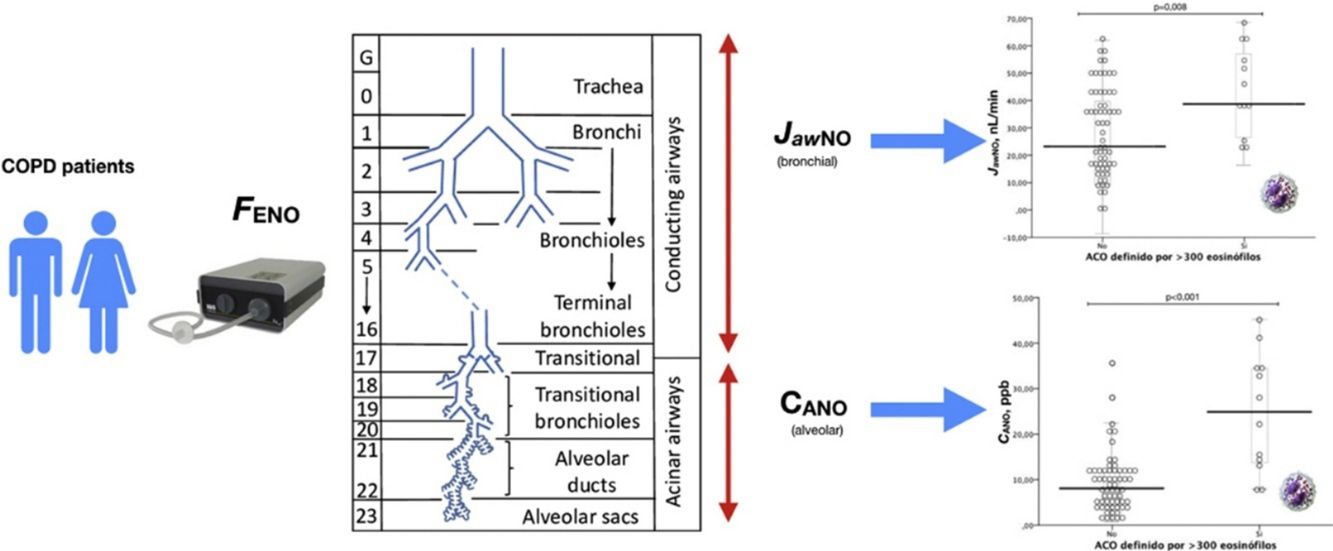
Exhaled nitric oxide (FENO) measurements differentiate COPD phenotypes from asthma–COPD overlap (ACO). To date, no study has been conducted to determine whether alveolar and bronchial components differ in this group of patients.
MethodsThis was an observational cross-sectional study recruiting ambulatory COPD patients. FENO was measured, differentiating alveolar (CANO) from bronchial (JawNO) components using a multiple-flow technique. CANO and JawNO values were compared between eosinophilic COPD patients (defined as ≥300 eosinophils/μL in peripheral blood test, or ≥2% eosinophils or ≥3% eosinophils), and a linear regression analysis was performed to determine clinical and biological variables related to these measurements.
Results73 COPD patients were included in the study. Eosinophil counts were associated with increased values of CANO and JawNO (for the latter only the association with ≥300 or ≥3% eosinophils was significant). CANO was also associated with CRP, and JawNO with smoking.
ConclusionsPatients with COPD and ACO characteristics show increased inflammation in the large and small airways. CANO and JawNO are associated with clinical and biological variables.
La medición del óxido nítrico en el aire exhalado diferencia fenotipos de pacientes con EPOC del solapamiento de asma y EPOC (ACO). Hasta el momento no se ha estudiado si existen diferencias entre los componentes alveolar y bronquial del FENO en este grupo de pacientes.
MétodosEstudio observacional transversal realizado en consultas externas de Neumología, incluyendo a pacientes con diagnóstico de EPOC a los que se les realizó una determinación del óxido nítrico en aire exhalado – FENO– diferenciando en esta medida el componente alveolar –CANO– y el de vía aérea central –JawNO–, y realizando las mediciones a distintos flujos. Se compararon los valores de CANO y JawNO entre los pacientes con eosinofilia (definidos como aquellos pacientes con ≥300 eosinófilos/μL en sangre periférica, o bien ≥2% eosinófilos o ≥3% eosinófilos) y se realizó un análisis de regresión lineal para estudiar las variables clínicas y biológicas que se asociaban a estas mediciones.
ResultadosParticiparon en el estudio 73 pacientes con EPOC. Los criterios de eosinofilia utilizados se asociaban a incrementos de los valores de CANO y de JawNO (en este último caso solo los criterios ≥300 eosinófilos y ≥3% eosinófilos). CANO se asoció al recuento de eosinófilos y PCR, y JawNO se asoció a tabaquismo y recuento de eosinófilos.
ConclusionesLos pacientes diagnosticados de EPOC y que tienen características de ACO muestran mayor inflamación a nivel bronquial y de vía aérea pequeña. CANO y JawNO se relacionan con variables clínicas y biológicas.
