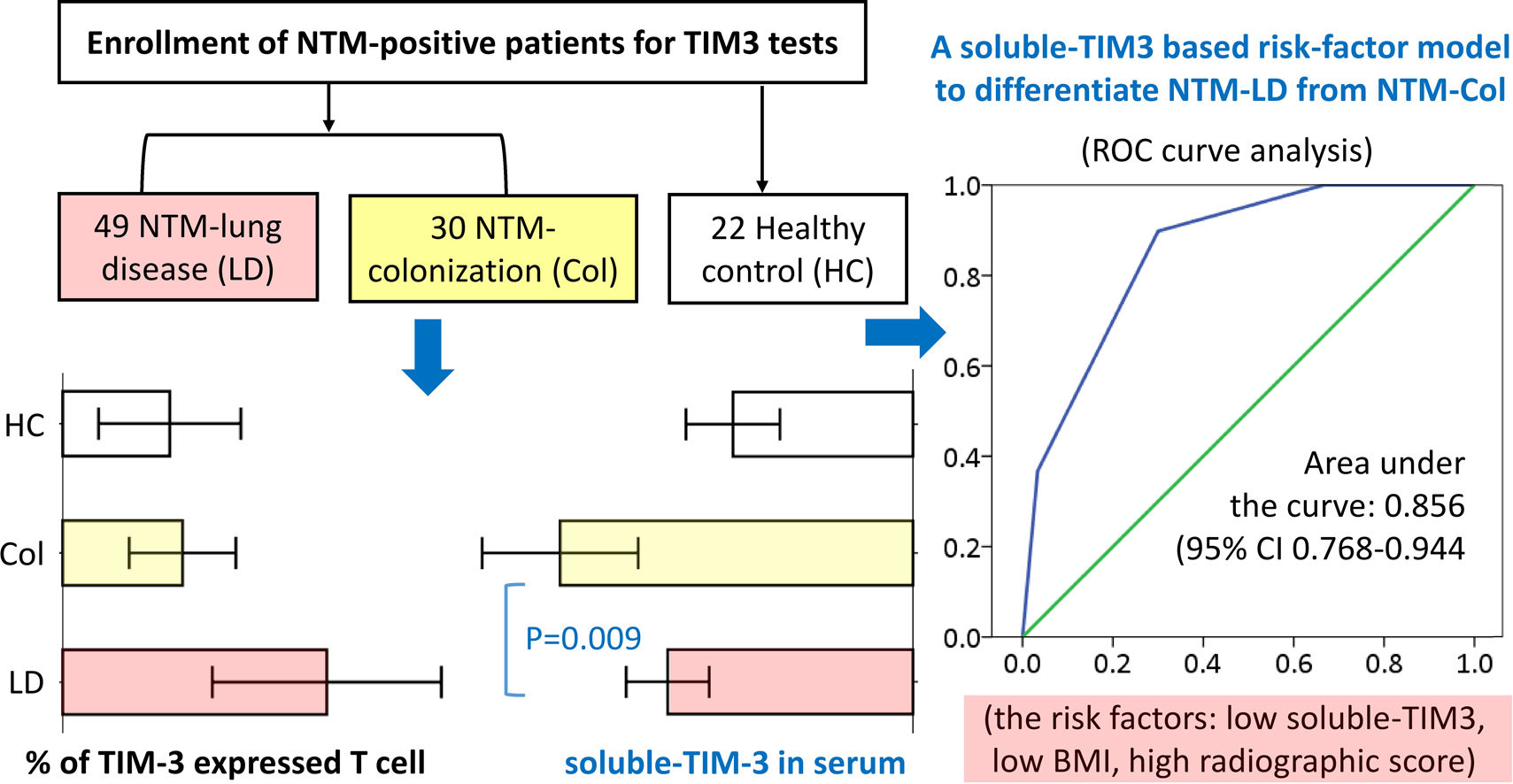
Differentiating between nontuberculous mycobacterial lung disease (NTM-LD) and pulmonary NTM colonization (NTM-Col) is difficult. Compared with healthy controls, patients with NTM-LD generally present immune tolerance along with increased expressions of T-cell immunoglobulin mucin domain-3 (TIM-3) and programmed cell death-1 (PD-1) on T lymphocytes. However, the role of soluble TIM-3 (sTIM-3) and soluble PD-1 (sPD-1) in differentiating NTM-LD from NTM colonization (NTM-Col) remains unclear.
MethodsPatients with NTM-positive respiratory samples and controls were enrolled from 2016 to 2019. Patients were classified into NTM-Col and NTM-LD groups. Levels of sTIM-3, sPD-1, soluble PD-ligand-1 (sPD-L1), and TIM-3 expression were measured. Factors associated with NTM-LD were analyzed by logistical regression.
ResultsTIM-3 expression on CD4+ and CD8+ T lymphocytes were highest in NTM-LD group, followed by NTM-Col, and control (P=.017 and P=.011 for trend). sTIM-3 elevated in the NTM-Col group compared with the NTM-LD and control groups (856.3±518.7 vs. 595.3±352.6pg/mL, P=.009; vs. 437.0±267.4pg/mL, P<.001). Levels of soluble PD-1 and its ligand were similar among groups. Among the 79 NTM-positive patients, sTIM-3 was associated with NTM-LD (100-pg/mL increase, adjusted odds ratio (aOR) 0.658 [95% CI, 0.502–0.864], P=.003). Patients with ≥2 risk factors (sTIM-3≤530pg/mL, BMI≤22.5, and radiographic score ≥5) were 13 times more likely to exhibit NTM-LD than those without (aOR 13.234 [2.983–58.709], P=.001).
ConclusionssTIM-3 was an independent factor for differentiating NTM-LD from NTM-Col, suggesting the immunologic role of sTIM-3 in NTM-LD pathogenesis. By assessing sTIM-3 levels and other risk factors, physicians may be able to identify NTM-LD cases in a simplified manner.
