Respiratory syncytial virus (RSV) is a major cause of respiratory tract infections in adults, particularly older adults and those with underlying medical conditions. Vaccination has emerged as a potential key strategy to prevent RSV-related morbidity and mortality. This Neumoexperts Prevention (NEP) Group scientific paper aims to provide an evidence-based positioning and RSV vaccination recommendations for adult patients. We review the current literature on RSV burden and vaccine development and availability, emphasising the importance of vaccination in the adult population. According to our interpretation of the data, RSV vaccines should be part of the adult immunisation programme, and an age-based strategy should be preferred over targeting high-risk groups. The effectiveness and efficiency of this practice will depend on the duration of protection and the need for annual or more spaced doses. Our recommendations should help healthcare professionals formulate guidelines and implement effective vaccination programmes for adult patients at risk of RSV infection now that specific vaccines are available.
Journal Information
Vol. 60. Issue 3.
Pages 161-170 (March 2024)
Vol. 60. Issue 3.
Pages 161-170 (March 2024)
Review Article
Respiratory Syncytial Virus Vaccination Recommendations for Adults Aged 60 Years and Older: The NeumoExperts Prevention Group Position Paper
Visits
1438
Esther Redondoa, Irene Rivero-Calleb,c,d, Enrique Mascaróse, Daniel Ocañaf, Isabel Jimenog, Ángel Gild,h, Manuel Linaresi, María Ángeles Onieva-Garcíaj,k, Fernando González-Romol, José Yusted,m, Federico Martinón-Torresb,c,d,◊,
Corresponding author
a Infectious, Migrant, Vaccines and Preventive Activities Group of SEMERGEN, International Healthcare Centre of the City Council of Madrid, Madrid, Spain
b Translational Paediatrics and Infectious Diseases Section, Paediatrics Department, Hospital Clínico Universitario de Santiago de Compostela, Santiago de Compostela, Spain
c Genetics, Vaccines, and Infections Research Group (GENVIP), Healthcare Research Institute of Santiago de Compostela, University of Santiago de Compostela, Santiago de Compostela, Spain
d CIBER of Respiratory Diseases (CIBERES), Instituto de Salud Carlos III, Madrid, Spain
e Health Department, Hospital la Fe, Primary Care Centre Arquitecto Tolsá, Valencia, Spain
f Primary Care, Health Care Centre Algeciras, Algeciras, Spain
g Primary Care Health Centre Isla de Oza, Vaccine Responsible of SEMG, Madrid, Spain
h Preventive and Public Health, Rey Juan Carlos University, Madrid, Spain
i Specialist in Primary Care and Clinical Microbiology, Infectious Diseases Group SEMERGEN, Fundación io, Madrid, Spain
j Preventive Medicine and Public Health Unit, Hospital Universitario Reina Sofía, Cordoba, Spain
k Maimonides Biomedical Research Institute of Cordoba (IMIBIC), Spain
l Clinical Microbiology Department, Hospital Universitario Clínico San Carlos, Madrid, Spain