Chronic obstructive pulmonary disease (COPD) is defined as chronic, irreversible airflow limitation, which is usually progressive and associated with an abnormal inflammatory reaction mainly caused by smoking.1,2 This characteristic inflammatory response in COPD is mediated by macrophages, neutrophils, and cytotoxic T-cells (CD9+), and is accompanied by structural changes that can cause narrowing of the airways, changes in the arteries, pulmonary parenchymal emphysema, or combinations of all three.3 These structural changes begin in the early stages of the disease,4 especially in the small airways. In some patients, the inflammatory response is mediated primarily by Th2 lymphocytes and eosinophils, generating clinical symptoms that share features with bronchial asthma. Although the characteristics have not been clearly defined, this syndrome is currently known as asthma–COPD overlap (ACO).5–7 One of the biological factors distinguishing COPD patients with this phenotype is their eosinophil count in peripheral blood,8 but so far, no optimal cut-off points have been clearly established.
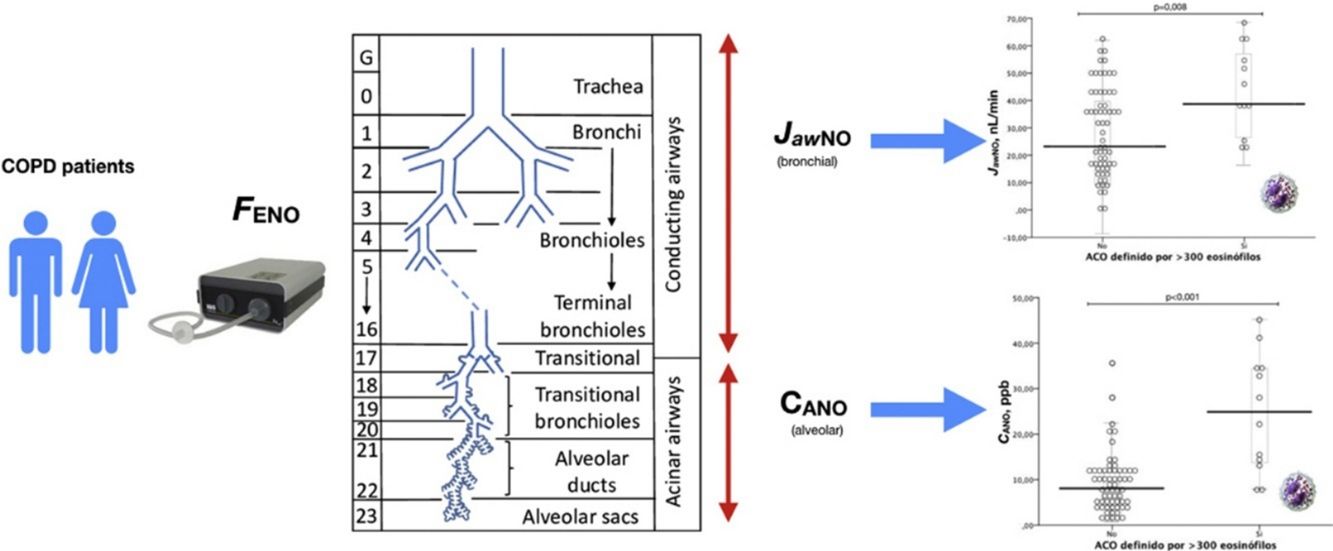
The measurement of nitric oxide in exhaled air as a marker of airway inflammation has advanced greatly in recent years, to the extent that several equations are now available that separate the alveolar or distal airway component (CANO) from the central bronchial component (JawNO).9–11 Both components have been widely studied in patients with bronchial asthma: the alveolar component (CANO) has been shown to be higher among patients with more severe asthma, suggesting greater inflammation in this region.12,13 Measuring CANO can also help identify patients who are likely to improve with the use of inhaled corticosteroids (ICS)14 and patients who present an increased risk of comorbidity.15
Different types of COPD patients can be identified by measuring nitric oxide in exhaled air (FENO).16,17 This variable is also associated with the presence of eosinophils in sputum,18,19 a typical finding in ACO. FENO levels are also a good predictor of response to the use of ICS.20,21 Studies differentiating the alveolar fraction from the bronchial fraction of FENO in COPD patients have shown that inflammation is distal in some cases,22 a phenomenon also observed in patients with severe asthma.
In a cross-sectional study conducted in Spain, elevated FENO levels were found in patients defined as having ACO, with an optimal cut-off of 20 parts per billion (ppb) for the diagnosis of ACO.16
However, the differences between the alveolar and bronchial components and the clinical and biological variables associated with inflammation in either of the territories have not been studied in depth.
The aim of this study is to determine differences in FENO levels between the alveolar and bronchial compartments in patients fulfilling biological criteria for ACO, and whether these differences are associated with other clinical or biological variables which might determine whether inflammation occurs more in a particular territory (CANO and JawNO).
Materials and MethodsStudy designThis was an observational, cross-sectional study to evaluate the differences in the production of CANO and JawNO and the relationship of these variables with clinical variables in a consecutive series of COPD patients who performed complete lung function testing (measurement of lung volumes and diffusion) in our respiratory outpatient clinic. The study was performed between November 2014 and May 2015.
Study populationThe study population comprised adult patients over 40 years of age, smokers, or non-smokers with an accumulated pack-year index of at least 10, and a diagnosis of COPD according to national and international guidelines and recommendations.1 Exclusion criteria were the presence of any respiratory disease other than COPD that might significantly affect the examination (including a history of bronchial asthma), a history of COPD exacerbation in the 4 weeks before the test, inability to perform the study procedures or complete the questionnaires, and participation in any other clinical trial or research study.
