Silicosis is associated with an increased risk of developing systemic autoimmune rheumatic disease (SARD). The prognostic implications of this association are poorly characterized. The aim of this study was to determine the prevalence of SARD and autoimmune markers in a cohort of patients with exposure to silica and assess their impact on prognosis.
MethodWe performed a prospective observational study of all patients attending the dedicated silicosis clinic of our pulmonology unit between 2009 and December 2017. Diagnosis was confirmed by a rheumatologist according to Spanish Rheumatology Society criteria. Autoimmune markers, pulmonary function tests, radiological progression, visits to the emergency department and primary care center, and hospital admissions for respiratory causes, and mortality were analyzed.
ResultsOverall, 489 cases of silicosis and 95 cases of exposure were studied. In total, 54 (11.0%) patients with silicosis had SARD: 12 (2.4%) rheumatoid arthritis, 10 (2.0%) systemic lupus erythematosus, 10 (2.0%) systemic sclerosis, 3 (0.6%) Sjögren syndrome, 2 (0.4%) vasculitis associated with anti-neutrophil cytoplasmic antibodies (ANCA +), 6 (1.2%) psoriatic arthritis, 3 (0.6%) ankylosing spondylitis, and 8 (1.6%) other autoimmune diseases with no special features. The patients with SARD visited the emergency room more often (63.0% vs. 42.5%; p = 0.004), and progressed more rapidly (22.2 vs. 11.7%; p = 0.030).
ConclusionsThe presence of systemic rheumatic autoimmune diseases involves radiological progression and a higher clinical impact.
La silicosis se asocia a un aumento del riesgo de padecer una de las enfermedades reumatológicas autoinmunes sistémicas (ERAS), aunque no se conocen las implicaciones clínicas de esta asociación. El objetivo del estudio es determinar la prevalencia de ERAS y de marcadores de autoinmunidad en una cohorte de pacientes con exposición a inhalación de polvo de sílice y evaluar su impacto clínico.
MétodoEstudio observacional prospectivo en pacientes atendidos en una consulta monográfica de silicosis desde 2009 hasta diciembre 2017. El diagnóstico de ERAS se confirmó por un especialista en Reumatología según criterios de la Sociedad Española de Reumatología. Se analizaron marcadores de autoinmunidad, pruebas de función respiaratoria, progresión radiológica e impacto clínico medido por visitas a Atención Primaria, a Servicio de Urgencias, ingresos hospitalarios por causa respiratoria y mortalidad.
ResultadosSe estudiaron 489 casos de silicosis y 95 de exposición a inhalación de polvo de sílice sin silicosis. De los pacientes con silicosis, 54 (11,0%) tenían ERAS: 12 (2,4%) artritis reumatoide, 10 (2,0%) lupus eritematoso sistémico, 10 (2,0%) esclerosis sistémica, 6 (1,2%) artritis psoriásica, 3 (0,6%) Síndrome de Sjögren, 2 (0,4%) vasculitis asociada a anticuerpos anticitoplasma de neutrófilos, 3 (0,6%) espondiloartritis y 8 (1,6%) enfermedad autoinmune sin características específicas. Los pacientes con ERAS realizaron más visitas a urgencias (63,0% vs. 42,5%; p = 0,004), y experimentaron mayor progresión (22,2 vs. 11,7%; p = 0,030).
ConclusionesLos pacientes con silicosis presentan una prevalencia de ERAS elevada y su presencia se asocia a una mayor progresión radiológica y un mayor impacto clínico.
Silicosis is an irreversible, currently incurable, chronic interstitial lung disease caused by chronic inhalation of silica powder in crystalline form. It is one of the most common occupational respiratory diseases in our setting1,2. The annual reports of the National Institute of Silicosis (INS) state that since 2008, there has been an increase in new diagnoses from sectors other than coal mining, including the granite, slate and artificial silica conglomerates industries3,4.
