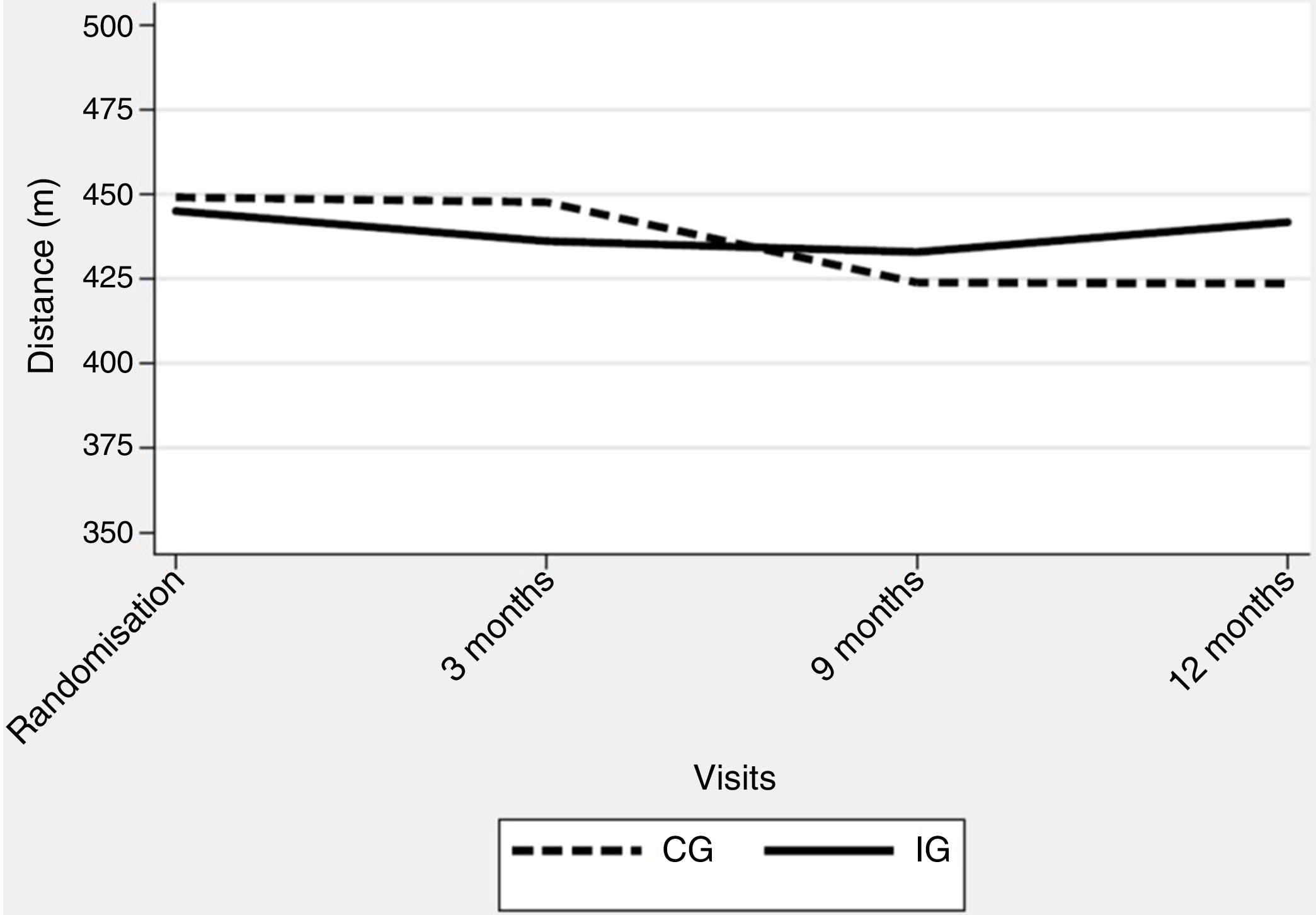
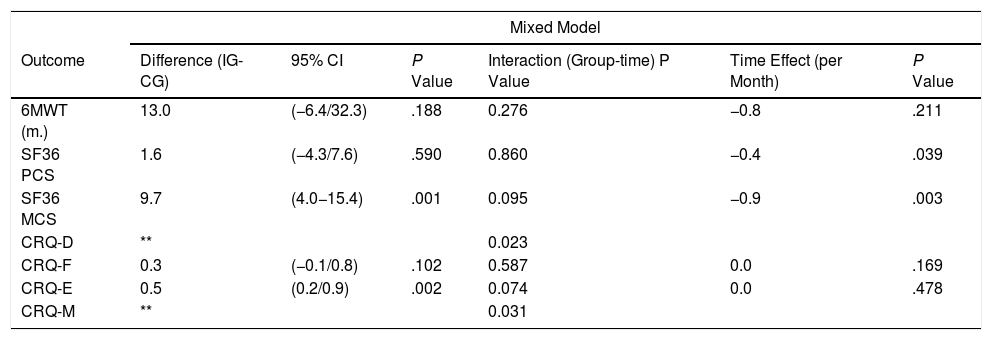
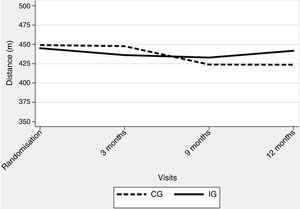
The corrected overall critical P-value was .007. No significant deterioration was observed over the study period, with an adjusted difference of 13.0m in 6MWT distance (95% CI −6.4/32.3; P=.188) in favour of the IG (Table 3) (Fig. 2). Regarding generic HRQoL, a significant deterioration was seen over the 12-month follow-up period in the SF-36 MCS (−0.9 points per month [P=.003]) but not in SF-36 PCS (−0.4 per month [P=.039]). Further, an adjusted significant difference of 9.7 points was found in the SF-36 MCS in favour of the IG (95% CI 4.0–15.4, P=.001) (Table 3).
Longitudinal Linear Analysis of Measures at 3, 9 and 12 Months, Adjusting for Values at Randomization Visit.
| Mixed Model | ||||||
|---|---|---|---|---|---|---|
| Outcome | Difference (IG-CG) | 95% CI | P Value | Interaction (Group-time) P Value | Time Effect (per Month) | P Value |
| 6MWT (m.) | 13.0 | (−6.4/32.3) | .188 | 0.276 | −0.8 | .211 |
| SF36 PCS | 1.6 | (−4.3/7.6) | .590 | 0.860 | −0.4 | .039 |
| SF36 MCS | 9.7 | (4.0−15.4) | .001 | 0.095 | −0.9 | .003 |
| CRQ-D | ** | 0.023 | ||||
| CRQ-F | 0.3 | (−0.1/0.8) | .102 | 0.587 | 0.0 | .169 |