Study variablesClinical variables: for each patient, data were collected on their respiratory disease, including time since onset, toxic habits, comorbidities, baseline dyspnea measured according to the modified Medical Research Council (mMRC) scale, COPD Assessment Test (CAT®) questionnaire, and history of exacerbations in the previous year (classified as moderate if treated with systemic corticosteroids and/or antibiotics in an outpatient setting, and severe in the case of admission > 24h to a hospital or emergency department).
Blood tests: before lung function tests were performed, peripheral blood was obtained for the determination of absolute eosinophil counts and percentages, and for the measurement of C-reactive protein (CRP).
Measurement of the alveolar (CANO) and bronchial (JawNO) component of nitric oxide in exhaled air: before lung function tests were performed, patients performed 3 FENO maneuvers at 50mL/s (FENO50) followed by additional determinations at 100mL/s, 200mL/s, and 350mL/s (at least 2 each) in order to obtain CANO and JawNO levels, according to international guidelines.23,24 An NO chemiluminescence analyzer (HypAir FENO®, Medisoft, Belgium) was used.
Lung function variables: patients performed spirometry at baseline and after inhaling salbutamol 400μg in accordance with national and international guidelines.25,26 Lung volumes and diffusing capacity of the lung for carbon monoxide were also determined according to applicable recommendations.27,28 All measurements were performed on the same lung function testing equipment (MasterLab, Jaeger GmbH, Germany).
Ethical aspectsThe study was conducted in compliance with the principles of the Declaration of Helsinki. Patients who were invited to participate signed informed consent forms. The study was approved by the reference clinical research ethics committee. All patient records generated in the database were confidential and handled in compliance with the Personal Data Protection Law 15/1999 of 13 December 1999.
Statistical analysisA descriptive statistical analysis was made of the study variables, using absolute and relative frequencies in the case of qualitative variables. Depending on whether distribution was normal or non-normal (applying the appropriate normality test for the sample size), quantitative variables were summarized by mean, standard deviation (Md ± SD), and range (minimum and maximum) or P50 (P25 - P75] (median, interquartile range), respectively.
Quantitative variables were compared by study groups using the Student's t-test for independent samples or the Kruskal–Wallis H (the appropriate test was selected from previously obtained results). Post hoc tests were conducted in the case of ANOVA; otherwise, the Mann–Whitney U-test with the Bonferroni correction was used. To determine the relationship between CANO, JawNO, and the clinical variables, a multiple linear regression analysis was performed with CANO and JawNO as dependent variables, respectively, using the forward stepwise method. The level of statistical significance was set at a p-value of 0.05. The statistical analysis was performed using the Statistical Package for Social Sciences (SPSS Inc., Chicago, IL, USA), version 24.0.