Nontuberculous mycobacteria (NTM) are opportunistic pathogens that are abundant in the human environment.1,2 NTM can cause NTM lung disease (NTM-LD) in patients with structural lung disease and patients who are immunocompromised. NTM can also cause pulmonary NTM colonization (NTM-Col). Alarmingly, the incidence of NTM-LD has been increasing worldwide over the last 3 decades.3–6 The American Thoracic Society (ATS)/Infectious Diseases Society of America (IDSA) guideline published in 2007 defines diagnostic criteria for NTM-LD, and retrospective surveys have determined that 25%–43% of patients with respiratory NTM isolates satisfy ATS/IDSA criteria for NTM-LD.7–9 However, a simple method for physicians to exclude NTM-Col and identify NTM-LD has not been developed. Female sex, low body mass index (BMI) or weight loss, previous pulmonary tuberculosis, comorbid chronic obstructive pulmonary disease, sputum acid-fast bacilli (AFB) smear positivity, nodular or tree-in-bud lesions, and cavitary and bilateral lung disease were risk factors for NTM-LD defined by the ATS/IDSA guideline (ATS-meeting NTM-LD) over NTM-Col.8–12 Furthermore, certain NTM species, such as Mycobacterium avium complex (MAC), M. abscessus, M. kansassi, and M. malmoense are highly correlated with NTM-LD.8,9,12,13 Specifically, a high titer of antibodies to the glycopeptidolipid core antigen of MAC is associated with MAC pulmonary infection but not colonization.14 However, reports on potential serologic biomarker assistance in the differentiation of NTM-LD from NTM-Col in patients with respiratory samples cultures that are positive for NTM (NTM-positive) remain scarce.
Patients with NTM-LD display chronic systemic inflammation and generally exhibit leukocytosis, elevated C-reactive protein, and erythrocyte sedimentation rates.13,15,16 However, these factors are highly correlated with other clinical characteristics of patients and have not been identified as independent factors for ATS-meeting NTM-LD. Studies have reported that patients with NTM-LD exhibit elevated programmed cell death-1 (PD-1), PD ligand-1 (PD-L1), and T-cell immunoglobulin and mucin domain-3 (TIM-3) protein expression on T lymphocytes compared with healthy controls; this finding may be associated with immune tolerance in patients with NTM-LD.17–19 PD-1 and TIM-3 are inhibitory receptors and a family of immune checkpoint proteins.20 However, whether these immune checkpoint profiles differ between NTM-LD and NTM-Col warrants investigation.
In contrast to immune checkpoint proteins expressed on the cell membrane, soluble-form TIM-3 (sTIM-3), soluble PD-1 (sPD-1), and soluble PD-L1 (sPD-L1) are present in humans and easy to detect in serum.21,22 Although their biological significance is not fully understood, these proteins are thought to be products of alternatively spliced variants or shedding from immune cells. Notably, sTIM-3 and sPD-L1 are prognostic factors in patients with certain chronic infectious diseases.23,24 However, the association between soluble immune checkpoint proteins and NTM-LD in patients who are NTM positive remains unknown. Therefore, given the potential differences in the immune exhaustion status between patients with NTM-LD and NTM-Col, this study aims to (1) to evaluate the values of immune checkpoint proteins, especially their soluble form, on differentiating NTM-LD and NTM-Col since 2016 and (2) further investigate the relationship between soluble-form and membrane-bound immune checkpoint proteins in subjects enrolled in 2019.
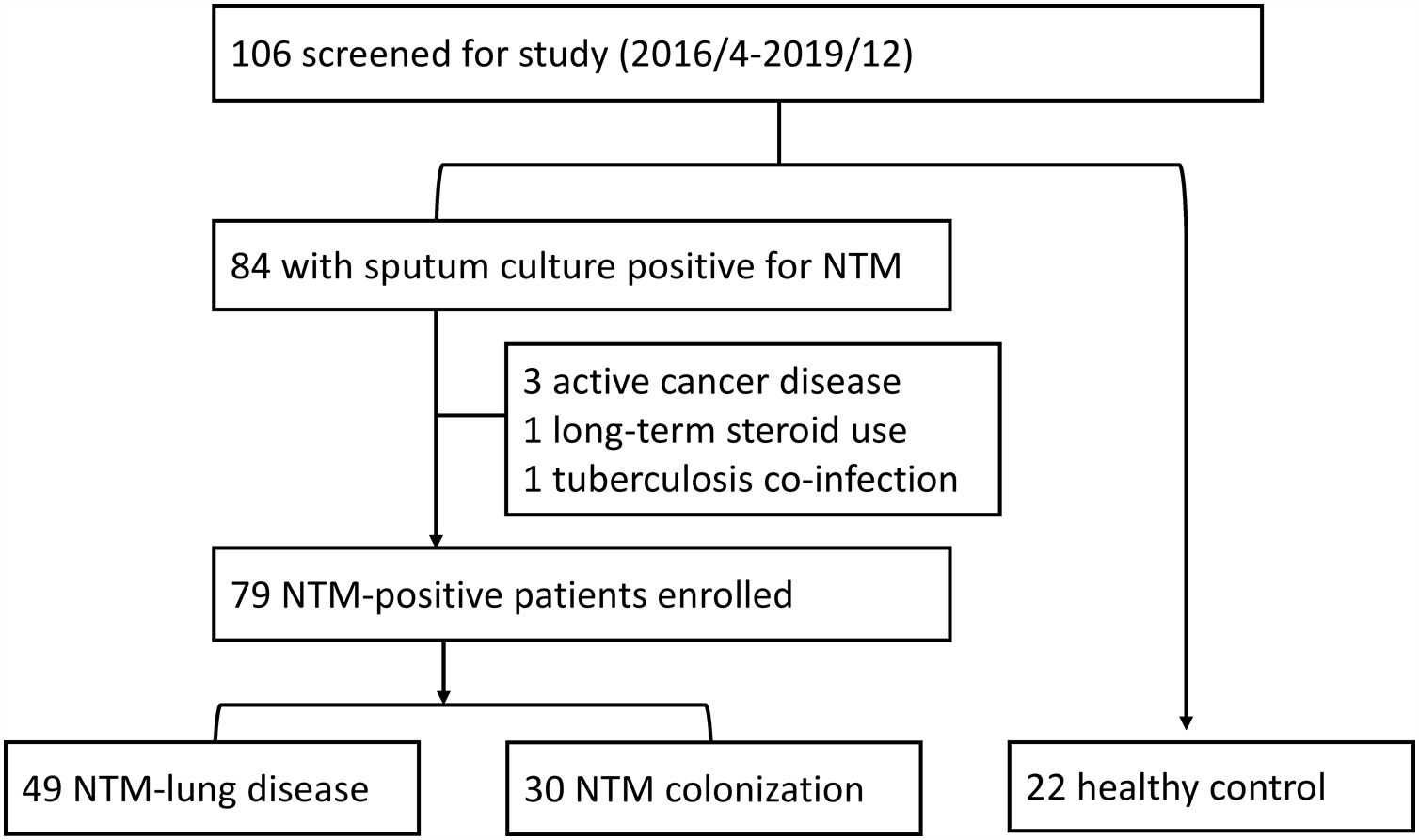
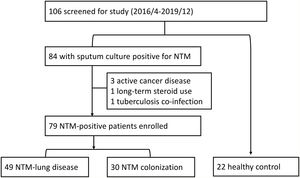
MethodsDesign and patient enrollmentThe present prospective study was conducted from April 2016 to December 2019 at Taipei Veterans General Hospital, a tertiary referral medical center in northern Taiwan. Adult patients with a respiratory sample culture positive for NTM were enrolled at the chest clinics of this hospital. Healthy participants were also recruited as the control group from outpatient clinics if their chest radiographic images revealed no abnormalities. The exclusion criteria were as follows: (1) testing positive for human immunodeficiency virus (HIV) infection, (2) having active cancer requiring treatment or other long-term immunosuppressive agents, and (3) having mixed pulmonary infection by NTM and Mycobacterium tuberculosis. This study was conducted in accordance with the Declaration of Helsinki. The institutional review board (IRB) of the Taipei Veterans General Hospital (IRB No. 2014-09-008BC, 2017-09-010C, and 2017-12-001C) approved this study. All participants provided written informed consent.