| CRQ-E | 0.5 | (0.2/0.9) | .002 | 0.074 | 0.0 | .478 |
| CRQ-M | ** | 0.031 | ||||
IG: intervention group; CG: control group; 6MWT: 6-min walk test; m.: metres; SF-36: 36-Item Short Form Survey; MCS: mental component summary; PCS: physical component summary; CRQ: Chronic Respiratory Disease Questionnaire; D: dyspnoea dimension; F: fatigue dimension; E: emotional function dimension; M: mastery dimension.
Scores for CRQ dimensions did not change significantly over the follow-up period. Nonetheless, significantly different time trajectories (time-group interactions) were detected for the CRQ-D and CRQ-M components, with a difference of 0.5 points in the CRQ-E score in favour of the experimental arm (95% CI 0.2–0.9; P=.002) (Table 3) (see online supplementary material).
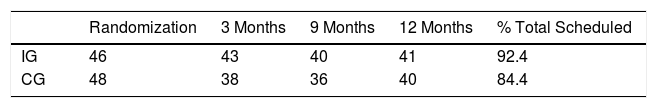
Adherence and ComplianceRates of adherence to the scheduled appointments were 92.4% in the IG and 84.4% in the CG (Table 4). Four patients in each group were non-attenders according to our prespecified definition. Overall compliance in the IG was 60% of the maximum scheduled days (weekly compliance is reported in the online supplementary material). Twelve patients in the IG were classified as non-compliers.