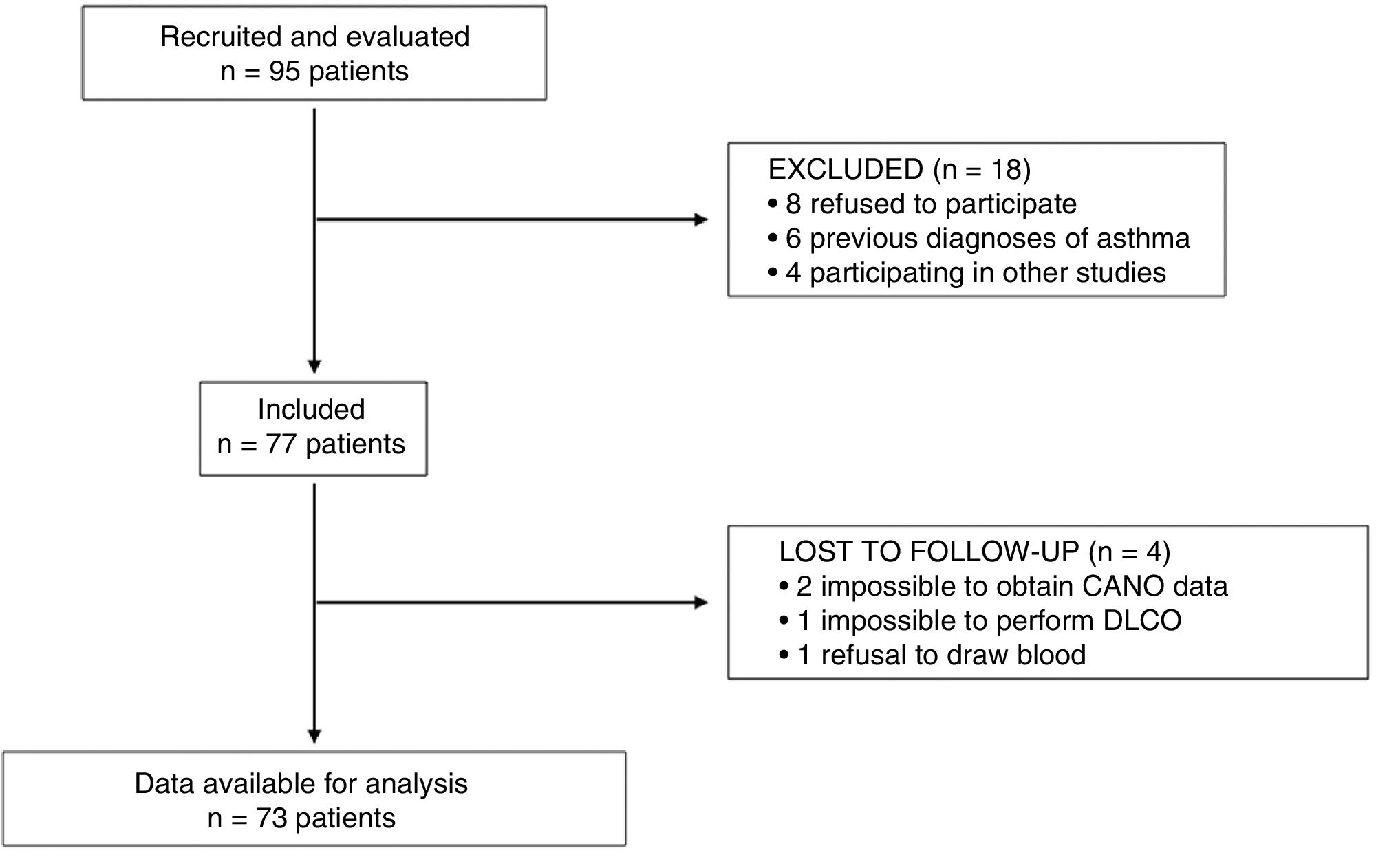
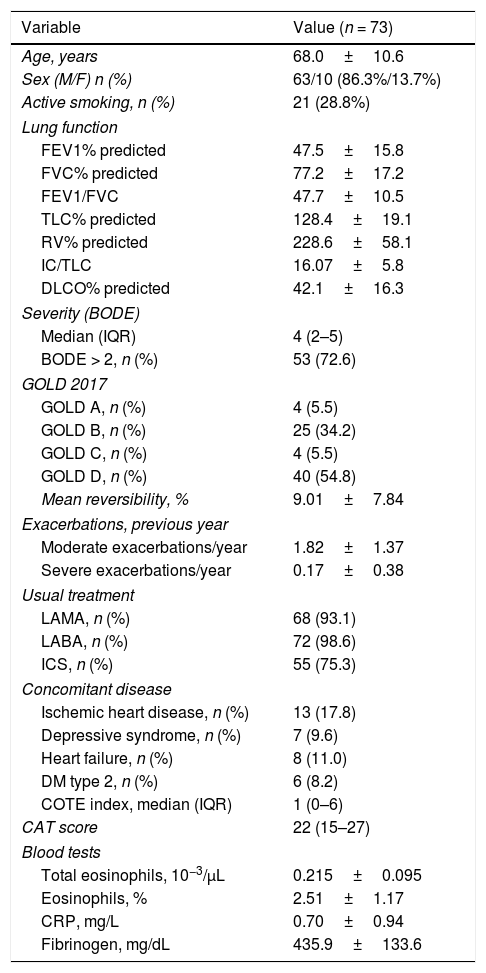
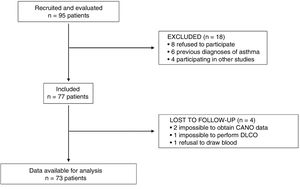
ResultsCharacteristics of the study populationNinety-five patients were invited to participate, 73 of whom finally completed all procedures. Fig. 1 is a STROBE diagram of the study participants.
Baseline patient characteristics are shown in Table 1. In summary, this is a series of patients with predominantly severe COPD, mean FEV1 < 50%, and a high burden of symptoms and exacerbations in the previous year. About 25% of the study population were active smokers.
Characteristics of study population.
| Variable | Value (n = 73) |
|---|---|
| Age, years | 68.0±10.6 |
| Sex (M/F) n (%) | 63/10 (86.3%/13.7%) |
| Active smoking, n (%) | 21 (28.8%) |
| Lung function | |
| FEV1% predicted | 47.5±15.8 |
| FVC% predicted | 77.2±17.2 |
| FEV1/FVC | 47.7±10.5 |
| TLC% predicted | 128.4±19.1 |
| RV% predicted | 228.6±58.1 |
| IC/TLC | 16.07±5.8 |
| DLCO% predicted | 42.1±16.3 |
| Severity (BODE) | |
| Median (IQR) | 4 (2–5) |
| BODE > 2, n (%) | 53 (72.6) |
| GOLD 2017 | |
| GOLD A, n (%) | 4 (5.5) |
| GOLD B, n (%) | 25 (34.2) |
| GOLD C, n (%) | 4 (5.5) |
| GOLD D, n (%) | 40 (54.8) |
| Mean reversibility, % | 9.01±7.84 |
| Exacerbations, previous year | |
| Moderate exacerbations/year | 1.82±1.37 |
| Severe exacerbations/year | 0.17±0.38 |
| Usual treatment | |
| LAMA, n (%) | 68 (93.1) |
| LABA, n (%) | 72 (98.6) |
| ICS, n (%) | 55 (75.3) |
| Concomitant disease | |
| Ischemic heart disease, n (%) | 13 (17.8) |
| Depressive syndrome, n (%) | 7 (9.6) |
| Heart failure, n (%) | 8 (11.0) |
| DM type 2, n (%) | 6 (8.2) |
| COTE index, median (IQR) | 1 (0–6) |
| CAT score | 22 (15–27) |
| Blood tests | |
| Total eosinophils, 10−3/μL | 0.215±0.095 |
| Eosinophils, % | 2.51±1.17 |
| CRP, mg/L | 0.70±0.94 |
| Fibrinogen, mg/dL | 435.9±133.6 |
BODE: body mass index, obstruction, dyspnea, and exercise tolerance; CAT: COPD assessment test; CRP: C-reactive protein; DLCO: diffusing capacity of the lung for carbon monoxide (expressed as a percentage of the predicted value); DM: diabetes mellitus; FEV1% predicted: peak expiratory flow in 1 second (expressed as a percentage of the predicted value); FVC: forced vital capacity (expressed as a percentage of the predicted value); IC: inspiratory capacity; ICS: inhaled corticosteroids; IQR: interquartile range; LABA: long-acting β adrenergic agonists; LAMA: long-acting muscarinic antagonists; RV: residual volume (expressed as a percentage of the predicted value); TLC: total lung capacity (expressed as a percentage of the predicted value).