Data collection and definitionsClinical data, including age, sex, BMI, comorbidities, and symptoms were collected upon enrollment. Mycobacterium culture results, grading of sputum AFB-smears, number of NTM-positive sputum samples in the first 3 months, radiographic findings, and radiographic score were recorded as previously reported.25,26 Routine identification of NTM species by using a molecular diagnostic biochip was a regular practice in 2016–2017 at this hospital.27 This procedure was no longer routine in 2018–2019 and had to be requested by a physician within 3–6 months after an NTM was isolated. Therefore, belated referrals to the chest clinic could prevent NTM species identification, despite the patient being enrolled.
Two pulmonologists (Dr Pan and Dr Feng) determined whether an NTM-positive case satisfied the ATS/IDSA-defined criteria for the diagnosis of NTM-LD within 3 months after enrollment.7 (see criteria in Supplementary material) Accordingly, patients were classified into the ATS-meeting NTM-LD group and the NTM colonization group (NTM-Col). All NTM-positive participants were observed for at least 6 months.
Enzyme-linked immunosorbent assay and flow cytometry dataAll participants received blood drawing upon enrollment. Serum samples were stored at −80°C. The serum levels of sPD-1, sPD-L1, and sTIM-3 were measured using enzyme-linked immunosorbent assays with commercially available kits according to manufacturer instructions (Catalog No. BMS2214, BMS2212, and BMS2219, respectively; Invitrogen; Thermo Fisher Scientific, Inc.). Technicians determined the concentrations of the markers but were unaware of the clinical characteristics of the enrolled participants.
To explore the association between soluble-form immune checkpoint proteins in serum and their membrane-bound form in T lymphocytes, flow cytometry of peripheral blood mononuclear cells (PBMCs) was performed in patients enrolled in 2019. First, using patients’ peripheral blood samples in heparin-containing tubes, PBMCs were isolated through density gradient centrifugation (Ficoll-Paque PLUS, GE Healthcare Life Sciences, Sweden). A trypan blue dye exclusion test was used to determine cellular viability, which always exceeded 95%. The cells were then stored in a 4% paraformaldehyde solution in PBS at 4°C for up to a week until the scheduled experiment. Second, the PBMCs for CD4, CD8, PD-1, PD-L1, and TIM-3 were stained and then measured using flow cytometry (FACSCanto II, BD Biosciences, USA). The staining antibodies were anti-CD4-APC-H7, anti-CD8-FITC, anti-PD-1-APC, anti-PDL1-APC, and anti-TIM-3-PE (BD Biosciences, USA). The lymphocyte population was identified using forward and side scatter. The CD4+ and CD8+ T lymphocytes were then gated, and the expression of PD-1, PD-L1, and TIM-3 was measured. Flow cytometry data were analyzed using the FlowJo software package (BD, Biosciences, USA).
Statistical analysisFirst, Student's t test and the Mann–Whitney U test were used to compare the groups for normally distributed continuous variables (expressed as mean±SD) and nonnormally distributed variables (expressed as median with interquartile range [IQR]), respectively. A chi-square test was used to analyze intergroup differences in percentages (%) for categorical variables. Logistic regression analysis was used to identify factors associated with ATS-meeting NTM-LD. Indicators with P values of <0.10 in univariate analyses were included in the multivariate models with forward stepwise regression. Adjusted odds ratios (ORs) with 95% CIs and corresponding P values were obtained. The analysis was repeated for patients without cavitary lung lesions in a sensitivity analysis. Receiver operating characteristic (ROC) curve analyses were performed and the Youden index was calculated to determine the optimal cutoff points for scores and biomarkers. SPSS v18 (SPSS Inc., Chicago, IL, USA) was employed to perform analyses.