Inhalation of silica dust causes respiratory conditions, such as silicosis, bronchial obstruction, mycobacterial infections, and lung cancer, and has also been associated with other diseases such as systemic autoimmune rheumatic disease (SARD)1,5. However, the mechanisms that link silica exposure with these autoimmune diseases are unknown.
Several SARDs have been associated with exposure to silica dust inhalation, including systemic sclerosis (SS)6,7, rheumatoid arthritis (RA)8,9, systemic lupus erythematosus (SLE), Sjögren’s syndrome (SjS), and anti-neutrophil cytoplasmic antibody-associated vasculitis (AAV)9. Studies, however, were carried out in heterogeneous populations, with few patients and only partial determinations of some antibodies or use of non-standard detection techniques10. Although the presence of RA has been described as a risk factor for progression to progressive massive fibrosis (PMF)11, the clinical impact in terms of functional status, symptoms, radiological progression, and events such as consultations, admissions and mortality in patients with silicosis, with or without SARD, has not been extensively analyzed in the literature.
The objectives of our study were to determine the prevalence of SARD and a series of autoimmune markers in a population of patients exposed to silica dust inhalation, with and without silicosis, and to analyze clinical outcomes, radiological progression, and mortality.
MethodThis was a prospective observational study conducted in a cohort of patients treated in a silicosis clinic between 2009 and 2017. Initially, all patients seen during the study period who had been exposed to silica dust inhalation, with or without a diagnosis of silicosis, and who signed informed consent were included. Patients with a differential diagnosis suggestive of other diseases were excluded (10 with sarcoidosis and 4 with idiopathic pulmonary fibrosis). The study was approved by the Network of Research Ethics Committees of Galicia and was conducted in accordance with the principles of the Declaration of Helsinki and its subsequent updates, and applicable biomedical research legislation.
At the initial visit, we collected sociodemographic variables, history of smoking (never smoker, former smoker or active smoker), cumulative pack-year index, comorbidity according to the Charlson index12, history of other specific diseases (pulmonary tuberculosis, non-tuberculous mycobacterial disease, pulmonary hypertension), history of exposure to silica dust inhalation, date of diagnosis of silicosis and severity, lung function, chest X-ray (Rx) and/or high-resolution computed tomography (CT) findings, blood tests, and body mass index.
Silicosis was diagnosed according to the SEPAR guideline13. Simple chronic silicosis was defined as opacities measuring <10 mm and complicated chronic silicosis as opacities >10 mm. A rheumatologist evaluated a possible diagnosis of SARD, establishing a definitive diagnosis according to the criteria of the Spanish Society of Rheumatology14. Antinuclear antibodies (ANA), antibody to extractable nuclear antigen (anti-ENA), anti-citrullinated cyclic peptide antibodies (anti-CCP), anti-neutrophil cytoplasmic antibodies (ANCA), and rheumatoid factor (RF) were evaluated as autoimmune markers. Radiological progression of silicosis, hospital admissions, emergency department or primary care consultations for respiratory causes, and death were assessed at follow-up visits until December 2018. The medical records were reviewed before the database was closed to complete any missing data.
Chest X-ray and CT were interpreted by radiologists with extensive experience in reading silicosis. The radiological evaluation was performed according to the ILO classification15. Small opacities on CT were classified in a manner similar to that used to classify radiological findings of the chest16. Large opacities were classified as: category A, in the presence of 1 or more opacities measuring more than 10 mm and less than 1 quadrant in the right side of the CT, at the level of the carina; category B, when opacity was greater than A but less than 2 quadrants in the right side of the CT, at the level of the carina; and category C in the presence of 1 or more opacities that together constituted more than 2 quadrants in the right side of the CT at the level of the carina17.
Any increase in nodule profusion, regardless of the amount, was interpreted as progression on Rx or CT. Progression involving a category change was defined as a change from simple to complicated silicosis, from complicated silicosis A to complicated silicosis B or C, or from B to C, confirmed on CT.
