Lungs represent the major site of infection in the intensive care unit (ICU).1 Bacteria recovered from patients undergoing mechanical ventilation (MV) can be part of the resident microbiome so its role as causative agents is still an unresolved issue. Colonization may be persistent even when the clinical symptoms improve.2Staphylococcus aureus remains a leading cause, both when methicillin susceptible (MSSA) and methicillin-resistant (MRSA). Colonization at ICU admission is a risk factor for developing pneumonia3,4 but clinical features can range from asymptomatic carriage to severe invasive disease, demonstrating an adaptation and switch in virulence regulation.5 It is of paramount importance to distinguish colonization from infection and to adequately prescribe antimicrobials.
This was a retrospective and observational study conducted during 2 years (2012–2013). Patients on MV admitted at ICU were selected based on the isolation of S. aureus in endotracheal aspirate (ETA). The objectives were to investigate host and microbial factors associated with (1) clinical outcome and (2) persistent isolation despite treatment adjusted to antibiotic susceptibility profile.
ETA and blood cultures were performed when there was clinical suspicion of respiratory tract infection, not systematically. Nasal swabs were obtained to check MRSA carriage. Study was performed according to confidentiality criteria and dissociation of patients’ identification data. Data recorded included epidemiological features, presence of comorbidities, Glasgow Coma Scale (GCS), central nervous system (CNS) involvement and severity of illness at admission assessed by Acute Physiology and Chronic Health Evaluation (APACHE)-II.
Patients (all with S. aureus ETA culture) were classified into pneumonia, tracheobronchitis or bronchial colonization by expert clinicians according to Clinical Pulmonary Infection Score (CPIS) and radiographic findings.6 Ventilator associated pneumonia was defined by presence of pulmonary infiltrate and CPIS ≥6. Ventilator associated tracheobronchitis was defined when there were fever (>38°C) with no other recognizable cause of purulent secretion and no radiographic signs of new pneumonia. Bronchial colonization was defined in the absence of the aforementioned clinical and radiological features. Data regarding antimicrobial therapy was recorded. Isolation in ETA was defined persistent when it lasted ≥7 days despite treatment adjusted to susceptibility profile.
Adverse clinical outcome was defined as the development of respiratory complications following definitions from Ferrando et al.,7 including empyema and septic shock. When after initial improvement, there was need for acute changes in ventilator support to enhance oxygenation, the patient was also considered to have a respiratory complication. Mortality was also recorded. VAP might lead to prolonged ICU length of stay (LOS) and prolonged MV so these parameters were also analyzed to define outcome. Clinical strains were phenotypically characterized by conventional identification and susceptibility testing methods. Antimicrobial treatment prescribed was recorded, and categorized as β-lactamcs, vancomycin, quinolones and linezolid.
Pearson's chi-square or Fisher test was applied for categorical variables and student's T test or ANOVA for numerical variables. Univariate/multivariate analyses were performed. Variables included in multivariate models were selected according to the univariate analysis results. Associations were considered statistically significant if p<0.05. Data were analyzed with SPSS v15 (SPSS Inc, Chicago, IL).