ExacerbationsThere were a total of 56 registered episodes of acute exacerbations in the IG, affecting 26 patients versus 47 in the CG affecting 21 patients, with no statistically significant differences between groups (Table 5). Including the fact that the patient had had at least one exacerbation episode as a covariate, ANCOVA indicated a relative increase in 6MWT distance of 23.2m (−1.4/47.7) (P=.064) in the IG. The occurrence of exacerbation was associated with an overall decrease of 14.9m (−39.6/9.7) (P=.231). Mixed models showed that IG patients were able to walk 14.9m more (−4.2/33.9) (P=.125) and the occurrence of exacerbation was associated with an overall decrease of 15.8m (−32.3/0.8) (P=.062).
Number of Exacerbations per Period Between Visits.
| No. Patients With ≥1 Exacerbation of Any Type per Period | ||||
|---|---|---|---|---|
| 0–3 Months | 3–9 Months | 9–12 Months | 0–12 Months | |
| IG | 10 | 14 | 17 | 26 |
| CG | 8 | 13 | 8 | 21 |
| No. Exacerbations (Any Type) per Period | ||||
|---|---|---|---|---|
| 0–3 Months | 3–9 Months | 9–12 Months | 0–12 Months | |
| IG | 11 | 22 | 23 | 56 |
| CG | 12 | 24 | 11 | 47 |
Top: Number of patients that showed exacerbation (the same patient may have an exacerbation in different periods). Bottom: Total number of exacerbations per period (the same patient may have more than one exacerbation in the same period). IG: Intervention Group; CG: Control Group.
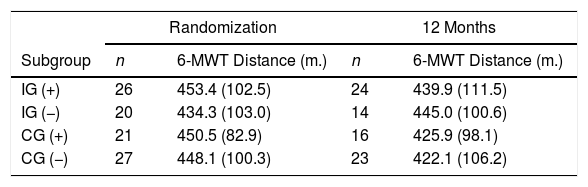
As illustrated in Table 6, only the subgroup of patients allocated to the IG who did not experience any exacerbation episode throughout the study period improved their exercise tolerance, although the difference did not reach significance.
Change Over Time in the 6-Minute Walk Test Distance in Each Group Stratified by the Presence of at Least One Exacerbation Episode.
| Randomization | 12 Months | |||
|---|---|---|---|---|
| Subgroup | n | 6-MWT Distance (m.) | n | 6-MWT Distance (m.) |
| IG (+) | 26 | 453.4 (102.5) | 24 | 439.9 (111.5) |
| IG (−) | 20 | 434.3 (103.0) | 14 | 445.0 (100.6) |
| CG (+) | 21 | 450.5 (82.9) | 16 | 425.9 (98.1) |
| CG (−) | 27 | 448.1 (100.3) | 23 | 422.1 (106.2) |
IG (+): IG and exacerbation; IG (−): IG no exacerbation; CG (+): CG and exacerbation; CG (−): CG no exacerbation. (m.): metres; data are presented as mean (SD).
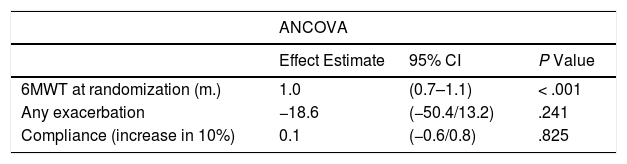
Table 7 shows, for the IG, the combined effect of exacerbation events and the degree of compliance on 6MWT distance, expressed in 10% increments of days with accomplished sessions over the total number of scheduled days. Occurrence of any exacerbation was associated with a decrease in the 6MWT of 18.6m (95% CI −50.4/13.2; P=.241) in the ANCOVA model. A 10% increase in compliance rate was associated with an increase in 6MWT distance by 0.1m (95% CI −0.6/0.8; P=.825). The mixed model showed similar results (Table 7).