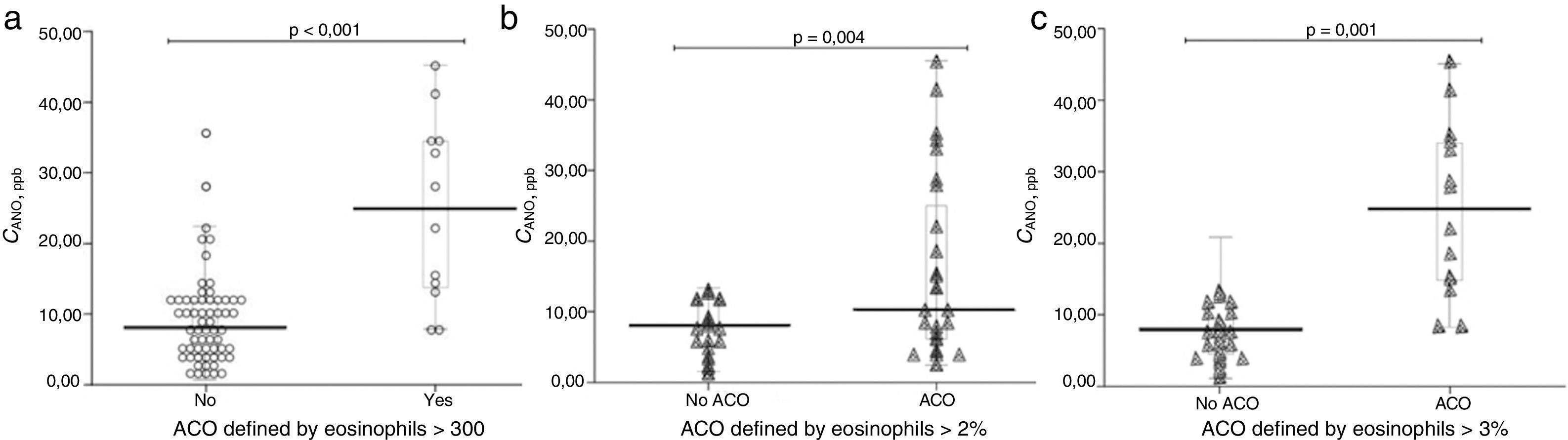
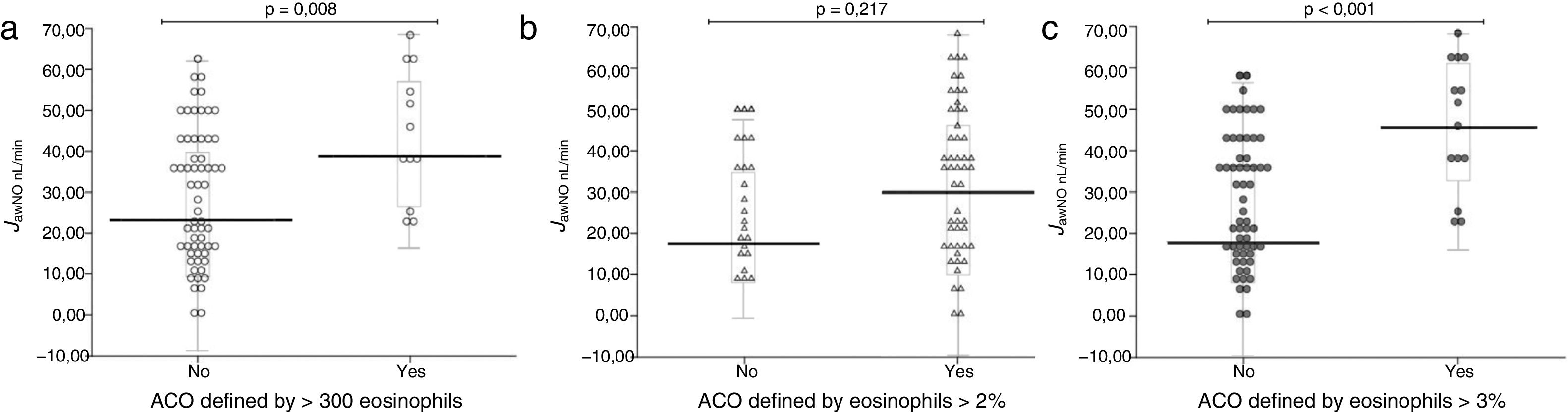
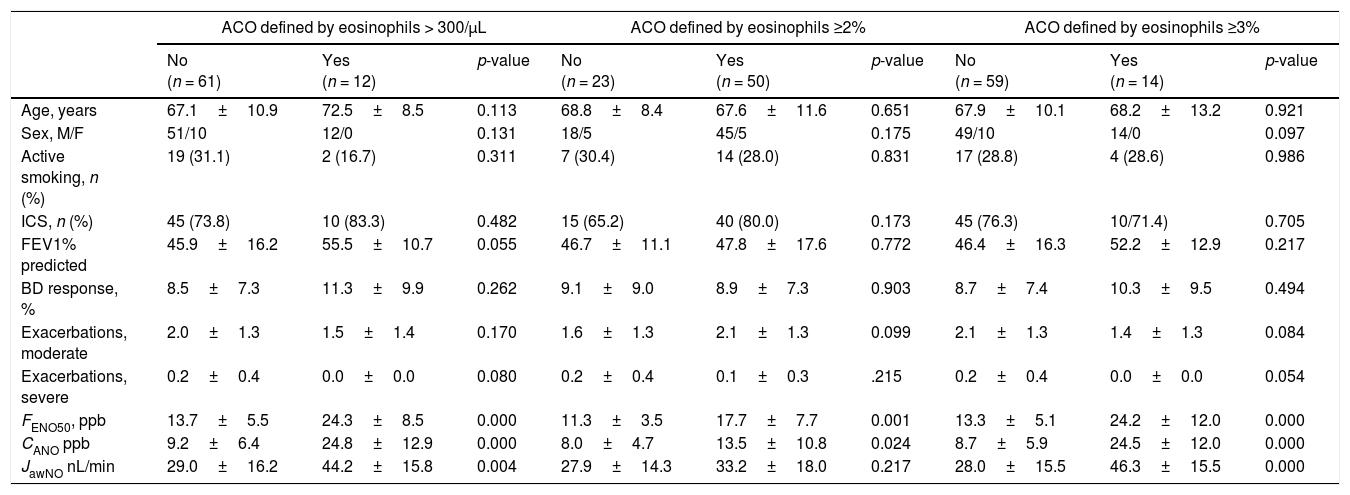
Table 2 shows patient characteristics according to the eosinophil count criteria for defining ACO (≥2% eosinophils, ≥3% eosinophils, or ≥300cells/μL). Clinical parameters did not differ among the groups according to the criterion for defining ACO, except for nitric oxide measurements (FENO50, CANO, and JawNO). Figs. 2 and 3 show CANO and JawNO levels, respectively, using different eosinophil cut-off points for the definition of ACO (2%, 3%, and > 300 eosinophils, respectively). In all cases, CANO levels were higher for patients defined as having ACO according to the eosinophil count, while differences were greater when the criteria of >3% and >300 eosinophils were used. JawNO levels were higher only in those patients with ACO defined by >3% or >300 eosinophils. The use of more restrictive criteria (>300 eosinophils and >3%) did not improve the detection of differences between patients classified as ACO and other patients.
Patient characteristics by ACO criterion.
| ACO defined by eosinophils > 300/μL | ACO defined by eosinophils ≥2% | ACO defined by eosinophils ≥3% | |||||||
|---|---|---|---|---|---|---|---|---|---|
| No (n = 61) | Yes (n = 12) | p-value | No (n = 23) | Yes (n = 50) | p-value | No (n = 59) | Yes (n = 14) | p-value | |