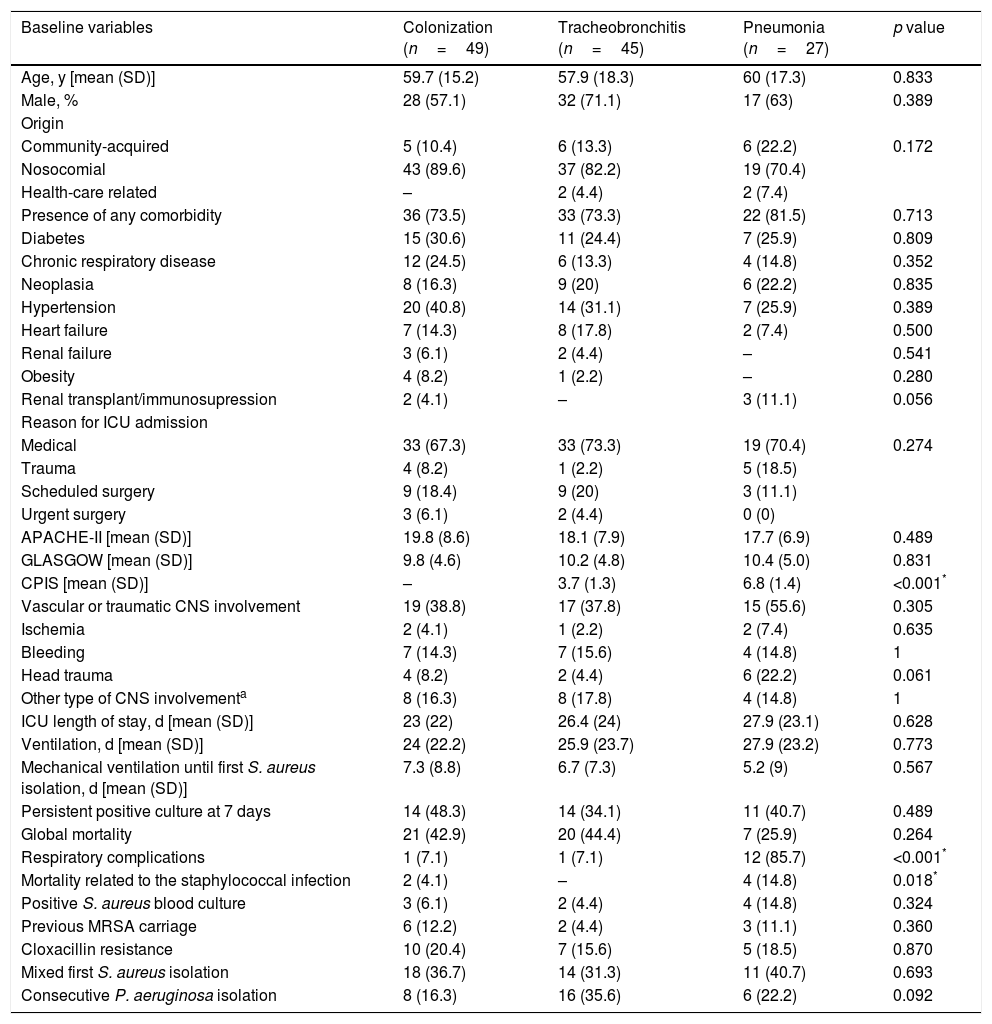
During the study period, the global incidence of VAP was 1.61 cases for 1000 days of stay and 2.34 cases for 1000 days of MV. Of the 3012 ETA samples collected, S. aureus was isolated in 270 (9%) corresponding to 121 patients classified as: pneumonia (27), tracheobronchitis (45) and bronchial colonization (49). Patients’ epidemiological and clinical characteristics and comparisons according to the study group are shown in Table 1. Among the 27 patients with pneumonia diagnosis, chest X-ray results were as follows; 13 cases were unilobar (48.2%), 12 multilobar (44.4%) and 2 had a diffuse pulmonary infiltrate (7.4%). Vascular or traumatic CNS involvement was frequent (42.1%) (previously described risk factor for S. aureus colonization). None of the variables allowed a satisfactory distinction between groups.
Patients epidemiological and clinical characteristics; and comparisons according to the study group considered.
