Prognosis in pulmonary arterial hypertension (PAH) has significantly improved after the discovery of some of the pathophysiological bases of the disease and the development of drugs involving these pathways.1 Treatment strategy is no longer based solely on functional class, as it was when first vasodilatory drugs were commercialized. Prognostic stratification and treatment strategy is based now in multiparametric evaluations of several clinical, analytical, functional, and cardiac imaging parameters. Current clinical practice guidelines recommend the combination of variables to discriminate individual prognosis at diagnosis and during follow-up, counselling the intensification of treatment according to the individual prognosis.2 Despite those advances, long-term survival is still far from being perfect, and the patient's response to the available drugs seems to be different. In this setting, prognostic scores are evolving and adapting to the different PAH phenotypes,3 although several gaps remain unsolved to date.
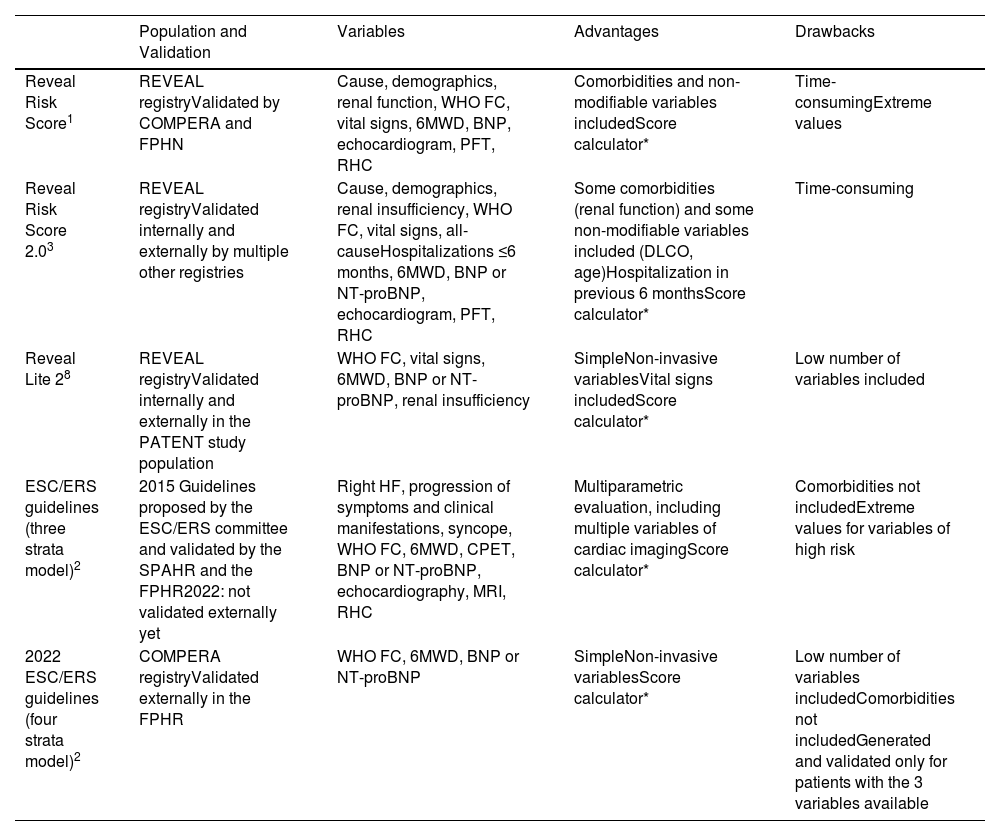
First, the European guidelines of pulmonary hypertension have recently updated the recommendations on prognostic stratification at diagnosis, including some parameters of right ventricle (RV) function.2 This was a long-standing request in the field since the RV function determines symptoms and prognosis. In PAH, the RV adapts to the initial afterload increase by augmenting its contractility and cardiac mass. In case of failure of this adaptative mechanism, dilation of the right heart chambers and systemic congestion appear.4 The tricuspid annular plane systolic excursion (TAPSE) and the systolic pulmonary artery pressure (sPAP) are easily measurable load-dependent parameters of RV contractility and pressure overload, respectively. The TAPSE/sPAP emerges as a parameter of ventriculoarterial coupling, with lower values indicating a maladaptive RV response to the pressure overload, eliminating the influence of the volume overload in this equation. This ratio has been validated in multiple clinical scenarios, improving the mechanistic interpretation of the failing RV in PAH.5 Whilst cardiac magnetic resonance, now considered in the European guidelines,2 measures profoundly the complex RV structure and function, this technique is still used barely in clinical practice. Related with the prior considerations on RV function, some authors defend an aggressive afterload lowering as the main therapeutic target to ameliorate prognosis by improving the RV adaptation to the disease.6 On the other hand, the need for regular risk assessment and the daily clinical practice barriers to implement more complex risk scores – especially the lack of time – was one of the main motivations for introducing simpler risk stratification methods at follow-up. This strategy is based on the adequate applicability of only three widespread parameters (six-minute walk test distance, functional class, and cardiac biomarkers).7 Equally, the REVEAL Lite 2.0 score defend this easier approach, with the sum of other quickly measurable important prognostic parameters: renal function, blood pressure and heart rate.8 Whereas the simplification is now the rule, other risk markers undoubtedly related with prognosis like the diffusing capacity of the lung for carbon monoxide, or the presence of complications associated with a pulmonary artery aneurism are now neglected9,10 (Table 1).