| Age, years | 67.1±10.9 | 72.5±8.5 | 0.113 | 68.8±8.4 | 67.6±11.6 | 0.651 | 67.9±10.1 | 68.2±13.2 | 0.921 |
| Sex, M/F | 51/10 | 12/0 | 0.131 | 18/5 | 45/5 | 0.175 | 49/10 | 14/0 | 0.097 |
| Active smoking, n (%) | 19 (31.1) | 2 (16.7) | 0.311 | 7 (30.4) | 14 (28.0) | 0.831 | 17 (28.8) | 4 (28.6) | 0.986 |
| ICS, n (%) | 45 (73.8) | 10 (83.3) | 0.482 | 15 (65.2) | 40 (80.0) | 0.173 | 45 (76.3) | 10/71.4) | 0.705 |
| FEV1% predicted | 45.9±16.2 | 55.5±10.7 | 0.055 | 46.7±11.1 | 47.8±17.6 | 0.772 | 46.4±16.3 | 52.2±12.9 | 0.217 |
| BD response, % | 8.5±7.3 | 11.3±9.9 | 0.262 | 9.1±9.0 | 8.9±7.3 | 0.903 | 8.7±7.4 | 10.3±9.5 | 0.494 |
| Exacerbations, moderate | 2.0±1.3 | 1.5±1.4 | 0.170 | 1.6±1.3 | 2.1±1.3 | 0.099 | 2.1±1.3 | 1.4±1.3 | 0.084 |
| Exacerbations, severe | 0.2±0.4 | 0.0±0.0 | 0.080 | 0.2±0.4 | 0.1±0.3 | .215 | 0.2±0.4 | 0.0±0.0 | 0.054 |
| FENO50, ppb | 13.7±5.5 | 24.3±8.5 | 0.000 | 11.3±3.5 | 17.7±7.7 | 0.001 | 13.3±5.1 | 24.2±12.0 | 0.000 |
| CANO ppb | 9.2±6.4 | 24.8±12.9 | 0.000 | 8.0±4.7 | 13.5±10.8 | 0.024 | 8.7±5.9 | 24.5±12.0 | 0.000 |
| JawNO nL/min | 29.0±16.2 | 44.2±15.8 | 0.004 | 27.9±14.3 | 33.2±18.0 | 0.217 | 28.0±15.5 | 46.3±15.5 | 0.000 |
BD: bronchodilator response; CANO: alveolar nitric oxide; FENO50: nitric oxide in exhaled air at 50mL/s; JawNO: bronchial nitric oxide.