Lung function, spirometry, and diffusion capacity tests for carbon monoxide (DLCO) were conducted following the recommendations of the Spanish Society of Pulmonology and Thoracic Surgery (SEPAR) and the European Respiratory Society (ERS)18,19. The results analyzed were forced vital capacity (FVC), forced expiratory volume in 1 s (FEV1), FEV1/FVC ratio, total lung capacity (TLC), and DLCO. Obstructive airflow limitation was defined as a ratio of FEV1/FVC <0.7 and decreases in FVC of >10% were analyzed: this value was taken as a clinically significant minimum difference20. Follow-up examinations of respiratory and radiological function were performed according to clinical judgment. The interval between lung function tests (first and last) was 53.9 ± 31.1 months. The intervals between chest X-rays were 76.1 ± 41.8 months and 54.5 ± 30.0 months between CT scans.
Descriptive and inferential analyses of the data were conducted. Results of the qualitative variables are presented as frequencies and percentages, and quantitative variables as mean ± standard deviations. We compared baseline characteristics of patients and lung function tests among patients, with and without SARD. A χ2 test was used to compare qualitative variables and the Student’s t test was used for quantitative variables. The same Kaplan-Meier survival tests and analyses were used to analyze the association with prognostic and progression variables. In the tests performed, the threshold of statistical significance was p < 0.05.
ResultsFrom January 2009 to December 2017, a total of 584 patients, all men, with a mean age of 52.8 ± 11.4 years, were evaluated. Overall, 489 had silicosis and 95 had been exposed but did not develop silicosis. Of the silicosis group, 203 had simple silicosis and 286 had complicated silicosis (165, A; 72, B; and 49, C). In terms of occupational history, 369 (75.5%) had worked in granite quarries, 44 (9.0%) in ornamental stone processing workshops, 11 (2.3%) were stone house builders, 15 (3.1%) worked in coal mines, 9 (1.8%) had been exposed to artificial quartz conglomerates, and 41 (8.4%) did other jobs, including working with ceramics, sand and gravel, and dental prosthetics. The mean duration of exposure to inhalation of silica dust was 23.9 ± 9.9 years and follow-up was 65.8 ± 32.7 months, during which 82 (16.8%) patients died.
Of the 489 patients with silicosis, 54 (11.0%) had SARD: 12 (2.4%) RA, 10 (2.0%) SLE, 10 (2.0%) SS, 3 (0.6%) SjS, 2 (0.4%) AAV, 6 (1.2%) psoriatic arthritis, three (0.6%) ankylosing spondylitis, and eight (1.6%) SARD without specific characteristics.
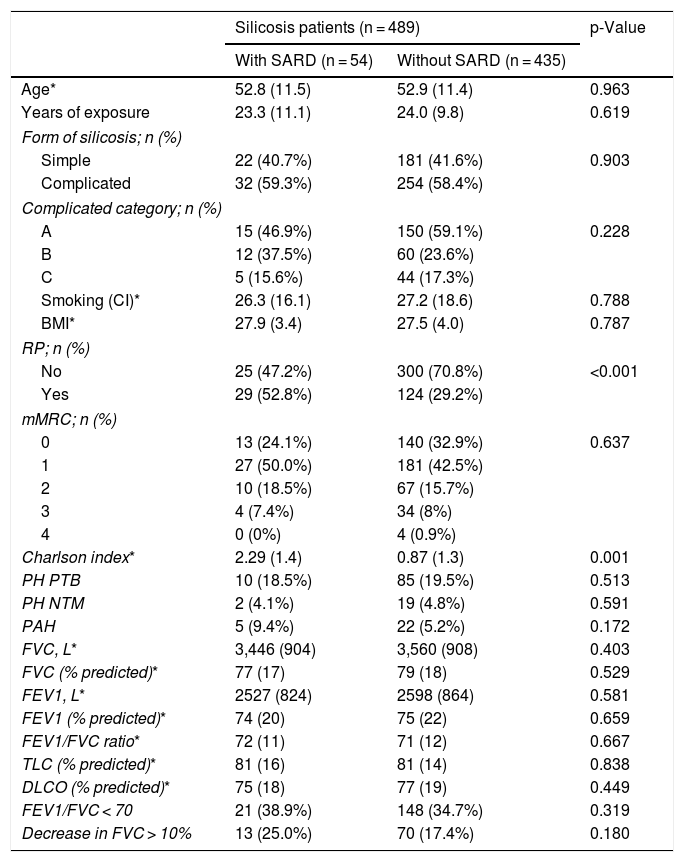
Demographic, clinical and functional characteristics are shown in Table 1. When comparing patients with SARD and without SARD, there were no significant differences in patient characteristics, except for a higher frequency of Raynaud’s phenomenon in patients with SARD [29 of 54 (52.8%)] than in patients without SARD [124 of 435 (29.2%)] (p < 0.001) and a higher number of comorbidities [Charlson index 2.3 ± 1.4 in patients with SARD versus 0.9 ± 1.3 in patients without SARD (p = 0.001)].