Main Characteristics of the Most Extended Prognostic Models for Pulmonary Arterial Hypertension.
| Population and Validation | Variables | Advantages | Drawbacks | |
|---|---|---|---|---|
| Reveal Risk Score1 | REVEAL registryValidated by COMPERA and FPHN | Cause, demographics, renal function, WHO FC, vital signs, 6MWD, BNP, echocardiogram, PFT, RHC | Comorbidities and non-modifiable variables includedScore calculator* | Time-consumingExtreme values |
| Reveal Risk Score 2.03 | REVEAL registryValidated internally and externally by multiple other registries | Cause, demographics, renal insufficiency, WHO FC, vital signs, all-causeHospitalizations ≤6 months, 6MWD, BNP or NT-proBNP, echocardiogram, PFT, RHC | Some comorbidities (renal function) and some non-modifiable variables included (DLCO, age)Hospitalization in previous 6 monthsScore calculator* | Time-consuming |
| Reveal Lite 28 | REVEAL registryValidated internally and externally in the PATENT study population | WHO FC, vital signs, 6MWD, BNP or NT-proBNP, renal insufficiency | SimpleNon-invasive variablesVital signs includedScore calculator* | Low number of variables included |
| ESC/ERS guidelines (three strata model)2 | 2015 Guidelines proposed by the ESC/ERS committee and validated by the SPAHR and the FPHR2022: not validated externally yet | Right HF, progression of symptoms and clinical manifestations, syncope, WHO FC, 6MWD, CPET, BNP or NT-proBNP, echocardiography, MRI, RHC | Multiparametric evaluation, including multiple variables of cardiac imagingScore calculator* | Comorbidities not includedExtreme values for variables of high risk |
| 2022 ESC/ERS guidelines (four strata model)2 | COMPERA registryValidated externally in the FPHR | WHO FC, 6MWD, BNP or NT-proBNP | SimpleNon-invasive variablesScore calculator* | Low number of variables includedComorbidities not includedGenerated and validated only for patients with the 3 variables available |
CPET: cardiopulmonary exercise test; COMPERA: Comparative, Prospective Registry of Newly Initiated Therapies for PH; FC: functional class; FPHR: French Pulmonary Hypertension Registry; PFT: pulmonary function tests; MRI: magnetic resonance imaging; REVEAL: Registry to Evaluate Early and Long-Term PAH Disease Management; RHC: right heart catheterization; SPAHR: Swedish Pulmonary Arterial Hypertension Registry; WHO: World Health Organization; 6MWT: 6-minute walk test.
In the ESC/ERS 3-strata model at diagnosis is recommended to consider as many variables as possible. Low-, intermediate- and high-risk variables are assigned 1, 2, and 3 points, respectively. Then, it is necessary to divide the sum of all numbers by the number of available variables and round to the nearest integer. In the ESC/ERS 4-strata model the three variables are needed. Low-, intermediate–low-, Intermediate–high-, and high-risk variables are assigned 1, 2, 3, and 4 points, respectively. Then, it is necessary to divide the sum of all grades by 3 and round to the nearest integer. In the REVEAL Lite 2 calculator a minimum of 3 variables are required, where at least 2 are of the most predictive variables: cardiac biomarker, 6MWT and FC. The score calculators of REVEAL, REVEAL 2.0 and REVEAL Lite 2 use categorized ranked equations.
Another important unmet question is the applicability of risk calculators in patients with comorbidities, the predominant PAH population. Recently, an analysis from the COMPERA registry identified that the 4-strata model is also useful in PAH with common cardiovascular risk factors (systemic hypertension, coronary artery disease, diabetes, and obesity), with similar precision for predicting mortality when compared with the remaining PAH population, also showing that the prognosis in the intermediate-low stratum was comparable with the low-risk stratum, hardly achieved in this population.11
Finally, it is important to note that novel molecular findings involved in the PAH pathophysiology could be interesting in terms of prognosis. The addition of the genetic results to the multiparametric evaluation could improve the identification of patients at higher risk strata.12 Concerning proteomics, some investigators have recently identified that a fully proteomic-based model (including SVEP1,PXDN, renin, neuropilin-1, thrombospondin-2, and peroxiredoxin-4) predicts very well the survival free of lung transplantation independent of common risk factors.13 Since these biomarkers relates with some vascular remodelling processes, angiogenesis, or fibrosis, they could predict outcomes before an overt RV failure develops. And others have created a cytokine-based model, both at diagnosis and during follow-up, suggesting the role of the pro-inflammatory hypothesis in the prognosis of this multidimensional disease.14
In conclusion, the prognostic evaluation in pulmonary arterial hypertension is an evolving complex scenario. The aim of this strategy is to treat depending on the individual risk of death. Despite recent important discoveries in terms of treatment of this rare disease,15 it is still necessary to design comprehensive prognostic tools to treat each patient more precisely, introducing novel mechanistic considerations in terms of right ventricle function and molecular discoveries, but also considering the applicability of this tools in clinical practice, as well as other currently neglected risk markers or frequent comorbidities.
Conflict of InterestsThe authors state that they have no conflict of interests.