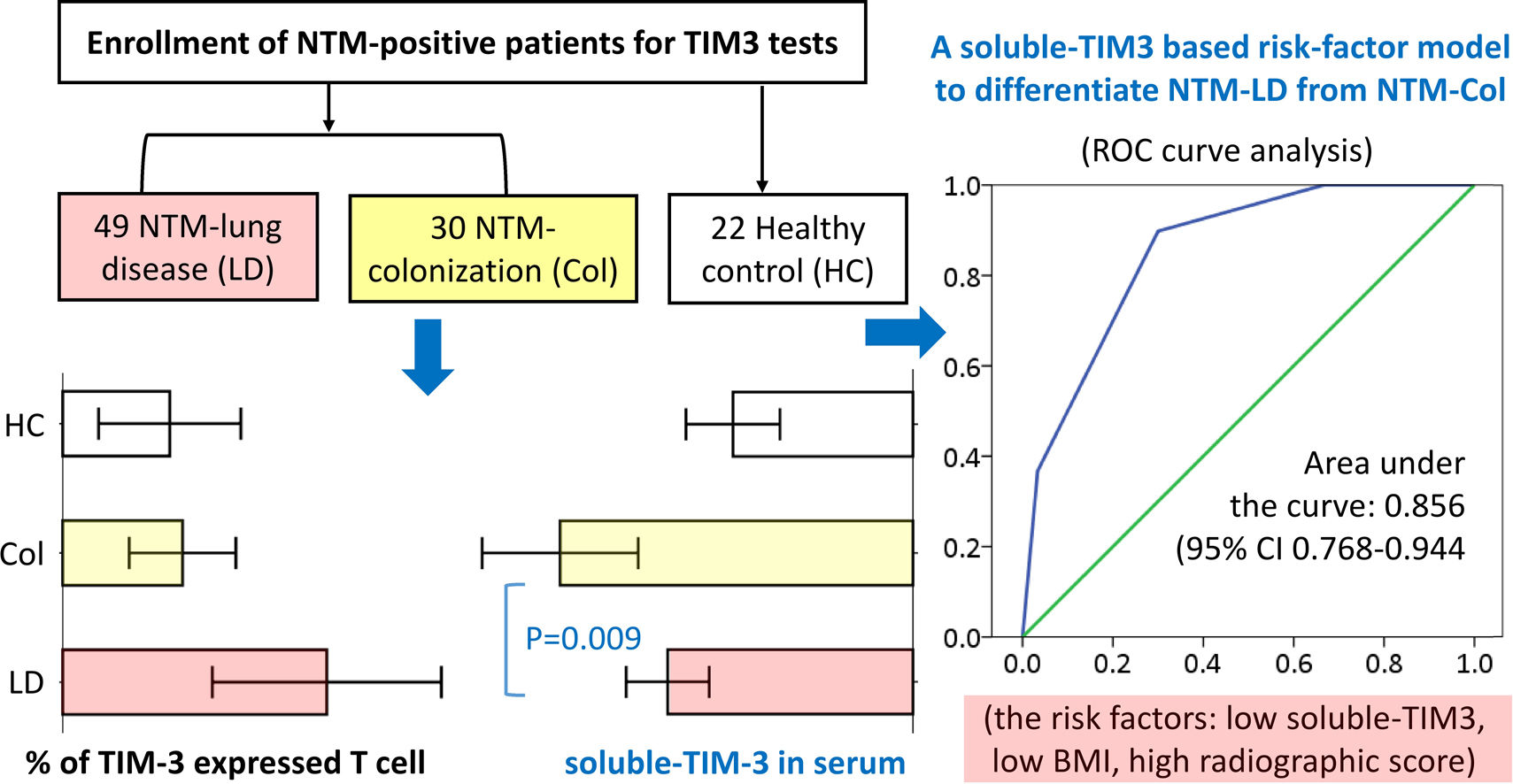
ResultsParticipant and immune checkpoint proteinsWe enrolled 101 participants, including 79 patients and 22 healthy controls (Fig. 1). Among the 79 patients, 49 satisfied the ATS diagnostic criteria for NTM-LD, and 30 did not (NTM-Col group). As presented in Table 1, the sTIM-3 level was significantly higher in the NTM-Col group than in the NTM-LD group (856.3±518.7 vs. 595.3±352.6pg/mL, P=.009). However, the levels of serum sPD-1 and sPD-L1 were not statistically different between the 2 groups. sTIM-3 level was also higher in the NTM-Col group than in the control group (P<.001).
Characteristics of patients with NTM-positive respiratory sample cultures and healthy controls (n=101).
| Variables | NTM-positive patients | Healthy control (n=22) | ||
|---|---|---|---|---|
| NTM-LD (n=49) | NTM-Col (n=30) | P value | ||
| Age, year | 67.0±13.7 | 69.9±15.3 | 0.386 | 51.1±21.4 |
| Male sex | 13 (27) | 18 (60%) | 0.004 | 10 (45%) |
| BMI (kg/m2)a | 19.3 [17.1–21.0] | 22.8 [19.6–27.4] | <0.001 | 22.8 [19.5–25.8] |
| Ever-smoker | 7 (14%) | 3 (10%) | 0.734 | 2 (9%) |
| Comorbidity number | ||||
| COPD | 7 (14%) | 4 (13%) | 1.000 | 0 |
| Autoimmune disease | 4 (8%) | 2 (7%) | 1.000 | 0 |
| Diabetes mellitus | 5 (10%) | 3 (10%) | 1.000 | 1 (5%) |
| Malignancy history | 5 (10%) | 8 (27%) | 0.068 | 2 (9%) |
| Prior TB history | 5 (10%) | 4 (13%) | 0.724 | 2 (9%) |
| Symptoms | ||||
| Cough | 33 (67%) | 20 (67%) | 1.000 | |
| Hemoptysis | 13 (27%) | 6 (20%) | 0.595 | |
| Shortness of breath | 3 (6%) | 2 (7%) | 1.000 | |
| Weight loss | 7 (14%) | 1 (3%) | 0.147 | |
| Initial radiographic pattern | ||||
| Fibro-cavitary lesion | 10 (20%) | 0 | 0.011 | |
| Bilateral lung disease | 42 (86%) | 14 (47%) | <0.001 | |
| Radiographic scorea | 6.0 [4.0–9.5] | 2.0 [1.0–5.0] | <0.001 | |
| Radiographic score ≥5 | 36 (73%) | 8 (27%) | <0.001 | |
| Sputum AFB-smear positivity | 21 (43%) | 10 (33%) | 0.480 | |
| No. of NTM-positive sputuma | 3 [2–3] | 2 [1–3] | 0.006 | |
| NTM identification requested | 47 (96%) | 21 (70%) | 0.002 | |
| NTM species (n=68)b | 0.027 | |||
| M. abscessus complex | 10 (21%) | 4 (9%) | ||
| M. avium complex | 31 (66%) | 7 (33%) | ||
| Other species | 8 (17%) | 10 (48%) | ||
| Soluble serum biomarkers | ||||
| sPD-1 (pg/mL) | 57.5±47.2 | 78.4±65.1 | 0.103 | 61.0±40.3 |
| sPD-L1 (pg/mL) | 8.5±16.8 | 13.9±27.6 | 0.276 | 8.9±7.9 |
| sTim-3 (pg/mL) | 595.3±352.6 | 856.3±518.7 | 0.009 | 437.0±267.4 |
Continuous and categorical data are expressed as mean±standard deviation (SD) and number (%), respectively.