CANO values by ACO criterion. (a) ACO defined by ≥300 eosinophils/μL. (b) ACO defined by ≥2% eosinophils. (c) ACO defined by ≥3% eosinophils. Horizontal lines show the mean value, figures show the individual values, the box shows the IQ range, and the whiskers show the mean standard error.
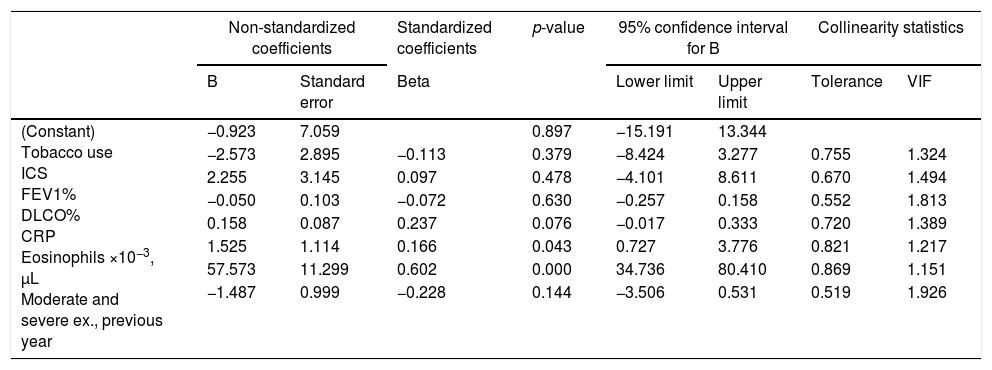
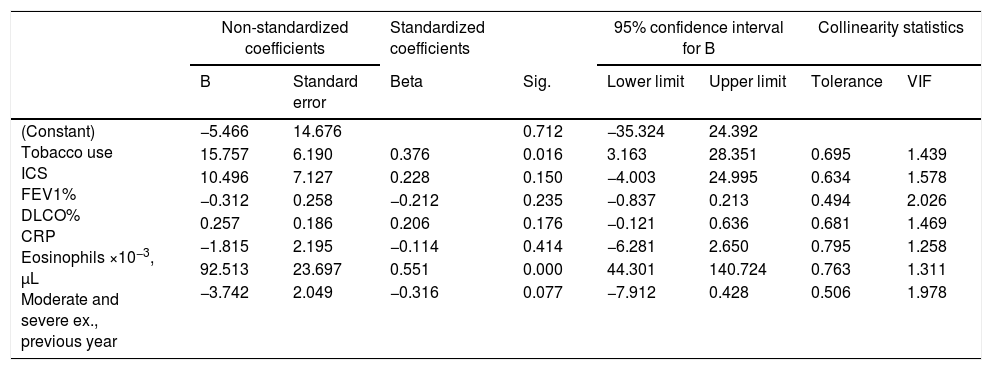
Multiple linear regression analyses aimed at evaluating the associations between CANO and JawNO and different clinical, biological, and lung function variables are shown in Tables 3 and 4, respectively. Variables associated with alveolar inflammation (CANO), and which predicted its finding, were CRP and total eosinophil count in peripheral blood, with an R2 of 0.412. Variables that showed the closest association with bronchial inflammation (JawNO), and which predicted its finding, were smoking, number of moderate exacerbations in the previous year, and eosinophil count in peripheral blood, with an R2 of 0.477. Appendix B (additional material) depicts CANO and JawNO values compared with the predicted values according to these models (expressed as regressions of the standardized predicted value).
