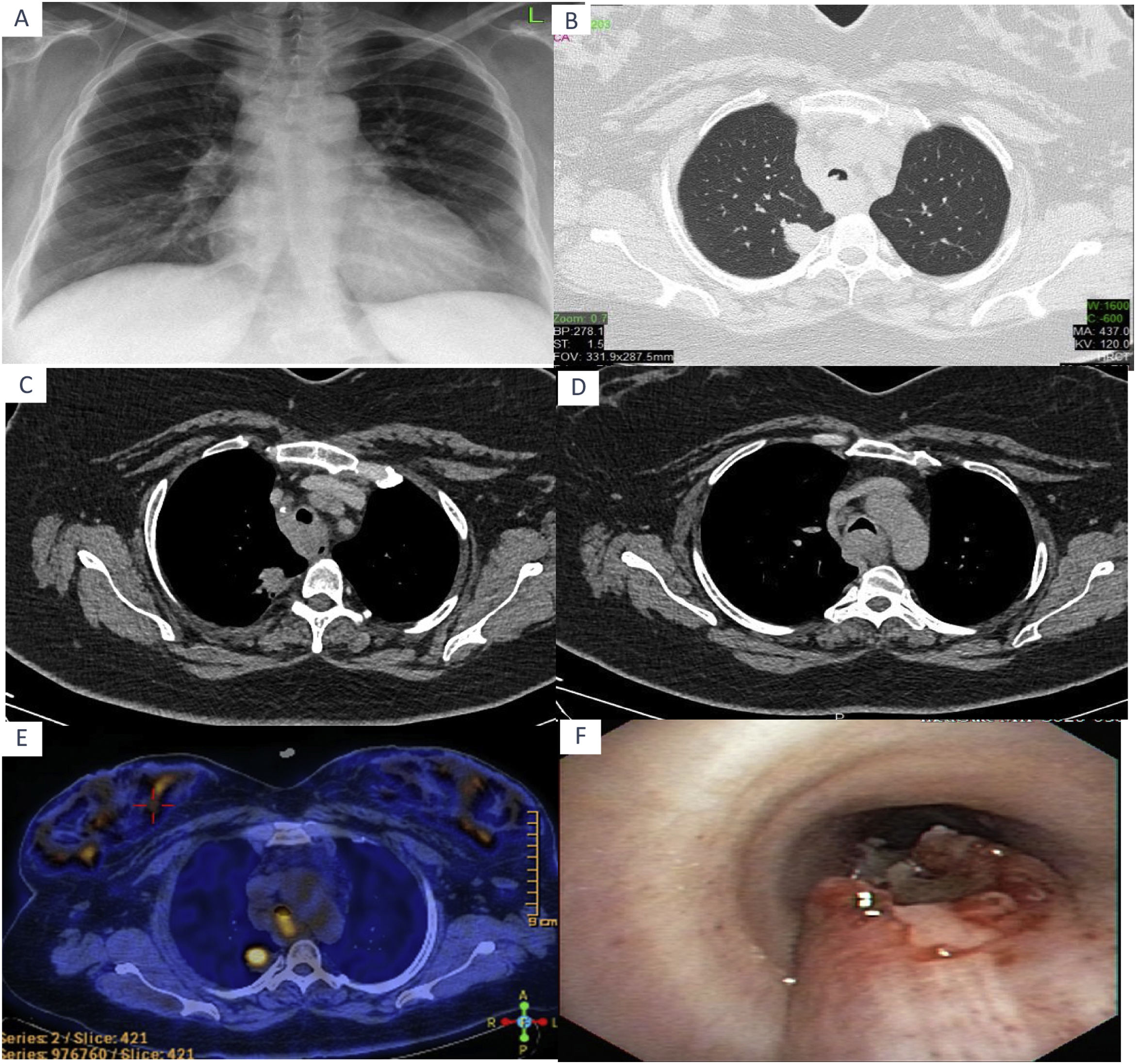
Pulmonary hyalinizing granuloma (PHG) first described by Engleman et al. in 1977, was generally reported as individual cases in the world literature.1 In most cases, PHG occurs as solitary or multiple pulmonary nodules with well defined boundaries.2 They usually grow slowly or may not grow at all; Although positron emission tomography CT is useful in ruling out metastatic lesions, increased metabolic activity may occur in PHG lesions.3 In histological analysis, homogeneous hyaline lamellae are usually detected in a perivascular distribution, surrounded by the collection of plasma cells, lymphocytes and histiocytes.2 Due to frequent coexistence with infectious, autoimmune and tumoral diseases, an abnormal immune reaction has been identified to explain the development of PHG.1,2 Twenty five percent of patients are asymptomatic. The most common symptoms in symptomatic patients are cough, shortness of breath and chest pain.1,4 Although corticosteroids show some effectiveness, usually specific treatment is not required.5 A 39 years old female patient with no complaints was admitted to our clinic with the suspicion of a mass in the right upper zone of the mediastinum on the chest X-ray taken for control purposes before her job application (Fig. 1A). She had no known chronic disease and no history of smoking. Physical examination findings were detected to be normal. In the thorax CT, an irregularly bounded mass lesion with a diameter of 14mm at the shortest and 31mm at the longest was detected in the upper lobe posterior segment in the right lung. At the same time there were irregularities in the trachea. Conglomerate lymphadenopathies which were calcified in places and whose boundaries were indistinguishable from the esophagus lumen in the lower paratracheal and subcarinal area were detected in the mediastinal upper-lower paratracheal, at prevascular subcarinal localization (Fig. 1B–D). PET CT was requested for the patient. 