| Baseline variables | Colonization (n=49) | Tracheobronchitis (n=45) | Pneumonia (n=27) | p value |
|---|---|---|---|---|
| Age, y [mean (SD)] | 59.7 (15.2) | 57.9 (18.3) | 60 (17.3) | 0.833 |
| Male, % | 28 (57.1) | 32 (71.1) | 17 (63) | 0.389 |
| Origin | ||||
| Community-acquired | 5 (10.4) | 6 (13.3) | 6 (22.2) | 0.172 |
| Nosocomial | 43 (89.6) | 37 (82.2) | 19 (70.4) | |
| Health-care related | – | 2 (4.4) | 2 (7.4) | |
| Presence of any comorbidity | 36 (73.5) | 33 (73.3) | 22 (81.5) | 0.713 |
| Diabetes | 15 (30.6) | 11 (24.4) | 7 (25.9) | 0.809 |
| Chronic respiratory disease | 12 (24.5) | 6 (13.3) | 4 (14.8) | 0.352 |
| Neoplasia | 8 (16.3) | 9 (20) | 6 (22.2) | 0.835 |
| Hypertension | 20 (40.8) | 14 (31.1) | 7 (25.9) | 0.389 |
| Heart failure | 7 (14.3) | 8 (17.8) | 2 (7.4) | 0.500 |
| Renal failure | 3 (6.1) | 2 (4.4) | – | 0.541 |
| Obesity | 4 (8.2) | 1 (2.2) | – | 0.280 |
| Renal transplant/immunosupression | 2 (4.1) | – | 3 (11.1) | 0.056 |
| Reason for ICU admission | ||||
| Medical | 33 (67.3) | 33 (73.3) | 19 (70.4) | 0.274 |
| Trauma | 4 (8.2) | 1 (2.2) | 5 (18.5) | |
| Scheduled surgery | 9 (18.4) | 9 (20) | 3 (11.1) | |
| Urgent surgery | 3 (6.1) | 2 (4.4) | 0 (0) | |
| APACHE-II [mean (SD)] | 19.8 (8.6) | 18.1 (7.9) | 17.7 (6.9) | 0.489 |
| GLASGOW [mean (SD)] | 9.8 (4.6) | 10.2 (4.8) | 10.4 (5.0) | 0.831 |
| CPIS [mean (SD)] | – | 3.7 (1.3) | 6.8 (1.4) | <0.001* |
| Vascular or traumatic CNS involvement | 19 (38.8) | 17 (37.8) | 15 (55.6) | 0.305 |
| Ischemia | 2 (4.1) | 1 (2.2) | 2 (7.4) | 0.635 |
| Bleeding | 7 (14.3) | 7 (15.6) | 4 (14.8) | 1 |
| Head trauma | 4 (8.2) | 2 (4.4) | 6 (22.2) | 0.061 |
| Other type of CNS involvementa | 8 (16.3) | 8 (17.8) | 4 (14.8) | 1 |
| ICU length of stay, d [mean (SD)] | 23 (22) | 26.4 (24) | 27.9 (23.1) | 0.628 |
| Ventilation, d [mean (SD)] | 24 (22.2) | 25.9 (23.7) | 27.9 (23.2) | 0.773 |
| Mechanical ventilation until first S. aureus isolation, d [mean (SD)] | 7.3 (8.8) | 6.7 (7.3) | 5.2 (9) | 0.567 |
| Persistent positive culture at 7 days | 14 (48.3) | 14 (34.1) | 11 (40.7) | 0.489 |
| Global mortality | 21 (42.9) | 20 (44.4) | 7 (25.9) | 0.264 |
| Respiratory complications | 1 (7.1) | 1 (7.1) | 12 (85.7) | <0.001* |
| Mortality related to the staphylococcal infection | 2 (4.1) | – | 4 (14.8) | 0.018* |
| Positive S. aureus blood culture | 3 (6.1) | 2 (4.4) | 4 (14.8) | 0.324 |
| Previous MRSA carriage | 6 (12.2) | 2 (4.4) | 3 (11.1) | 0.360 |
| Cloxacillin resistance | 10 (20.4) | 7 (15.6) | 5 (18.5) | 0.870 |
| Mixed first S. aureus isolation | 18 (36.7) | 14 (31.3) | 11 (40.7) | 0.693 |
| Consecutive P. aeruginosa isolation | 8 (16.3) | 16 (35.6) | 6 (22.2) | 0.092 |
ICU: intensive care unit, CNS: central nervous system, APACHE-II: Acute Physiology and Chronic Health Evaluation II, CPIS: clinical pulmonary infection score.
Initial S. aureus isolation was mixed with another microorganism in 35.5% of cases: Haemophilus influenzae (16.3%), Pseudomonas aeruginosa (14%) and other Gram negative bacilli (58.1%). P. aeruginosa was subsequently isolated in 30 patients (24.8%). Twenty-one patients (17.3%) had a positive blood culture, and in 9 cases, the isolated microorganism was S. aureus (7.4%).
