Journal Information
Vol. 60. Issue 5.
Pages 261-262 (May 2024)
Vol. 60. Issue 5.
Pages 261-262 (May 2024)
Editorial
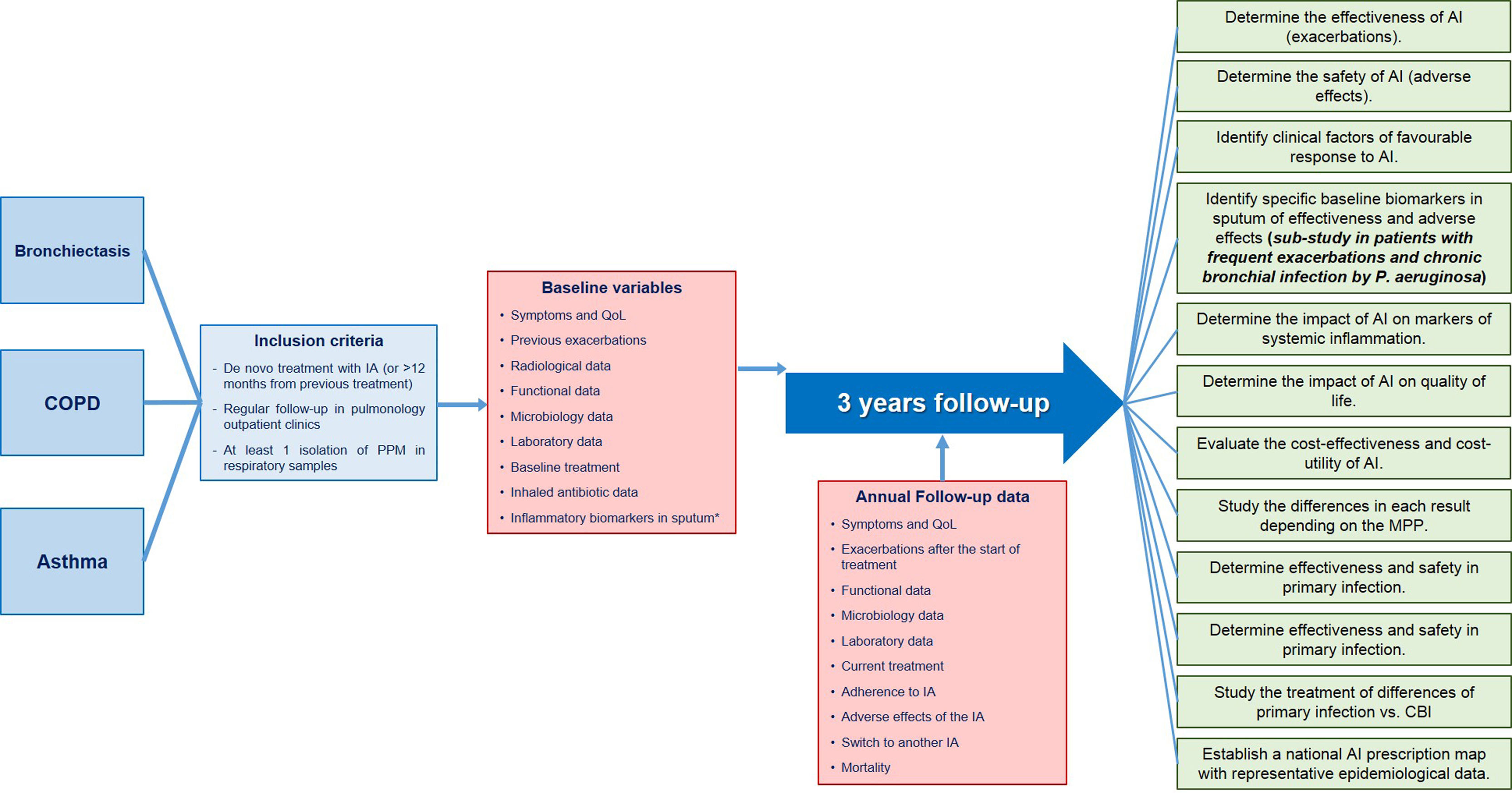
Unraveling the Enigma: Spain Takes a Pioneering Step With REPAIR in the World of Inhaled Antibiotics
Visits
813
David de la Rosa-Carrilloa,
, Marc Miravitllesb, Xavier Pomaresc, Miguel-Ángel Martínez-Garcíad,e
Corresponding author
a Pneumology Department, Hospital de la Santa Creu i Sant Pau, Barcelona, Spain
b Pneumology Department Hospital Universitari Vall d’Hebron, Vall d’Hebron Institut de Recerca (VHIR), Vall d’Hebron Hospital Campus, Barcelona, Spain
c Department of Respiratory Medicine, Hospital de Sabadell, Hospital Universitari Parc Taulí, Institut d’Investigació i Innovació Parc Taulí I3PT, Universitat Autònoma de Barcelona, Sabadell, Spain
d Pneumology Department, Hospital Universitario y Politécnico La Fe, Valencia, Spain
e CIBERES de Enfermedades Respiratorias, ISCIII, Madrid, Spain
This item has received
Article information
These are the options to access the full texts of the publication Archivos de Bronconeumología