Characteristics of patients with silicosis with and without SARD.
| Silicosis patients (n = 489) | p-Value | ||
|---|---|---|---|
| With SARD (n = 54) | Without SARD (n = 435) | ||
| Age* | 52.8 (11.5) | 52.9 (11.4) | 0.963 |
| Years of exposure | 23.3 (11.1) | 24.0 (9.8) | 0.619 |
| Form of silicosis; n (%) | |||
| Simple | 22 (40.7%) | 181 (41.6%) | 0.903 |
| Complicated | 32 (59.3%) | 254 (58.4%) | |
| Complicated category; n (%) | |||
| A | 15 (46.9%) | 150 (59.1%) | 0.228 |
| B | 12 (37.5%) | 60 (23.6%) | |
| C | 5 (15.6%) | 44 (17.3%) | |
| Smoking (CI)* | 26.3 (16.1) | 27.2 (18.6) | 0.788 |
| BMI* | 27.9 (3.4) | 27.5 (4.0) | 0.787 |
| RP; n (%) | |||
| No | 25 (47.2%) | 300 (70.8%) | <0.001 |
| Yes | 29 (52.8%) | 124 (29.2%) | |
| mMRC; n (%) | |||
| 0 | 13 (24.1%) | 140 (32.9%) | 0.637 |
| 1 | 27 (50.0%) | 181 (42.5%) | |
| 2 | 10 (18.5%) | 67 (15.7%) | |
| 3 | 4 (7.4%) | 34 (8%) | |
| 4 | 0 (0%) | 4 (0.9%) | |
| Charlson index* | 2.29 (1.4) | 0.87 (1.3) | 0.001 |
| PH PTB | 10 (18.5%) | 85 (19.5%) | 0.513 |
| PH NTM | 2 (4.1%) | 19 (4.8%) | 0.591 |
| PAH | 5 (9.4%) | 22 (5.2%) | 0.172 |
| FVC, L* | 3,446 (904) | 3,560 (908) | 0.403 |
| FVC (% predicted)* | 77 (17) | 79 (18) | 0.529 |
| FEV1, L* | 2527 (824) | 2598 (864) | 0.581 |
| FEV1 (% predicted)* | 74 (20) | 75 (22) | 0.659 |
| FEV1/FVC ratio* | 72 (11) | 71 (12) | 0.667 |
| TLC (% predicted)* | 81 (16) | 81 (14) | 0.838 |
| DLCO (% predicted)* | 75 (18) | 77 (19) | 0.449 |
| FEV1/FVC < 70 | 21 (38.9%) | 148 (34.7%) | 0.319 |
| Decrease in FVC > 10% | 13 (25.0%) | 70 (17.4%) | 0.180 |
BMI: body mass index; CI: cumulative index; DLCO: lung diffusion capacity; FEV1: forced expiratory volume in 1 min; FVC: forced vital capacity; mMRC: modified Medical Research Council dyspnea index; NTM: non-tuberculous mycobacteria; PAH: pulmonary arterial hypertension; PH: personal history; PTB: pulmonary tuberculosis; RP: Raynaud’s phenomenon; TLC: total lung capacity.