m National Centre for Microbiology, Instituto de Salud Carlos III, Madrid, Spain
Ver másRelated content
Esther Redondo, Irene Rivero-Calle, Enrique Mascarós, Daniel Ocaña, Isabel Jimeno, Ángel Gil, Manuel Linares, María Ángeles Onieva-García, Fernando González-Romo, José Yuste, Federico Martinón-Torres
Podcast
Article information
Abstract
Full Text
Bibliography
Download PDF
Statistics
Tables (6)
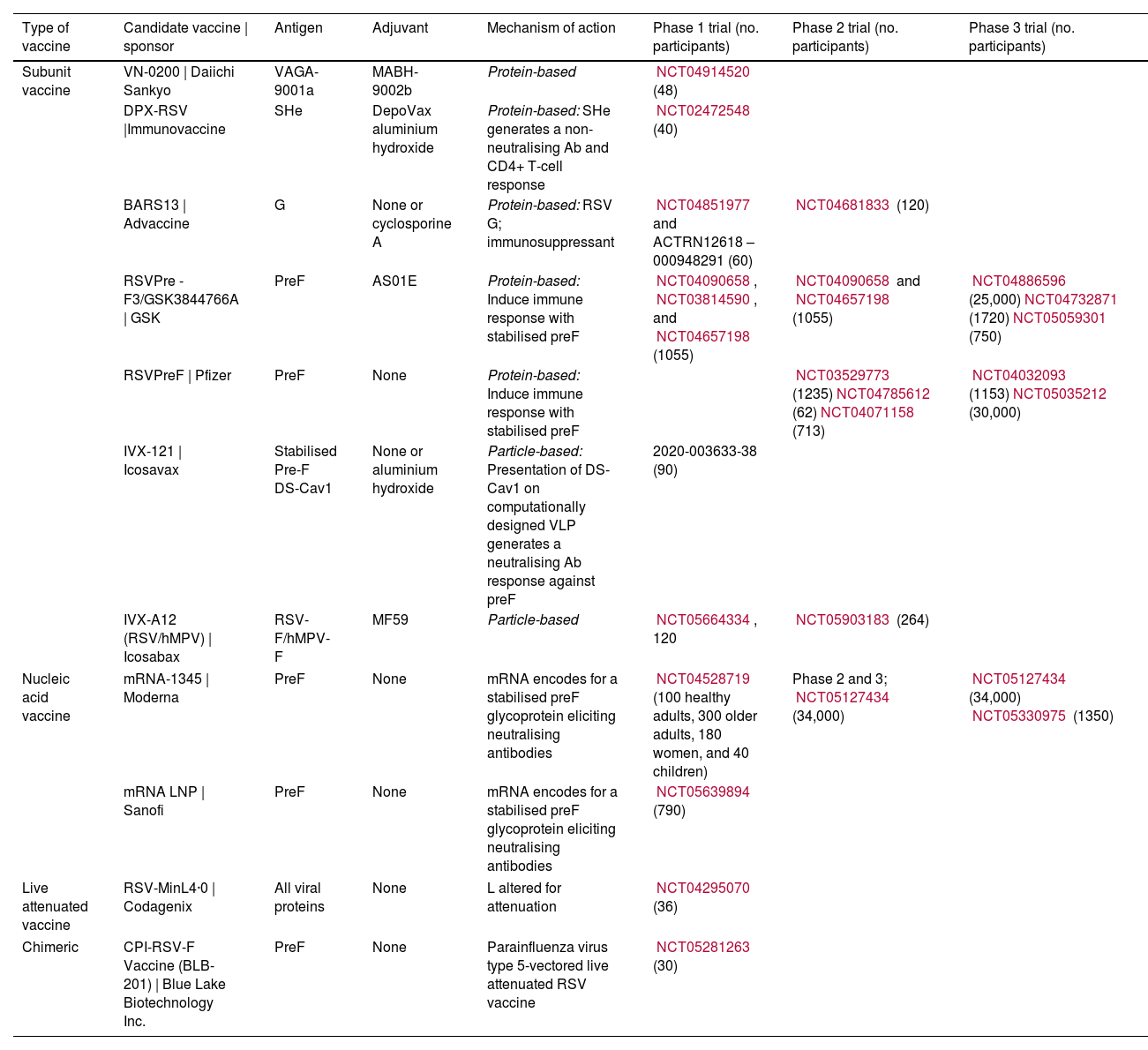
Table 1. Summary of Vaccine Candidates in Development for Older Adults Categorised by Design Strategy.11
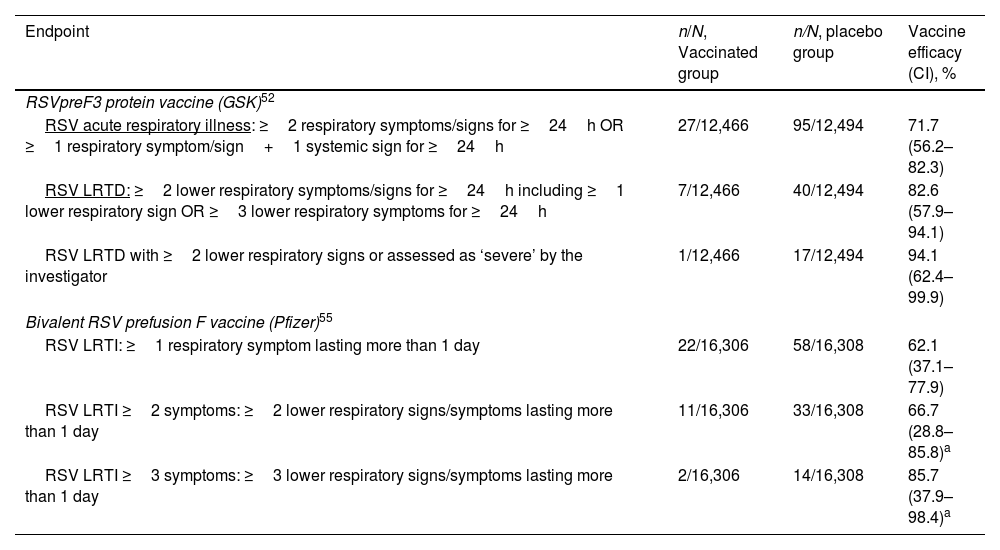
Table 2. Efficacy of Currently Licensed RSV Vaccines for Adults 60 Years or Older Based on Phase 3 Trial Results.52,55
Table 3. Efficacy of One Dose of Currently Licensed RSV Vaccines for Adults Aged ≥60 Years During the First RSV Season, the Second RSV Season and Both Seasons, 2021–2023.53,54
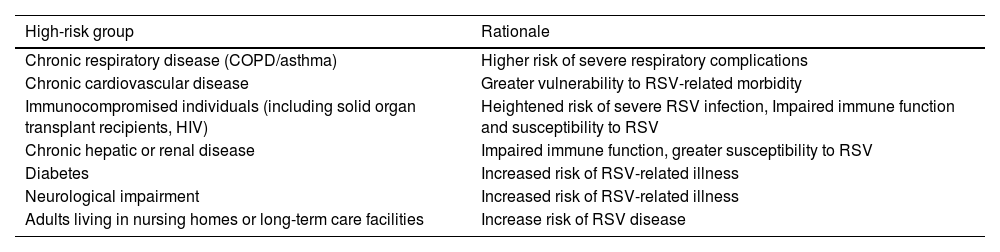
Table 4. High-risk Groups for RSV Vaccination Recommendations Irrespective of Age.
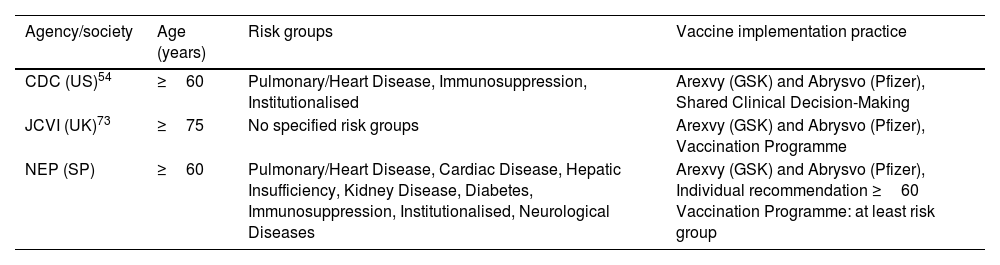
Table 5. Official Recommendations on the Use of Licensed RSV Vaccines for Older Adults.
Table 6. Summary of NEP Group Positioning on RSV Vaccination in Older Adults.
Show moreShow less
Abstract
Keywords:
Respiratory syncytial virus
RSV
Adult patients
Vaccination
Recommendations
Preventive measures
High-risk populations
Chronic obstructive pulmonary disease
These are the options to access the full texts of the publication Archivos de Bronconeumología