Joint Effect of Several Variables on 6MWT Distance Walked at 12 Month Visit.
| ANCOVA | |||
|---|---|---|---|
| Effect Estimate | 95% CI | P Value | |
| 6MWT at randomization (m.) | 1.0 | (0.7–1.1) | < .001 |
| Any exacerbation | −18.6 | (−50.4/13.2) | .241 |
| Compliance (increase in 10%) | 0.1 | (−0.6/0.8) | .825 |
| Mixed Model | |||
|---|---|---|---|
| Effect Estimate | 95% CI | P Value | |
| 6MWT at randomization (m.) | 0.9 | (0.8–1.0) | < .001 |
| Any exacerbation | −14.6 | (−35.8/6.6) | .177 |
| Compliance (increase in 10%) | −0.1 | (−0.5/0.3) | .689 |
| Time (month of visit) | 0.7 | (−0.9/2.3) | .403 |
Top: Analysis of covariance including 6MWT distance walked at randomization (visit 0), occurrence of at least one exacerbation event and increase in compliance by 10% of scheduled training days. Bottom: Linear mixed model with the same explanatory variables and time.
Our randomized study has not demonstrated the hypothesized superiority of a telerehabilitation maintenance programme over usual maintenance care in patients with moderate-to-severe COPD after an 8-week intensive hospital-based rehabilitation programme. Neither clinically meaningful nor statistically significant differences were found in the main ANCOVA-based analyses after 12 months comparing maintenance exercises following advice from a physician, in line with common local practice, with those directly monitored through the use of a web-based platform with input data provided by the patient via a mobile phone. Overall compliance with the TelePR programme was good, allowing close monitoring of the frequency and quality of maintenance exercises completed. The intervention programme was also safe, with no significant major adverse effects detected during follow-up.
Despite overall negative findings, some results are worth looking at in detail. Firstly, patients in the IG showed, on average, minimal changes over the 12-month follow-up period, with a globally more stable pattern in exercise tolerance and several HRQoL dimensions (Fig. 2 and online supplementary material). This is compatible with the view that the TelePR programme would be beneficial as a maintenance strategy at least in some patients with COPD. Specifically, we observed an average relative improvement of nearly 20m in the 6MWT and nearly 0.5 points in several CRQ scores.
Secondly, our data, gathered in the context of a pragmatic trial, show that the selection criteria applied allowed enrolment of patients with a highly variable clinical and functional baseline status. This is reflected, for instance, in the huge variation observed in baseline 6MWT distance (range 150–650m). This high dispersion in initial data might have hampered the ability of the trial to show potential overall benefits of the intervention.
Thirdly, our control group also had only minor deterioration in most endpoints over the course of the study. That might be due to the fact that regular visits to a specialized clinic along with reminders to keep active could also exert some positive effect. Interestingly, similar patterns were detected in several outcome variables, with early differences in favour of the IG, reaching a peak at the 9-month visit and subsequent attenuation of differences by the last (12-month) visit. Whether these time profiles are related to a short-time effect of the telerehabilitation intervention or to other factors remains unclear.
Lastly, our secondary analysis, with a linear mixed model, that makes use of all available longitudinal data, showed both clinically and statistically significant improvements in two dimensions related to quality of life (SF-36 MCS and CRQ-E) (Table 2). In addition, significantly different time profiles were detected in CRQ-D and CRQ-M, showing a relevant difference achieved at month 3 visit with progressive loss of differences at later visits (see online supplementary material).
Maintenance StrategiesSeveral studies have described difficulties in maintaining the benefits obtained after an intensive PR programme. Unfortunately, improvements achieved during intensive rehabilitation schedules tend to diminish over time.17–20 Possible contributing factors include programme duration, intensity, location, and psychosocial factors.21–23 Some research has suggested that supervised post-rehabilitation exercise programmes could be effective in preserving exercise capacity.24,25 In a 12-month study, Spencer26 did not find any difference between two groups doing once-a-week supervised outpatient-based exercise and unsupervised exercise, both maintaining the improvements obtained in the initial phase over the study duration.