18F-FDG uptake (SUVmax: 7.1) was observed in a 27mm×23mm lesion in the paravertebral region in the posterior segment of the right lung upper lobe. In the right lateral neighborhood of the esophagus, at the carina level 18F-FDG uptake (SUVmax: 6.5) was observed in lesion with a diameter of approximately 1cm. (Fig. 1E). First, flexible bronchoscopy was planned for diagnosis taking into account endobronchial control. In flexible bronchoscopy, lesion invasive toward the posterior wall and vascularized at the mid level of the trachea was observed, bronchoscop could not be progressed toward its distal region (Fig. 1F). Numerous bronchial biopsy and bronchial lavages were taken. Pathologies were not diagnostic. Tuberculosis EZN and its culture were negative. Transthoracic trucut biopsy was performed to the lesion in the posterior of the right lung upper lobe. In pathology; thick, keloid-like collagen fibers and inflammatory cell infiltration rich in lymphoids were observed. Reactive lymphoid population with antigenic properties was observed. Because the patient was asymptomatic and the physical examination findings were normal, interventional or systemic treatment was not considered. In the 3rd month follow-up thorax CT, the lesions remained stable, she still had no symptoms. PHG is rare, difficult to diagnose and should be kept in mind in patients who are followed up with suspicion of malignancy but cannot be diagnosed.3
(A) Chest X-ray. (B) In the Thorax CT, an irregularly bounded mass lesion in the upper lobe posterior segment in the right lung and irregularities in the trachea. (C, D) In the Thorax CT Mediastynal conglomerate lymphadenopathies. (E) Focally increased 18F-FDG uptake is observed in the nodular lesion in the right upper lobe and at the carina level. (F) Invasive lesion to the posterior wall at the middle level of the trachea in bronchoscopy.
GK writes first version of the manuscript, and MD completed all documents. GK and MD were responsible for the treatment of the patient. MD supervised the treatment and the completion of this case report. Two authors read and approved the final manuscript.
Availability of data and materialsThe data are available from the authors upon reasonable request.
Consent for publicationWritten informed consent was obtained from the patient for the publication of this case report and any accompanying images. The data presented in this case report do not allow identification of the patient.
FundingNo funding was obtained for this study.
Conflicts of interestsNo conflicts of interest were declared by the authors.