Multiple linear regression analysis with CANO as the dependent variable.
| Non-standardized coefficients | Standardized coefficients | p-value | 95% confidence interval for B | Collinearity statistics | ||||
|---|---|---|---|---|---|---|---|---|
| B | Standard error | Beta | Lower limit | Upper limit | Tolerance | VIF | ||
| (Constant) Tobacco use ICS FEV1% DLCO% CRP Eosinophils ×10−3, μL Moderate and severe ex., previous year | −0.923 | 7.059 | 0.897 | −15.191 | 13.344 | |||
| −2.573 | 2.895 | −0.113 | 0.379 | −8.424 | 3.277 | 0.755 | 1.324 | |
| 2.255 | 3.145 | 0.097 | 0.478 | −4.101 | 8.611 | 0.670 | 1.494 | |
| −0.050 | 0.103 | −0.072 | 0.630 | −0.257 | 0.158 | 0.552 | 1.813 | |
| 0.158 | 0.087 | 0.237 | 0.076 | −0.017 | 0.333 | 0.720 | 1.389 | |
| 1.525 | 1.114 | 0.166 | 0.043 | 0.727 | 3.776 | 0.821 | 1.217 | |
| 57.573 | 11.299 | 0.602 | 0.000 | 34.736 | 80.410 | 0.869 | 1.151 | |
| −1.487 | 0.999 | −0.228 | 0.144 | −3.506 | 0.531 | 0.519 | 1.926 | |
Ex.: exacerbations; VIF: variance inflation factor.
Multiple linear regression analysis with JawNO as the dependent variable.
| Non-standardized coefficients | Standardized coefficients | 95% confidence interval for B | Collinearity statistics | |||||
|---|---|---|---|---|---|---|---|---|
| B | Standard error | Beta | Sig. | Lower limit | Upper limit | Tolerance | VIF | |
| (Constant) Tobacco use ICS FEV1% DLCO% CRP Eosinophils ×10−3, μL Moderate and severe ex., previous year | −5.466 | 14.676 | 0.712 | −35.324 | 24.392 | |||
| 15.757 | 6.190 | 0.376 | 0.016 | 3.163 | 28.351 | 0.695 | 1.439 | |
| 10.496 | 7.127 | 0.228 | 0.150 | −4.003 | 24.995 | 0.634 | 1.578 | |
| −0.312 | 0.258 | −0.212 | 0.235 | −0.837 | 0.213 | 0.494 | 2.026 | |
| 0.257 | 0.186 | 0.206 | 0.176 | −0.121 | 0.636 | 0.681 | 1.469 | |
| −1.815 | 2.195 | −0.114 | 0.414 | −6.281 | 2.650 | 0.795 | 1.258 | |
| 92.513 | 23.697 | 0.551 | 0.000 | 44.301 | 140.724 | 0.763 | 1.311 | |
| −3.742 | 2.049 | −0.316 | 0.077 | −7.912 | 0.428 | 0.506 | 1.978 | |
Ex.: exacerbations; VIF: variance inflation factor.
The results of this study show that COPD patients who meet one of the proposed ACO criteria (elevated eosinophil count in peripheral blood) showed differences in nitric oxide production in the alveolar territory (CANO) or in the bronchial territory (JawNO), depending on the threshold considered for eosinophil counts, while values were similar if the eosinophil criteria applied were ≥300cells/μL or ≥3%.
Our findings are in line with the previous study published by our group and show that FENO50 may be an accessible marker for the detection of ACO.16 Our results contribute additional, clinically useful data, and define in more detail the contribution of eosinophil levels in peripheral blood.
Measuring FENO and differentiating the alveolar and bronchial components is an approach that has been developed mainly in the field of bronchial asthma, in which it has previously shown utility in the evaluation of disease control and for predicting the future risk or adjustment of inhaled treatment.29–31
Fewer studies have been published on determinations of CANO and JawNO in COPD patients, and the results have been less convincing,32–34 although until now, no analyses have been based on the criteria for defining ACO, i.e., eosinophils in peripheral blood or bronchodilator response. Our findings in the group of patients without biological characteristics of ACO are comparable with series reported by other authors.32,33 Similarly, we found a relationship between smoking and low JawNO values, also noted in another study, suggesting that smoking influences the production of bronchial but not alveolar nitric oxide. In our study, the COPD patient group showed no differences with respect to alveolar and bronchial inflammation (CANO and JawNO) from the patient group with no signs of ACO.
Our study has limitations: it is cross-sectional, it was conducted in a single center, and few patients were included, due to the technical demands of the test procedure. However, the series size is similar to that of other studies.32 Moreover, in order to eliminate other possible confounding factors, we excluded patients previously diagnosed with bronchial asthma, so the results obtained are not applicable to all patients with ACO. We should also mention that not all patients are classified as ACO due to their eosinophil count. Another weakness of our study is that some individuals were active smokers and some were receiving treatment with ICS, which may alter CANO and JawNO levels.
From the perspective of clinical utility, the use of the variables included in the linear regression models of this study could help clinicians identify patients or patient groups who may have greater bronchial or alveolar Th2 inflammation,35 and who therefore require an inhalation system or molecule that deposits preferentially in the bronchi or the alveoli. This would individualize treatment and reduce the risk of side effects.36
In summary, we present evidence that FENO production in COPD patient differs depending on eosinophil levels in peripheral blood, particularly with regard to nitric oxide produced by peripheral airways and its associated clinical variables, factors that are easy for the clinician to identify.
Conflict of interestThe authors state that they have no conflict of interests.
Please cite this article as: Alcázar-Navarrete B, Castellano Miñán F, Santiago Díaz P, Ruiz Rodrígueza O, Romero Palacios PJ. Óxido nítrico alveolar y bronquial en la enfermedad pulmonar obstructiva crónica y el solapamiento de asma y EPOC (ACO). Arch Bronconeumol. 2018;54:414–419.