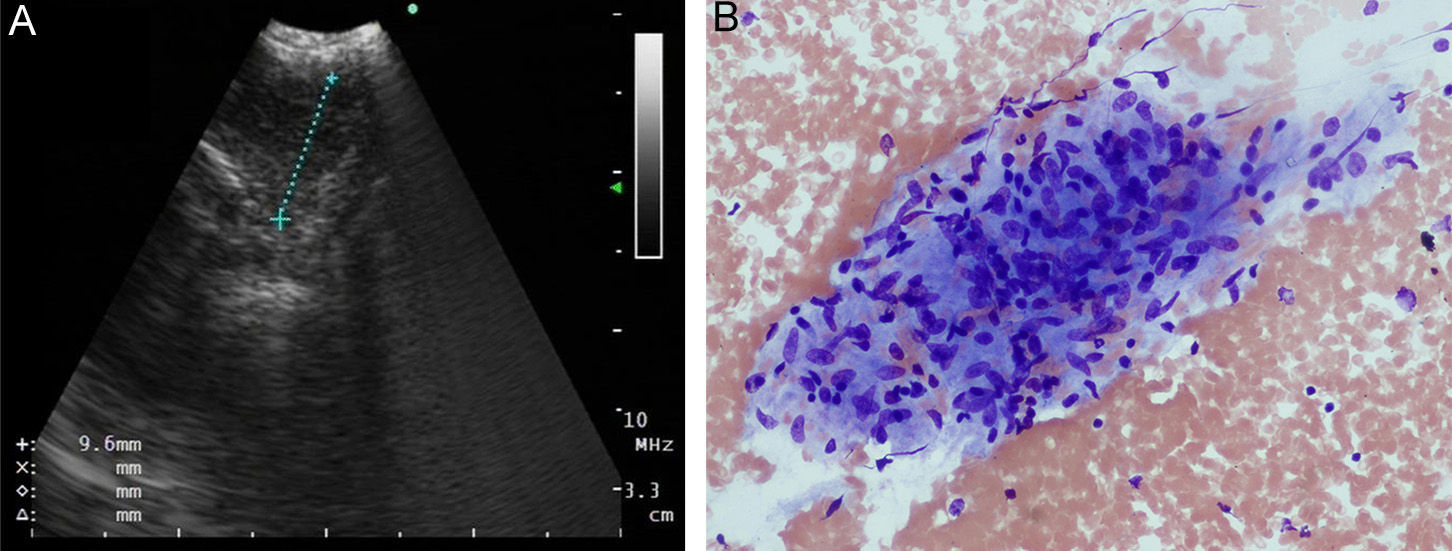
A 38-year-old man was admitted to our department with a 1-month history of chest pain and sensitivity that had gradually worsened in the previous few days. Systemic examination and vital signs were normal. His medical record indicated a history of asymptomatic microscopic hematuria diagnosed as bladder carcinoma 3 years previously. He had completed 16 cycles of bacillus Calmette–Guérin (BCG) approximately 16 months earlier. A whole body bone scan was performed to rule out distance metastasis in the chest wall, but after detecting increased activity at the level of the 5th rib, we decided to perform whole body positron emission tomography/computed tomography (PET/CT) with 18F-fluoro-2-deoxy-d-glucose (18F-FDG PET/CT). This revealed increased activity (SUVmax: 5.6) in lymph node station 11L, in the left hilar region. Although the preliminary diagnosis had been primary bladder carcinoma metastasis, histological analysis of the samples obtained by endobronchial ultrasound-guided fine needle aspiration (Fig. 1A) revealed granulomatous lymphadenitis (Fig. 1B). The patient therefore received empirical quadruple pharmacological tuberculosis treatment, consisting of isoniazide (300mg/day), rifampicin (600mg/day), ethambutol (800mg/day) and pyrazinamide (1g/day) for 6 months. Follow-up PET performed 6 months later showed a decrease in FDG uptake (SUVmax: 1.1) compared to previous levels. The patient did not report any discomfort at his 1-year check-up.
BCG is an important adjuvant treatment option for reducing the risk of recurrence and progression of bladder carcinoma.1 Although rare, intravesical BCG treatment can present systemic and localized complications. Most localized adverse effects are generally self-limiting, and include hematuria, dysuria, cystitis, prostatitis, and orchitis. Systemic adverse effects, however, are less common, and can range from simple febricula to potentially fatal multi-organ failure.2
Several cases of pulmonary or extra-pulmonary granulomatous lymphadenitis secondary to BCG instillation have been described in the English-language literature. However, there is considerable debate on whether the infectious complications secondary to BCG are due to a hypersensitivity reaction or to active infection. Some studies have found viable bacteria in different tissues, such as the lung, pancreas and liver, suggesting active infection.3,4 The hypersensitivity hypothesis, meanwhile, is supported by studies in which microorganisms could not be isolated.5 This was the case in our patient, as we were unable to demonstrate the presence of acid-fast bacilli in 3 sputum samples and 1 bronchoalveolar lavage sample; culture results were also negative.
In summary, this case is of interest insofar as it illustrates a characteristic complication of intravesical BCG instillation. We hope this report will highlight the importance of bearing in mind the different complications of intravesical BCG treatment, even when there is clinical suspicion of BCG infection.
Conflict of interestsThe authors have no conflict of interests or funding to declare.
Please cite this article as: Dogan D, Zor M, Ozkisa T, Ayten O, Yigit N, Yavas I. Linfadenitis granulomatosa mediastínica tras tratamiento con bacilo Calmette–Guérin intravesical que simula metástasis distante de carcinoma vesical primario. Arch Bronconeumol. 2015;51:526–527.