Statins [hydroxyl-methyl-glutaryl-coenzyme-A reductase (HMGCR) inhibitors] are used to treat patients with hypercholesterolemia. One recently described adverse effect of these drugs is necrotizing autoimmune myopathy (NAM).1–3 In statin-induced NAM, patients present with subacute symmetrical proximal limb weakness and elevated serum levels of the muscle enzyme, creatine kinase (CK). The clinical course is severe, and patients present histologically significant necrosis of muscle fibers with minimal or no inflammation.2,3
To date, no association between statin-induced NAM and interstitial lung disease (ILD) has been reported in the literature, and only one publication has addressed the use of rituximab in NAM.4 We report the first case of ILD secondary to statin-induced NAM, correlating muscle disease with pulmonary functional deterioration, responding to rituximab.
A 52-year-old male patient complained of a 3-year history of myalgia, proximal limb muscle weakness (grade 3) and progressive breathlessness (currently grade 2 according to the Modified Medical Research Council dyspnea scale), which started 3 months after taking rosuvastatin for dyslipidemia. He also had diabetes mellitus and hypothyroidism, and was a non-smoker.
Statin use was withdrawn due to elevated CK (3000U/L, reference values <150U/L) and aldolase (19.3U/L, reference values <7.6U/L) levels; however, the side effects persisted. He was hospitalized due to increasing CK levels (7000U/L). Physical examination was normal, and oxygen saturation (SpO2) was 96% on room air. Electroneuromyography showed mild proximal myopathy. The biceps brachii muscle was biopsied, and histopathology indicated homogeneous muscle size, with moderate necrosis in muscle fibers without significant inflammation (CD4+/CD8+ negative and CD 68+ positive). MHC I on the surface of all muscle fibers was positive. Additional laboratory tests revealed normal serum levels of thyroid hormones, negative autoimmune antibodies, and negative serologies for hepatitis and HIV.
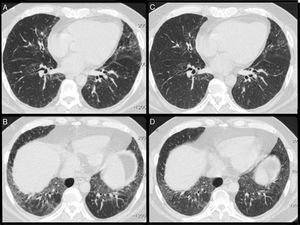
Lung function tests (LFTs) showed a restrictive pattern with reduction of carbon monoxide diffusion capacity (DLCO) (Table 1). High-resolution chest computed tomography (CT) showed a pattern of non-specific interstitial pneumonia (Fig. 1A and B).
Drug Regimens, Lung Function Parameters and Clinincal Laboratory Results During Treament.
| Diagnosis | May/13 | Aug/13 | Dec/13 | Apr/14 | Jun/14 | Aug/14 | Nov/14 | |
|---|---|---|---|---|---|---|---|---|
| Drug regimen | No drug | Aza150mg Pred40mg | Aza 200mg Pred 20 | MMF 1g | CyC IV (5th pulse) | 1st month after Rtx | 3rd month after Rtx | 6th month after Rtx |
| FVC | 2.24 (53%) | 3.28 (71%) | 2.71 (59%) | 2.65 (60%) | 2.73 (59%) | 2.63 (58%) | 2.78 (63%) | 2.81 (63%) |
| FEV1 | 2.02 (60%) | 2.86 (77%) | 2.29 (62%) | 2.27 (64%) | 2.34 (63%) | 2.19 (61%) | 2.40 (67%) | 2.43 (68%) |
| TLC | 3.75 (58%) | 4.18 (63%) | 4.38 (65%) | 4.05 (63%) | 3.82 (59%) | 4.14 (64%) | 4.05 (63%) | 4.25 (63%) |
| RV | 1.20 (62%) | 0.90 (44%) | 1.52 (34%) | 1.35 (68%) | 1.09 (50%) | 1.31 (65%) | 1.21 (60%) | 1.44 (71%) |
| DLCO | 19.50 (58%) | 20.38 (62%) | 22.1 (66%) | 21.2 (67%) | 21.2 (67%) | 22 (67%) | 18.1 (56%) | 20.3 (63%) |
| MIP | NA | −88 (72%) | −60 (49%) | NA | NA | NA | −81 (67%) | NA |
| MEP | NA | 78 (63%) | 62 (50%) | NA | NA | NA | 70 (68%) | NA |
| CK | >7000 | 90 | 193 | 713 | 1994 | NA | 744 | 148 |
| Aldolase | 19.3 | 4.6 | 6.3 | 9.4 | 27.2 | NA | 14.7 | 4.9 |
Aza: azathioprine (∼3mg/kg/day); CyC: cyclophosphamide (750mg/m2); MMF: mycophenolate mofetil, RTX: rituximab (two infusions of 1000mg, two weeks apart); Pred: prednisolone; FVC: forced vital capacity; FEV1: forced expiratory volume in 1second; TLC: total lung capacity; RV: residual volume; DLCO: carbon monoxide diffusion capacity; MIP: maximal inspiratory pressure; MEP: maximal expiratory pressure; CK: creatine kinase; NA: not available.
(A and B) High-resolution computed tomography showing raised diaphragm, ground glass opacities, interlobular thickening, and traction bronchiectasis predominantly in the lower lobes, a pattern compatible with non-specific interstitial pneumonia. (C and D) CT scans after rituximab treatment showing improvement in ground glass opacities.
Statin-induced NAM was diagnosed and the patient was treated with intravenous methylprednisolone and human immunoglobulin. He was discharged with prednisone (40mg/day) and azathioprine. Clinical, laboratory and PFT findings improved gradually after 3 months. Prednisone was gradually tapered to 20mg/day, but muscle strength and dyspnea continued to worsen, while CK and aldolase levels rose. Azathioprine was switched to mycophenolate mofetil but later discontinued due to gastrointestinal intolerance. Cyclophosphamide (CyC) was then introduced monthly (750mg/m2), but after six CyC doses, a high dose of prednisone had to be maintained (Table 1). Rituximab was then introduced (1000mg, 2 weeks apart), leading to reduced CK levels and improvements in LFTs and CT results (Fig. 1C and D).
Rituximab, a chimeric monoclonal anti-CD20 antibody, has been used as a rescue drug in the treatment of refractory myositis and ILD associated with antisynthetase syndrome5 Our case demonstrated a successful outcome with rituximab, and one possible explanation for this response could be that B cell depletion prevents not only persistent autoantibody production in NAM but also antigen presentation and interaction with other T cells.
A major limitation of our case report is that anti-HMGCR autoantibodies were not analyzed. Nevertheless, the temporal association between the onset of symptoms and statin use, the presence of significant necrosis without inflammation on muscle biopsy, and the requirement of intense immunosuppression allowed a confident diagnosis of statin-related NAM.
We emphasize the importance of considering statins as potential etiologic factors of ILD. Patients with statin-related NAM should be actively tested for ILD, and rituximab seems to have a role in refractory cases.
Please cite this article as: Dias OM, Baldi BG, Costa AN, Shinjo SK, Miossi R, Kairalla RA. La enfermedad pulmonar intersticial con miopatía autoinmune necrosante asociada a estatinas responde al rituximab. Arch Bronconeumol. 2016;52:395–397.