The variables that were nonnormally distributed were also expressed as the median [IQR] and analyzed using the Mann–Whitney U test.
Two patients who had mixed infection by M. abscessus complex and M. avium complex were assigned to the M. abscessus complex in the chi-square test.
Abbreviations: AFB, acid-fast bacilli; ATS, American Thoracic Society; BMI, body mass index; COPD, chronic obstructive pulmonary disease; NTM-Col, nontuberculous mycobacterium colonization; NTM-LD, NTM lung disease; sPD-1, soluble programmed cell death-1; sPD-L1, soluble programmed cell death ligand-1; sTIM-3, soluble T-cell immunoglobulin mucin domain-3; TB, tuberculosis.
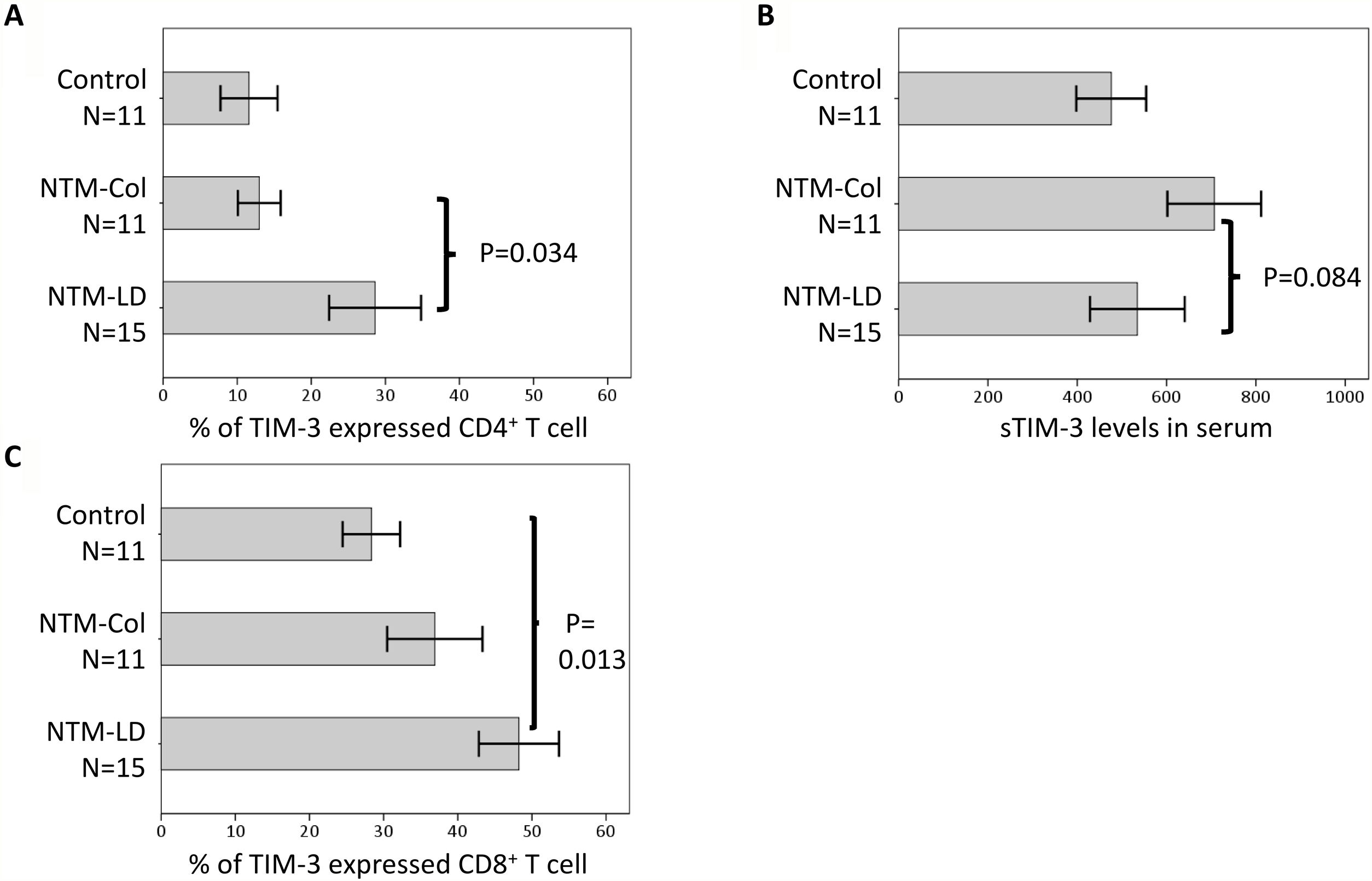
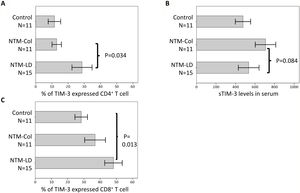
For the subgroup enrolled in 2019 with flow cytometry data (n=37, Fig. 2), the proportions of TIM-3 expression on CD4+ T cells were highest in the NTM-LD group (n=15), followed by the NTM-Col group (n=11), and the controls (n=11; P=.017 for trend). This expression trend was also observed on CD8+ T cells (P=.011). No statistical differences were observed in the percentages of PD-1 and PD-L1 expression on CD4+ and CD8+ T cells among the 3 groups (Supplemental Table S1). Compared with NTM-Col, NTM-LD was marginally associated with an elevated TIM-3 expression in CD4+ T cells and a reduced sTIM-3 level after adjustment for age, sex, and BMI in the subgroup multivariate logistic analysis (P=.098 and P=.060).