A decrease in FVC > 10% was observed in 25.0% (13) of patients with SARD, and in 17.4% without SARD (p = 0.180).
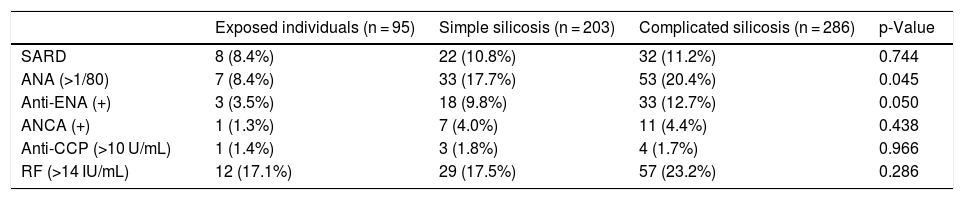
Table 2 shows the presence of autoantibodies (ANA, anti-ENA, anti-CCP, ANCA, and RF) in exposed individuals without silicosis and in patients with silicosis. Higher levels of ANA were observed in silicosis than in exposed individuals, with no differences in RF, ANCA, and anti-CCP.
Autoantibodies in exposed individuals without silicosis and in patients with silicosis.
| Exposed individuals (n = 95) | Simple silicosis (n = 203) | Complicated silicosis (n = 286) | p-Value | |
|---|---|---|---|---|
| SARD | 8 (8.4%) | 22 (10.8%) | 32 (11.2%) | 0.744 |
| ANA (>1/80) | 7 (8.4%) | 33 (17.7%) | 53 (20.4%) | 0.045 |
| Anti-ENA (+) | 3 (3.5%) | 18 (9.8%) | 33 (12.7%) | 0.050 |
| ANCA (+) | 1 (1.3%) | 7 (4.0%) | 11 (4.4%) | 0.438 |
| Anti-CCP (>10 U/mL) | 1 (1.4%) | 3 (1.8%) | 4 (1.7%) | 0.966 |
| RF (>14 IU/mL) | 12 (17.1%) | 29 (17.5%) | 57 (23.2%) | 0.286 |
ANA: antinuclear antibodies; ANCA: anti-neutrophil cytoplasmic antibodies; Anti-CCP: anti-neutrophil cytoplasmic antibodies; Anti-ENA: antibody to extractable nuclear antigen; RF: rheumatoid factor; SARD: systemic autoimmune rheumatologic disease.
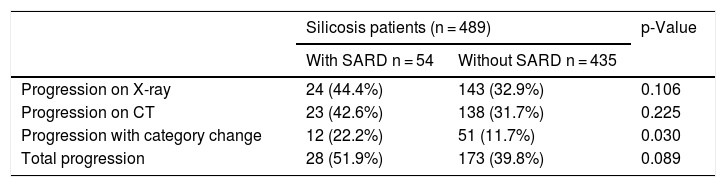
Radiological progression was observed in 26 (12.8%) patients with simple silicosis and in 175 (61.2%) patients with complicated silicosis. A statistically significant proportion of patients with SARD had progression with a change in silicosis category (Table 3).
Radiological progression by presence of SARD.
| Silicosis patients (n = 489) | p-Value | ||
|---|---|---|---|
| With SARD n = 54 | Without SARD n = 435 | ||
| Progression on X-ray | 24 (44.4%) | 143 (32.9%) | 0.106 |
| Progression on CT | 23 (42.6%) | 138 (31.7%) | 0.225 |
| Progression with category change | 12 (22.2%) | 51 (11.7%) | 0.030 |
| Total progression | 28 (51.9%) | 173 (39.8%) | 0.089 |
CT: Computed tomography.