TelerehabilitationTelemedicine-based interventions promoted through phone calls, websites, or mobile phones have shown promising results in COPD patients and several studies have demonstrated the feasibility of these programmes.26–28,4,29 In our study, all patients were able to use the technology and it was not necessary to withdraw any patients for difficulties managing the devices provided.
Most telehealth studies reported have major methodological limitations such as a short duration, small sample sizes and high dropout rates.30–34 There is also a paucity of evidence on the long-term benefits of telerehabilitation interventions conducted to help maintain benefits gained during initial rehabilitation. One of the two studies found is a small non-controlled study of 10 COPD patients attending PR and followed up with a telerehabilitation programme over 2 years by Zanaboni et al., 7 and they concluded that exercise maintenance via telerehabilitation was feasible and effective, achieving improvements in physical performance, health status and quality of life. The other is a randomized study of the same length as ours conducted by Vasilopoulou et al.,8 in which 6MWT distance (a secondary endpoint) was stable in both active interventions: multimodal home-based maintenance and hospital-based maintenance rehabilitation groups, with a similar pattern to that in our TelePR group. They only found significant deterioration in exercise tolerance in their control group, which did not receive any initial intensive PR intervention.
Exacerbations and ComplianceIn our study, we have also observed that the occurrence of exacerbation episodes during the maintenance period can have a major influence on exercise tolerance, to such an extent that it may even counterbalance any potential benefit of a planned regular maintenance strategy (Tables 6 and 7).
Level of compliance with the planned maintenance exercises was not a significant predictor of final physical performance in our study, either alone or after accounting for the patient having experienced at least one acute exacerbation episode (Table 7). The deleterious influence of exacerbations on functional capacity is widely acknowledged. Symptoms and physiological performance usually return to baseline pre-exacerbation levels after 7 (range 4–14) days for dyspnoea, but full recovery is not achieved in all patients even more than 1 month after the start of the acute event.22 In relation to this, our estimates of around 16m of deterioration in 6MWT distance associated with at least one acute exacerbation episode are, to the best of our knowledge, the first data provided on the potential magnitude of this detrimental effect during a PR maintenance programme. Alahmari et al. state that having attended a PR programme in the past is associated with an attenuated short-term impact of exacerbation on exercise tolerance, but their patients were not undergoing an active maintenance intervention.35
Furthermore, the only subgroup in our trial that showed some improvement in exercise tolerance at the end of the maintenance period, although not statistically significant, was that composed of IG patients without any exacerbation event during the study (Table 5). We suggest that any clinical study aimed at assessing the effects of PR maintenance programmes on physical, emotional and HRQoL endpoints should also carefully register the occurrence, main features and clinical consequences of acute exacerbations as a way to gain deeper insight into the potential effect-modifying role played by these common intercurrent events. Frequency and/or intensity of exacerbations could also be used as an additional selection criterion for inclusion of a COPD patient on a maintenance programme or adaption of such a programme to specific clinical characteristics.
There is little experience on the issue of whether interventions using telerehabilitation programmes have a protective effect and reduce the rate of exacerbations. A recent study8 reports a protective effect of a telerehabilitation programme on the number of hospital admissions due to COPD acute exacerbations. In our study, as in others,26 the number of episodes of acute exacerbation was indeed higher in the IG (56 episodes versus 47 in the CG), but without statistical significance (Table 5). Although all patients were instructed to report any adverse events and medical records were systematically assessed to identify exacerbation events, it cannot be ruled out the possibility that the TelePR programme, with an accurate tally of any period of physical inactivity, could strengthen the detection and reporting of intercurrent episodes.