Proportions of T-cell immunoglobulin mucin domain-3 (TIM-3) expression on CD4+ and CD8+ T lymphocytes (A and B) and soluble sTIM-3 in serum (sTIM-3) (C) in patients enrolled in 2019. The data in the bar charts are mean values, and error bars are standard errors. Intergroup difference is not statistically different in cases without P values.
The factors associated with NTM-LD in the univariate analyses were female sex, low BMI, elevated sTIM-3 level, bilateral lung disease, radiographic score ≥5, NTM-positive sputum, and certain NTM species (Table 2). In the multivariate analysis using forward selection, the independent factors retained were BMI (per 1-kg/m2 increase; OR, 0.759; 95% CI, 0.626–0.920; P=.005), sTIM-3 level (per 100-pg/mL increase; OR, 0.732; 95% CI, 0.612–0.877; P=.001), and radiographic score ≥5 (OR, 7.649; 95% CI, 1.904–30.731; P=.004; Table 2).
Analysis of factors associated with NTM-LD in patients with NTM-positive respiratory sample cultures (n=79).
| Variables | Univariate | Multivariate, forward selection | Multivariate, fully adjusted | |||
|---|---|---|---|---|---|---|
| Crude OR (95% CI) | P value | Adjusted OR (95% CI) | P value | Adjusted OR (95% CI) | P value | |
| Age (years) | 0.985 (0.953–1.019) | 0.383 | 1.002 (0.954–1.052) | 0.947 | ||
| Male sex | 0.241 (0.092–0.633) | 0.004 | – | 0.233 (0.039–1.402) | 0.112 | |
| BMI (kg/m2) | 0.777 (0.674–0.895) | <0.001 | 0.759 (0.626–0.920) | 0.005 | 0.825 (0.664–1.026) | 0.083 |
| Ever-smoker | 1.500 (0.357–6.308) | 0.580 | ||||
| sTim-3 (100-pg/mL increment) | 0.865 (0.768–0.974) | 0.016 | 0.732 (0.612–0.877) | 0.001 | 0.658 (0.502–0.864) | 0.003 |
| Bilateral lung disease | 6.857 (2.341–20.083) | <0.001 | – | |||
| Radiographic score ≥5a | 7.615 (2.724–21.291) | <0.001 | 7.649 (1.904–30.731) | 0.004 | 11.304 (1.612–79.253) | 0.015 |
| Sputum AFB-smear positivity | 1.500 (0.582–3.866) | 0.401 | 2.611 (0.483–14.118) | 0.265 | ||
| No. of NTM-positive sputumb | 1.729 (1.009–2.964) | 0.047 | – | |||
| NTM subspecies (n=68) | ||||||
| Other species | 1.0 (reference) | 1.0 (reference) | ||||
| M. avium complex | 3.125 (0.707–13.814) | 0.133 | 3.330 (0.380–29.154) | 0.277 | ||
| M. abscessus complex | 5.179 (1.494–17.953) | 0.010 | 12.569 (1.378–114.60) | 0.025 | ||
Abbreviation: AFB, acid-fast bacilli; BMI, body mass index; NTM-LD, nontuberculous mycobacterium (NTM) lung disease; sTIM-3, soluble T-cell immunoglobulin mucin domain-3.
sTIM-3 level remained an independent factor for NTM-LD after adjustment for age, sex, BMI, radiographic score ≥5, sputum-smear positivity, and NTM species (per 100-pg/mL increase; OR, 0.658; 95% CI, 0.502–0.864; P=.003; Table 2). A sensitivity analysis with further adjustment for the levels of sPD-1 and sPD-L1 revealed similar results. Because all participants with cavitary lesions (n=10, 13%) had NTM-LD, the presence of cavitary lesions was not included in the model. In a subgroup analysis of patients without cavitary lesions (n=69, 87.3%), sTIM-3 levels remained independently associated with NTM-LD (per 100-pg/mL increase; OR, 0.618; 95% CI, 0.450–0.847; P=.003; Supplemental Table S2).