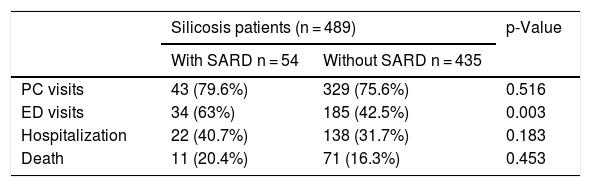
Patients with SARD made a greater number of visits to the emergency department (63.0 vs 42.5%, p = 0.004) than those without SARD, but the number of visits to primary care, number of hospital admissions and mortality rate did not differ among groups (Table 4). There were also no significant differences in survival between patients with and without SARD (log rank = 0.06; p = 0.802).
Visits to emergency department, primary care, hospitalization, and mortality.
| Silicosis patients (n = 489) | p-Value | ||
|---|---|---|---|
| With SARD n = 54 | Without SARD n = 435 | ||
| PC visits | 43 (79.6%) | 329 (75.6%) | 0.516 |
| ED visits | 34 (63%) | 185 (42.5%) | 0.003 |
| Hospitalization | 22 (40.7%) | 138 (31.7%) | 0.183 |
| Death | 11 (20.4%) | 71 (16.3%) | 0.453 |
ED: emergency department; PC, primary care.
This study has determined the prevalence of SARD for the first time in a Spanish cohort of confirmed cases of silicosis (11%) and in cases with significant exposure to silica dust inhalation without silicosis (8.4%). In addition, the presence of SARD in patients with silicosis has been associated with a higher number of emergency department visits and a greater silicosis category progression.
Compared with the EPISER study (Epidemiological study of the prevalence of rheumatic diseases in the adult population in Spain), we observed a higher prevalence of RA, SLE, SjS, and psoriatic arthritis among the various rheumatic diseases21. Although our sample comprises only men and is older than the EPISER study cohort21, our results reveal a possible relationship between SARD and exposure to inhalation of silica dust.
A study of SS in a Spanish population22 showed a prevalence of 0.03% compared to 2.0% in our study. With regard to vasculitis, the rate of AAV was 0.4%, while in the general population it was 0.003%23.
The prevalence of SARD varies considerably when compared with other studies in patients with silicosis. Makol et al.24 determined the prevalence of different SARDs in a cohort of 790 silicosis patients, mainly foundry workers, and detected a prevalence of RA of 4.2%, greater than the 2.4% reported in our population of silicosis patients, while their SS prevalence was lower than in our series (0.3% vs 2.0%). The prevalence of SjS associated with silicosis in our series was 0.6%, similar to a another cohort, although there are fewer reports of this association in the literature25. We observed SLE rates of 2.0%, higher than those reported by Makol et al.24 and similar to those described by Parks et al.9.
Despite the heterogeneity among the studies, a meta-analysis conducted in 2013 of 6 case-control studies underlined the association of exposure to silica dust inhalation and AAV26. Bartůnková et al.27 published a series of 86 men with a history of least 5 years’ exposure to silica. They detected ANCA more frequently in the group of exposed individuals (7.1%) and in the silicosis group (30.3%) than in the control group, but they only described the concentration of the marker and not the presence of vasculitis. Our cohort included 2 cases (0.4%) of AAV, a rate that is higher than the 0.003% estimated by Koldingsnes et al.23 in a population in northern Europe where this disease is more common, but similar to that reported by Makol et al.24.
In a series of patients with advanced silicosis associated with exposure to artificial quartz agglomerates, 9 of the 40 patients (23%) had autoimmune rheumatic diseases28. In our series, 9 cases of silicosis were reported in workers exposed to artificial quartz conglomerates, 2 of whom had SARD (22%).
A group of 8 patients (1.6%) had autoimmune rheumatic disease without specific characteristics, a finding that is usually not reported but is consistent with the pathogenesis of silicosis as an uncontrolled immune process29. It may be that this is an undifferentiated autoimmune disease associated with silica.