There is general agreement that compliance with long-term maintenance programmes is often low and decreases over time. In our study, the rate of compliance with the TelePR was 60% of the overall planned exercise days. We do not know of any other study that has given such a detailed account of the extent of accomplishment of planned exercises (see online supplementary material). If we focus on attendance to scheduled appointments, our rates are higher than those reported by most studies regardless of their design. Brooks19 in a study involving 109 patients followed over 1 year observed that 24 (22%) did not return for follow-up appointments after the baseline visit and less than half attended the last visit. Further, Spencer26 reported an adherence rate of 30% at 12 months. Zanaboni7 observed that rates of daily card registration decreased from 42.9% to 13.9% over the 2-year study, and Guell36 in a 3-year randomized trial reported adherence rates of 66% and 17% for the IG and CG respectively. In contrast, the multimodal home-based maintenance TelePR intervention by Vasilopoulou et al.8 showed notably high adherence rates (of over 90%) to each component of their telehealth strategy.
LimitationsThe design of the study as a pragmatic trial and the correspondingly broad inclusion criteria led to recruitment of a study population with substantial clinical and functional heterogeneity which, in turn, may have diminished the likelihood of observing either clinically meaningful or statistically significant differences. Another limitation is the lack of assessment of compliance with exercise recommendations in the control group. The design of the study meant that this group were supposed to continue maintenance training according to current usual care in the recruiting hospitals. Their degree of adherence was defined according to the number of follow-up appointments attended, which is a non-specific indicator of the amount and type of maintenance exercise actually performed. It is likely that other types of compliance monitoring techniques, such as daily cards, could have given more accurate information on exercise activity in this group.
Lastly, although our rate of withdrawals and losses to follow-up is lower than that reported in most other published studies, our analysis has also had to deal with some missing data. In this regard, there was an unexpected slight increase in the number of patients attending the last appointment compared to previous ones. This phenomenon cannot be explained by the pattern of occurrence of exacerbation events but perhaps might be the result of clinicians making intensive efforts to record functional and health status of their patients at the last study appointment.26
ConclusionWe have not shown overall superiority of our TelePR maintenance strategy over our own current clinical practice. Longitudinal analyses have shown improvements in some HRQoL domains and a more stable time pattern in the IG.
Our approach to a TelePR maintenance programme is both feasible and safe and allows close monitoring of maintenance exercise. Nonetheless, we found no association between level of compliance and extent of functional maintenance in our IG.
We have identified a potentially important negative influence of exacerbations on final physical function and only patients in the intervention subgroup with no exacerbations in the study period seemed to increase their average 6MWT distance from baseline by 12 months.
Further research is warranted using more homogeneous COPD populations, considering in the design initial exercise capacity, the observed effect of the intensive PR phase and the clinical profile of the patient, especially regarding the frequency and severity of acute exacerbation episodes.
Prior Abstract PublicationPartial data have been presented as a poster (“Challenges and opportunities for independent clinical research”) at the Scientific Meeting of the Spanish Clinical Research Network (SCReN), Madrid, 9 June 2016 and as a poster discussion (“A Respiratory Telerehabilitation programme as maintenance in patients with chronic obstructive pulmonary disease”) at the European Respiratory Congress, Paris, 15–19 September 2018. We have no conflicts of interest to disclose.
ContributorsJBG and JIP conceived the study and devised the design. JIP planned and carried out the statistical analysis plan and interpreted the results. JBG and JIP drafted the manuscript. All authors critically reviewed the manuscript draft, made suggestions and approved the final version prior to submission.
CollaboratorsLuis Paz del Rio, Macarena Segura, Fátima Morante, Jaime Navarro, Leire Argoitia, Juan Agüero, Elena López de Santa María, Liliana Pérez.
DisclaimerThe funders of the trial had no role in trial design, data collection, data analysis, data interpretation or writing of the report.
Patient Consent for PublicationObtained.
Ethics ApprovalBasque Country Ethics Committee (code 2014011).
Data Availability StatementData are available on reasonable request.
Conflict of InterestThe authors have not conflict of interest to disclose.
The authors would like to thank the participants involved in the trial, the nurses and rehabilitation personnel at each study site, and the editors of Ideas Need Communicating Language Services for their help with improving the use of English in the manuscript (supported by the Basque Foundation for Research and Innovation in Health [BIOEF]). They are also very grateful to the Spanish Clinical Research Network (SCReN) for its support, and especially to Ana Irasarri (SCReN project manager) for her gracious and tenacious backing in many aspects of the trial management.