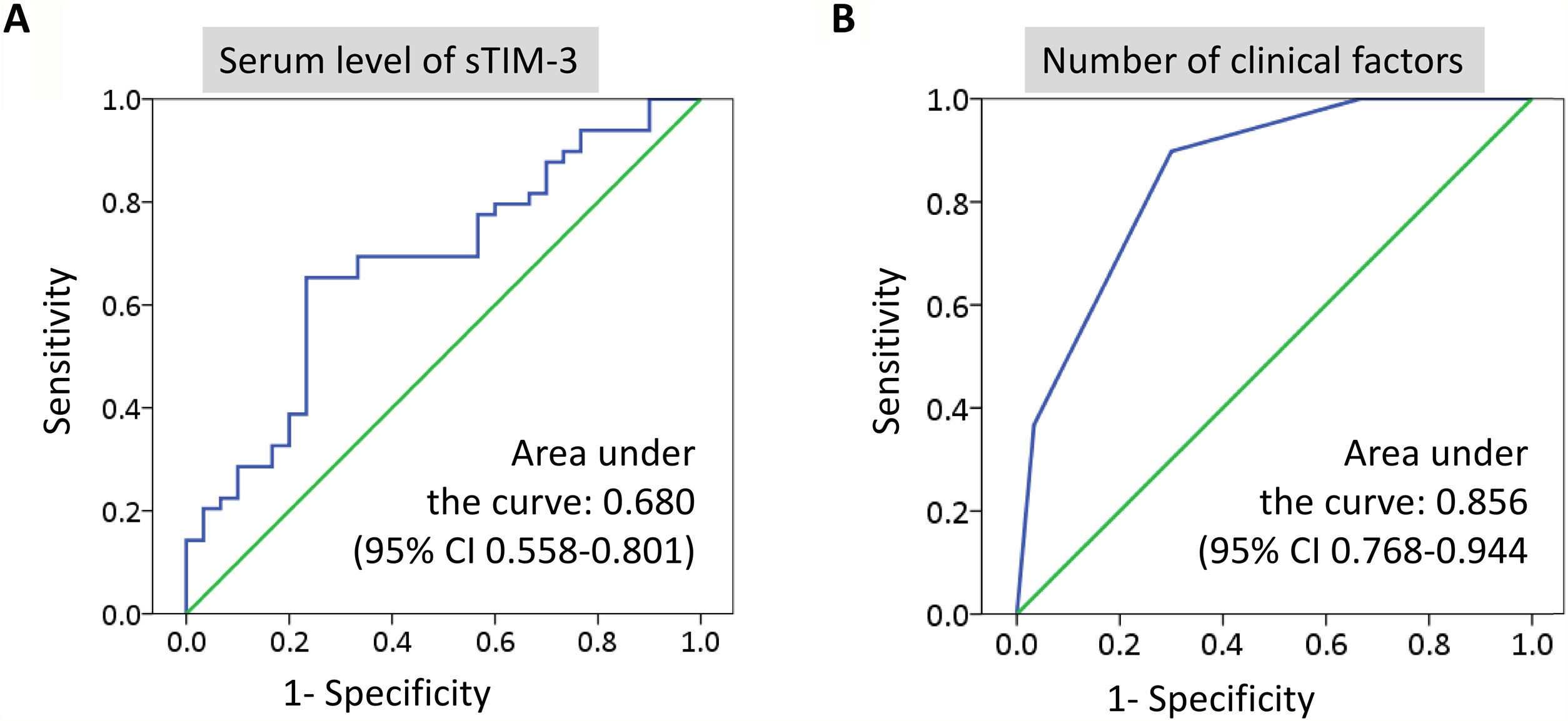
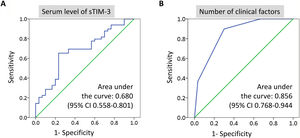
Synergistic effect of sTIM-3 and other indicators in identifying NTM-LDROC curve analysis revealed that the optimal cutoff point for sTIM-3 to detect ATS-meeting NTM-LD is 530pg/mL (area under the curve: 0.680; 95% CI, 0.558–0.801; P=.008). Sensitivity and specificity of using sTIM-3 ≤530pg/mL to identify NTM-LD cases were 76.7% and 65.3%, respectively. Furthermore, sTIM-3 ≤530pg/mL was significantly associated with the NTM-LD group (OR, 5.176; 95% CI, 1.904–14.077; P=.001; Fig. 3A). (See Supplementary material for factors associated with sTIM-3 level).
Receiver operating characteristic curve analysis of soluble T-cell immunoglobulin mucin domain-3 (sTIM-3) level (A) and the number of risk factors (B) to differentiate nontuberculous mycobacterial (NTM)-lung disease and NTM colonization. The risk factors were low sTIM-3 levels (≤530pg/mL), body mass index ≤22.5 (kg/m2), and radiographic score ≥5.
Using Youden index to evaluate the cutoff values of relevant biomarkers, we defined 3 risk factors for NTM-LD: BMI ≤22.5, sTIM-3 ≤530, and radiographic score ≥5. We classified NTM-positive cases into 4 groups with 0–3 risk factors, respectively (see Supplemental Table S3). ROC curve analysis revealed that the optimal cutoff for identifying ATS-meeting NTM-LD was ≥2 risk factors with a sensitivity of 89.8% and a specificity of 70.0% (area under the curve: 0.856; 95% CI, 0.768–0.944; P<.001; Fig. 3B). Patients with ≥2 risk factors exhibited a 13-fold increase in the odds of exhibiting NTM-LD compared with those with <2 factors after adjustment for age, sex, smear positivity, and NTM species (OR, 13.234; 95% CI, 2.983–58.709; P=.001).
DiscussionThis study revealed that, sTIM-3 was an independent factor for differentiating NTM-LD from NTM-Col in NTM-positive patients. Moreover, patients with ≥2 risk factors (low sTIM-3 level [≤530pg/mL], BMI<22.5, and radiographic score ≥5) were 13 times more likely to have NTM-LD than were those without. Our findings may suggest that sTIM-3 combining other biomarkers can be sensitive tools to differentiate NTM-LD from NTM-Col.
Patients with immunosuppression or structural lung disease are prone to NTM-LD. However, NTM-Col is frequently observed in these patients. Antibiotic treatment is generally not necessary for NTM-Col. Differentiating true infection from NTM-Col is crucial in caring for patients who are NTM positive. However, differentiating between NTM-LD and NTM-Col is difficult because they have similar symptoms and image patterns, especially among patients with underlying lung disease.28 The ATS/IDSA diagnostic criteria, consisting of symptom, microbiologic, and radiographic indicators, do not have clear cutoff ranges.7 Furthermore, although NTM-LD is a chronic inflammatory infectious disease with immune tolerance in nature,18,19 the diagnostic standard does not account for the effect of patients’ immune profiles. We revealed that the level of sTIM-3 in the NTM-Col group was significantly different from that in the NTM-LD group, whereas sPD-1 and sPD-L1 levels did not differ. Although the relevance between soluble and membrane-bound TIM-3 in NTM-LD remain unclear, our study is the first to provide evidence that sTIM-3 is independently associated with NTM-LD.