In silicosis, autoantibodies such as ANA, anti-Scl-70, anti-centromere, and ANCA are common, even if they are not associated with symptoms of autoimmune disease30. In our silicosis series, the prevalence of ANA was 17%, lower than that reported by Lippmann et al.31 of 34%, but higher than Zaghi et al.10.
Some hypotheses have been proposed regarding the pathophysiology of the mechanisms underlying the relationship of silica exposure with these autoimmune diseases30. One argues that inhalation of silica particles would trigger the activation of macrophages and their posterior apoptosis, which would lead to T and B lymphocyte activation, inducing an autoimmune response32. Another hypothesis contends that lymphocyte apoptosis, which is more dominant in regulatory T lymphocytes than in responsive T lymphocytes, would lead to an autoimmune imbalance32,33.
Nearly half of our silicosis patients progressed during the follow-up period. Similar findings were reported in the few published studies on the natural history and progression of silicosis, showing that between a third and a half of patients suffer progression of silicosis despite curtailing exposure34. For example, in the study by Lee et al.35 in 260 individuals with granite silicosis, 141 of whom had comparative X-rays, 37% showed progression, while Karataş et al.36 in a study of ceramic workers, 165 of whom were followed up for least 24 months and presented at least two X-rays, 38% showed progression. Very few studies have evaluated the role of SARD in the progression of silicosis. In 1986, Sluis-Cremer et al.11 published a case-control study that found that miners with rheumatoid arthritis showed faster silicosis progression and a tendency toward an increased risk of progressive massive fibrosis. Patients with SARD (not just RA) in our study showed a higher frequency of silicosis progression than patients without SARD.
No data on hospital stay and mortality due to silicosis are available in Spain, although a study carried out in our hospital between 2009 and 2016 found that 30% of patients with complicated silicosis were admitted at least once and had a mortality rate of 18% during the follow-up period37,38. In that paper, we reported that individuals with SARD have higher admission and mortality rates, although the difference was not statistically significant.
To date, no evidence has been generated on the deterioration of lung function in relation to the presence of SARD in patients with silicosis39. In this study, we have seen differences in lung function, although they have not been statistically significant.
Our study has some limitations. Although the design was prospective, no predefined intervals were specified for radiologic or lung function studies, so the evaluation of progression between tests may be influenced by differences in time periods. Radiological progression was estimated on the assumption that if the patients did not have 2 chest X-rays and/or 2 CTs (3 did not have 2 X-rays and 12 did not have 2 CTs), they had not progressed, which may underestimate the actual percentage of progression. Moreover, our data were collected from a single geographical area, with specific occupational exposures, working materials and climatic characteristics, so the external validity of our results must be interpreted with caution.
In conclusion, our study suggests that exposure to inhalation of silica dust and especially silicosis are associated with the development of SARD. The presence of SARD in patients with silicosis is associated with radiological progression and more visits to the emergency department.
Maintaining respirable dust levels within legal limits and early diagnosis are key to preventing silicosis. Clinicians need to bear in mind that silicosis patients can present different SARDs, and this must be evaluated in each visit. Likewise, patients with autoimmune rheumatic disease should be evaluated for occupational exposure to inhalation of silica dust40.
FundingThis study was funded by the Spanish Society of Pulmonology and Thoracic Surgery, under the auspices of SEPAR Research Grants Project No. 779, 2018.
Competing interestsThe authors declare no competing interests.
We thank all the professionals of the Respiratory Medicine department.
Please cite this article as: Blanco-Pérez JJ, Arnalich-Montiel V, Salgado-Barreira Á, Alvarez-Moure MA, Caldera-Díaz AC, Melero-Gonzalez R, et al. Prevalencia e impacto clínico de las enfermedades reumatológicas autoinmunitarias sistémicas en pacientes con silicosis. Arch Bronconeumol. 2021;57:571–576.