Respiratory adverse clinical outcome, including mortality related was associated with diagnosis of pneumonia (p<0.001) and staphylococcal bacteraemia (p=0.022) in univariate analysis, and only pneumonia in multivariate (p=0.001; OR: 38.4; IC95%: 4.606–320.165). Interestingly, clinical outcome was similar regardless of cloxacillin resistance. No statistical differences were found when considering ICU LOS, days on MV, age, APACHE-II and GCS.
When considering global mortality, it accounted for 48 patients (39.7%), and 6 cases were staphylococcal infection related. Mortality was significantly associated with the presence of comorbidities (p=0.017), positive S. aureus blood culture (p=0.028), previous MRSA carriage (p=0.025) and consecutive isolation of P. aeruginosa in respiratory sample (p=0.052). Patients that died were older (p=0.004) and had a higher APACHE index at admission (p<0.001).
Selecting the 97 patients that received antimicrobial treatment adjusted to susceptibility profile, persistence at ≥7days was documented in 39 cases (40.2%), being 27 MSSA and 12 MRSA. Persistence was frequent independently of the antimicrobial used (p=0.036). In the univariate analysis, persistence correlated with: younger age (p=0.009), higher CPIS (p=0.041), ICU LOS (p<0.001) and days on MV (p<0.001), cloxacillin resistance (p=0.016) and consecutive isolation of P. aeruginosa (p<0.001). In multivariate only age (p=0.003; OR: 0.949; IC 95%: 0.918–0.982) and cloxacillin resistance (p=0.003; OR: 7.891; IC 95%: 1.980–31.454) remained significant. Correlation to younger age could be associated to differences in antibiotic pharmacokinetics/pharmacodynamics and inflammatory response. An important aspect is the absence of differences regarding clinical outcome, suggesting that persistence is more related to bacterial adaptation.2 Potential reasons for persistence could be a limited antimicrobial penetration into lung parenchyma, together with the ability of S. aureus to reside intracellularly within host cells and to produce biofilm.8 The impact of long-term effect of persistent bacterial isolation remains to be elucidated. Persistent isolation was more frequent in MRSA cases, as expected since antimicrobial treatments such as glycopeptides have poor penetration into alveolar tissues and less intrinsic activity than β-lactams.9 Still, MSSA isolates were also recovered from persistent cases indicating that cloxacillin resistance or type of antimicrobial are not the only factors intervening in this phenotype. Adequacy of treatment was considered according to the results of in vitro susceptibility testing, but antimicrobial characteristics such as lung penetration, bactericidal/bacteriostatic/intracellular activity were not analyzed.
S. aureus has a complex set of virulence factors, with defined implications in severity in community acquired infections but less clear in nosocomial.10 In recent years, preclinical investigations using monoclonal antibodies against virulence factors have shown promising results.11 Regulatory systems, such as the accessory gene regulator (agr) control virulence factors secretion and its dysfunction has been related to persistent bacteremia and higher mortality. However, we have previously shown that about 80% of isolates from the respiratory tract had a functional agr with no correlation with unfavorable outcome.12
In summary, our study suggests that persistent S. aureus isolation despite adjusted antibiotic treatment is often reported in MV patients, with no correlation with adverse clinical outcome. Although it is more frequent in MRSA cases, it is also reported for MSSA isolates. Thus, in order to optimize antimicrobial treatment, besides considering positive bacterial culture result it will also be important to identify microbial factors that contribute to virulence in order to initiate a highly-focused personalized therapy.
FundingThis work has been funded by the project PI13/01418 which is part of “Plan Nacional de I+D+I” and co-funded by ISCIII – Subdirección General de Evaluación and “Fondo Europeo de Desarrollo Regional” (FEDER). This work also received a grant from the Spanish Society of Pneumology and Thoracic Surgery (SEPAR 054/2011). None of the funding sources had a role in study design, collection, analysis and interpretation of data; in the writing of the report; and in the decision to submit the article for publication.
We thank Irma Casas for statistical assessment, Isabel Carrasco for her technical assistance and Maisem Laabei for his critical review.