Pulmonary hemorrhage is a potentially life-threatening condition that sometimes requires immediate intervention to stop the bleeding. When the source of the bleeding is not identifiable, for example, in alveolar hemorrhage, therapeutic options are limited and include correction of the coagulation defects, along with life-sustaining treatment as needed.1–3
In this context, tranexamic acid (TA), a synthetic antifibrinolytic agent, has been approved for the oral or intravenous treatment or prophylaxis of bleeding episodes in patients with coagulation disorders. Based on its effectiveness in the control of local mucosal bleeding (nose, colon, rectum, and mouth), it has been evaluated for use in pulmonary hemorrhage of different etiologies.4–6
We report the case of an 18-year-old man, diagnosed with idiopathic pulmonary hemosiderosis, who had received several lines of treatment, and who was currently receiving corticosteroids, hydroxychloroquine, and immunoglobulins.
He was admitted to the ICU with a clinical picture of dyspnea and frank hemoptysis, requiring orotracheal intubation due to global respiratory failure. Chest X-ray showed bilateral infiltrates, and anemia with a 2g/dL decrease in hemoglobin from baseline was detected. Fiberoptic bronchoscopy was performed, showing the bronchial mucosa covered with red blood, surging up from both lower lobes. Antimicrobial treatment began with a wide-spectrum antimicrobial and the corticosteroid dose was increased to 1mg/kg/12h.
The patient's progress in the following hours was poor, and collection of bloody fluid from the orotracheal tube persisted. Anemia developed again with a fall in hemoglobin levels of 1.5g/dL, and ventilation and oxygenation became more difficult, with reduced lung compliance. Chest X-ray showed increased patchy bilateral consolidations. Fiberoptic bronchoscopy was repeated, showing aspiration of fresh blood from both lower lobes.
The patient's clinical status continued to worsen and diffuse alveolar hemorrhage was suspected. Treatment began with nebulized TA on day 3 of admission, and antimicrobial treatment, sedation, and analgesia were maintained. A 22mm Cirrus®2 nebulizer breathing kit was used for nebulization. Using this system and a gas flow of 8L/min, 77% of the output volume contains particles at least 5μm in diameter with a mean mass diameter of 2.7μm.7
We used a TA dose of 500mg/5mL/12h, during which time the patient remained on mechanical ventilation.
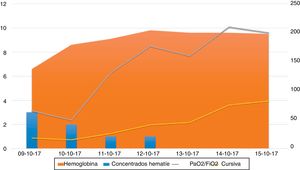
A significant improvement was observed in the following days, with no new episodes of bleeding or anemia, and the patient's respiratory status improved, so sedation could be discontinued. He awakened correctly and was weaned from mechanical ventilation, with extubation on day 9 of hospitalization. Figure 1 describes the course of the event, in terms of lung compliance, PaO2/FiO2, anemia, and need for transfusion during the first 7 days in the ICU, at which time mechanical ventilation weaning maneuvers were initiated.
Course of the event, described in terms of hemoglobin levels and the need for transfusion (number of packed red blood cell units) on the left vertical axis of the figure. The right vertical axis shows the course of lung compliance and PaO2/FiO2 until the start of mechanical ventilation weaning maneuvers.
Diffuse alveolar hemorrhage, whether idiopathic or associated with hematological disorders is an entity with a mortality rate ranging between 70% and 90%.8,9
TA, a synthetic derivative of the amino acid lysine, has anti-fibrinolytic activity, as it binds with plasminogen, thus inhibiting fibrin binding and subsequent plasmin activation.4
A Cochrane review identifies two clinical trials which evaluate the use of oral and intravenous TA (Anchafibrin®).10 There is insufficient evidence to recommend its use, but some small studies suggest that it may reduce hemorrhage duration. However, a review article33 of the published series concludes that, while a recommendation with strong evidence cannot be given, TA can reduce both bleeding duration and volume, with a low short-term risk of thromboembolic disease (weak recommendation, 2B).11
The recommendation to administer nebulized TA in pulmonary hemorrhage may be based on pathophysiological evidence that it increases the activity of anti-fibrinolytic factors that are depleted in sites of continuous bleeding throughout the bronchial tree. Studies indicate that the drug is more effective when administered locally rather than systemically, suggesting that local administration provides improved, more durable inhibition of fibrinolysis, with less systemic absorption, thus reducing the risk of thrombosis associated with the latter. Indeed, the local application of TA has proven efficacy in the control of bleeding of different etiologies, with no significant adverse effects.12,13
The data suggest that administering TA by inhalation is an effective, inexpensive, safe, and easy-to-manage technique, making it a good option for the treatment of pulmonary hemorrhage in emergency rooms or even general hospitalization units.
However, no cause-and-effect relationship can be inferred, as favorable progress may be due to a spontaneous resolution of the case. For this reason, we believe that additional prospective randomized clinical trials are needed to evaluate the use of TA in this syndrome, since treatment duration and dose have not yet been fully clarified.
Please cite this article as: González-Castro A, Rodriguez-Borregán JC, Chicote E, Escudero P, Ferrer D. Ácido tranexámico nebulizado como alternativa de tratamiento en la hemorragia pulmonar. Arch Bronconeumol. 2018;54:442–443.