As a negative regulatory immune checkpoint, TIM-3 is a transmembrane protein expressed on T cells and monocytes, and its expression is associated with T-cell exhaustion in chronic viral infection, tuberculosis, and NTM-LD.19,20,29,30 We determined that TIM-3 expression on CD4+ T cells is elevated in patients with NTM-LD compared with the controls; this finding is consistent with a recent report.19 We also noted that TIM-3 expression in NTM-LD was significantly higher than that in the NTM-Col subgroup. Notably, TIM-3 can depart from the cell surface to become a surface-shed sTIM-3 after cleaving by certain matrix metalloproteinases. An alternatively spliced sTIM-3 may exist in mice, but one has not been identified in humans.21,23,31 Studies in patients with HIV infections revealed that sTIM-3 level was positively associated with the level of TIM-3 expression on the cell membrane and was correlated with disease progression.23,32 Our study targeted NTM-Col subgroups without true infection and demonstrated that patients with NTM-Col had a relatively low degree of TIM-3 expression but a high level of sTIM-3 compared with patients with NTM-LD. We think, because sTIM-3 is most likely membrane-shed in humans, one possible explanation for this phenomenon is that NTM-Col patients have an elevated TIM-3-shedding against the increase in TIM-3-expression on T cells. This mechanism may protect patients with NTM-Col from profound immune exhaustion, which occurs in patients truly infected by NTM. Some experts proposed that sTIM-3 may block the TIM-3-ligand pathway to restore T-cell function in patients with HIV.33 However, the biological roles of sTIM-3 in patients with NTM-LD and NTM-Col warrant further study.
Studies have indicated that similar to TIM-3, the degrees of PD-1 expression on T lymphocytes are higher in patients with NTM-LD than in healthy controls.17–19 However, we determined that the degrees of PD-1 expression on CD4+ T cells and the serum levels of sPD-1 were not significantly different between the NTM-LD and NTM-Col groups. The limited sample size of this study may partially contribute to these results. In addition, the association between NTM-LD and healthy groups is dissimilar to the association between NTM-LD and NTM-Col because, unlike patients with NTM-Col, healthy controls have no NTM related host–pathogen interaction. Notably, the level of sTIM-3 was highly correlated with sPD-1 and sPD-L1 levels in patients who were NTM positive. A basic study in patients with HIV revealed that TIM-3 and PD-1 blocking synergistically restored macrophage and T cells function and the ability to control tuberculosis.34 Although the mechanism is unknown, the interaction between TIM-3 and PD-1 pathways must be complex in patients with infectious diseases. Crucially, our sensitivity analysis with further adjustment for sPD-1 and sPD-L1 levels confirmed that low sTIM-3 was independently associated with NTM-LD instead of NTM-Col diagnosis.
Several studies have reported potential serological biomarkers for differentiating NTM-LD from NTM-Col. Placenta growth factor and vascular endothelial growth factor are local pulmonary biomarkers in NTM-LD. However, the use of plasma level ratios to differentiate NTM-LD from NTM-Col yielded relatively low sensitivity.11 Similarly, Krebs von den Lungen-6 (KL-6) levels were correlated with the disease progression of NTM-LD. However, because KL-6 also elevated in patients with bronchiectasis without pulmonary NTM isolates, its specific value that could be used to differentiate NTM-LD from NTM-Col was unclear.35 By contrast, we determined that sTIM-3 is an immunologic biomarker for NTM-LD in NTM-positive patients, even in more difficult-to-diagnose populations without cavitary lesions. A suppressed level of sTIM-3 ≤530pg/mL combined with other clear indicators, including BMI ≤22.5 and radiographic score ≥5, could be used as reliable screening tools to detect NTM-LD in patients who are NTM positive with a sensitivity of 89.8%.
This study had several limitations. First, the sample size was relatively small. Nevertheless, our findings were robust because the multivariate models in sensitivity and subgroup analyses confirmed the results. However, because we enrolled only 10 patients with cavitary disease in this NTM study, the diagnostic value and pathogenic role of sTIM3 in patients with cavitary disease deserves further large-scale investigation. Second, no long-term follow-up data were acquired to evaluate changes in sTIM-3 in patients with NTM-LD with and without disease progression. Further research investigating the prognostic value of sTIM-3 on progressive NTM-LD is warranted. Third, the association between sTIM-3 and its membrane-bound form remains unclear. However, the flow cytometry data analysis indicated that sTIM-3 has an independent role in NTM infection because it remained marginally associated with NTM-LD diagnosis after adjustment for TIM-3 expression.
In conclusion, sTIM-3 was an independent factor for differentiating NTM-LD from NTM-Col, suggesting an immunologic role of sTIM-3 in NTM infection. Physicians may be able to identify NTM-LD cases in a simplified manner among patients with NTM-positive respiratory samples by assessing sTIM-3 levels and other risk factors.
Author contributionsDrs Pan SW, Feng JY, and Chan YJ collected data. Drs Pan SW, Shu CC, Lee CC, Feng JY, and Su WJ performed data analysis. Drs Pan SW, CC Shu, and Feng JY wrote the manuscript. Profs. Chan YJ, Chen YM, and Su WJ provided critical revisions for the paper. Drs Feng JY and Su WJ were the guarantors of the paper, taking responsibility for the integrity of the work as a whole, from inception to published article.
FundingThis study was supported by the Taipei Veterans General Hospital/National Taiwan University Hospital Joint Research Program [grant number: VN105-11, VN106-15, VN108-14, VN109-08], Taipei Veterans General Hospital [V107B-016, V108B-039 and V109C-053], and the Ministry of Science and Technology, Taiwan [MOST106-2314-B-075-007, 107-2314-B-075-057, 108-2314-B-075-001, 109-2314-B-075-094 and 109-2628-B-075-026].
Conflicts of interestThe authors do not have any conflicts of interest.
The authors thank the staff of the Division of Microbiology, Department of Pathology and Laboratory Medicine, Taipei Veterans General Hospital, for their assistance in collecting microbiological data. The authors thank the Medical Science and Technology Building of Taipei Veterans General Hospital for providing research facilities. This manuscript was edited by Wallace Academic Editing.