The goal of this investigation is to assess the association between prehospital use of aspirin (ASA) and patient-centered outcomes in a large global cohort of hospitalized COVID-19 patients.
MethodsThis study utilizes data from the Society of Critical Care Medicine Discovery Viral Infection and Respiratory Illness Universal Study (VIRUS) Registry. Adult patients hospitalized from February 15th, 2020, to September 30th, 2021, were included. Multivariable regression analyses were utilized to assess the association between pre-hospital use of ASA and the primary outcome of overall hospital mortality.
Results21,579 patients were included from 185 hospitals (predominantly US-based, 71.3%), with 4691 (21.7%) receiving pre-hospital ASA. Patients receiving ASA, compared to those without pre-admission ASA use, were generally older (median 70 vs. 59 years), more likely to be male (58.7 vs. 56.0%), caucasian (57.4 vs. 51.6%), and more commonly had higher rates of medical comorbidities. In multivariable analyses, patients receiving pre-hospital ASA had lower mortality (HR: 0.89, 95% CI 0.82–0.97, p=0.01) and reduced hazard for progression to severe disease or death (HR: 0.91, 95% CI 0.84–0.99, p=0.02) and more hospital free days (1.00 days, 95% CI 0.66–1.35, p=0.01) compared to those without pre-hospital ASA use. The overall direction and significance of the results remained the same in sensitivity analysis, after adjusting the multivariable model for time since pandemic.
ConclusionsIn this large international cohort, pre-hospital use of ASA was associated with a lower hazard for death in hospitalized patients with COVID-19. Randomized controlled trials may be warranted to assess the utility of pre-hospital use of ASA.
Severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) has infected over 258 million people causing more than 5.1 million deaths worldwide.1 The severity of coronavirus disease 2019 (COVID-19) ranges from asymptomatic infection to mild illness, critical illness, or death.2,3 Although isolated respiratory manifestation of acute illness, COVID-19 may have multisystem involvement with short- and long-term sequela.4,5 In an attempt to better understand outcomes of COVID-19, the Society of Critical Care Medicine Discovery Viral Infection and Respiratory Illness Universal Study (VIRUS) COVID-19 Registry was established and has been engaged in exploring the use of repurposed medication and identification of the practice patterns that could be related to improved patient centric outcomes for hospitalized patients with COVID-19 across the globe.6
It is now well established that microvascular thromboses, arterial thrombotic events (cardiac and cerebrovascular), and major venous thromboembolic events (VTEs) contribute to morbidity and mortality in COVID-19 disease, with dysregulated inflammation thought to be a major driver of these events.7,8 Aspirin or acetylsalicylic acid (ASA) and other anti-platelet agents may counteract inflammation-related neutrophil recruitment and platelet aggregation, thereby reducing intravascular thrombin activity.9 Similar findings have been shown in vitro for SARS-CoV-2 as well, however it is unclear if ASA use may translate to clinical benefit in patients with COVID-19. Several small observational studies have observed lower mortality associated with pre-hospital use of ASA, whereas a recent randomized trial from the RECOVERY collaborative group found no mortality benefit with newly initiated ASA therapy in hospitalized patients, despite finding modestly shorter hospital stays.10–12
In this investigation, we use data from the international VIRUS: COVID-19 registry to assess the associations between prehospital ASA use and clinical outcomes in hospitalized patients with laboratory confirmed COVID-19, with a primary outcome of overall hospital mortality. We hypothesized that pre-hospital ASA use would be associated with reduced risk mortality and improvement in other secondary outcomes such as lower hazard or progression to severe disease and shorter hospitalizations.
Materials and methodsStudy population and settingThis is an observational cohort study of data collected prospectively through the VIRUS: COVID-19 registry. VIRUS: COVID-19 registry was established through the Society of Critical Care Medicine (SCCM) Discovery Network and has provided a centralized platform for international clinical research related to COVID-19. Details of the registry, inception and design are described elsewhere.13,14
The registry includes hospitalized patients of all ages, however, for the purpose of this study only adult patients with laboratory-confirmed COVID-19 are included (>18 years of age). Patients were enrolled at 185 international sites, including academic, community and private hospitals between February 15, 2020 through September 30, 2021. Patients were followed until hospital discharge or death, whichever came first. The VIRUS registry received exempt status for human subject research by the Institutional Review Board (IRB) at the Mayo Clinic (20-002610). Individual study sites submitted a separate proposal to the local IRBs for approval and signed a data use agreement prior to being granted permission to extract and enter de-identified data into the registry case report forms (CRFs). Patients without research authorization were excluded.
Data collectionPrior COVID-19 registry work from the World Health Organization was used as an initial template in VIRUS design.3 Building upon that, clinically relevant data elements were added including patient demographics, medical comorbidities, details on pre-hospital use of medications, acute illness characteristics, various level of oxygen and organ support therapies, medical interventions in the hospital, critical care utilization, and outcomes. We ensured data quality by maintaining bidirectional open communication with all the domestic and international sites and the central registry organization by weekly remote training meetings. Regular quality checks were performed to ensure adherence to the study protocol and accuracy of the data collection procedure. In addition, the registry also performed multistep data cleaning, including active surveillance for entries out of range or of improper data type and contacted sites for correction of data aberrancy. Study data were recorded and managed using the Research Electronic Data Capture system.15 The study was determined to be exempt from Institutional Review Board. The study is registered on Clinicaltrials.gov: NCT04323787.
Exposure of interest and outcome measuresPre-hospital ASA use was the exposure of interest for this study, which was defined by documented use of ASA or any ASA-containing medication at any dose prior to the index hospitalization. This was further confirmed by the prescription information in the electronic medical records and endorsement by the patients. Duration of pre-hospital medication use or most recent administration prior to hospitalization was not documented through the VIRUS registry.
The primary outcome of interest was overall hospital mortality. Secondary outcomes were need for intensive care unit (ICU) admission, progression to severe disease (requiring invasive mechanical ventilation) or death (WHO score 6 or higher),16 hospital and invasive mechanical ventilation (IMV) free days. Other exploratory outcomes included need for any blood product transfusion, new in-hospital complications such as cerebrovascular accident (CVA), cardiac arrest, gastrointestinal hemorrhage, and myocardial infarction. Patient outcomes were censored at the time of hospital discharge.
Statistical analysisPatients were stratified based on whether they received pre-hospital use of ASA or not. Continuous variables were summarized as median (interquartile range [IQR]) and compared between the two groups with Mann–Whitney or Kruskal–Wallis tests, as appropriate. Categorical variables were presented as n (%) and were compared between groups using the Pearson's χ2 test or Fisher's exact test as appropriate. P values less than 0.05 were considered statistically significant.
Unadjusted analyses of outcomes were assessed between groups using Chi-squared tests for categorical variables and t test for continuous variables.
Cox-proportional hazard models were subsequently employed to assess exposure-outcome relationships adjusting the model for potential confounding variables selected a priori, including age, gender, race, ethnicity, comorbidities including coronary artery disease (CAD), hypertension (HTN), congestive heart failure (CHF), chronic kidney disease (CKD), previous history of stroke, chronic pulmonary diseases (including COPD, asthma, and pulmonary circulatory diseases), history of VTE, DM, and obesity. We also performed a sensitivity analysis by removing “race” from the models due to missing data on 2963 patients.
All statistical analyses were performed using JMP statistical software base version 14.0 (SAS Institute Inc., Cary, NC).
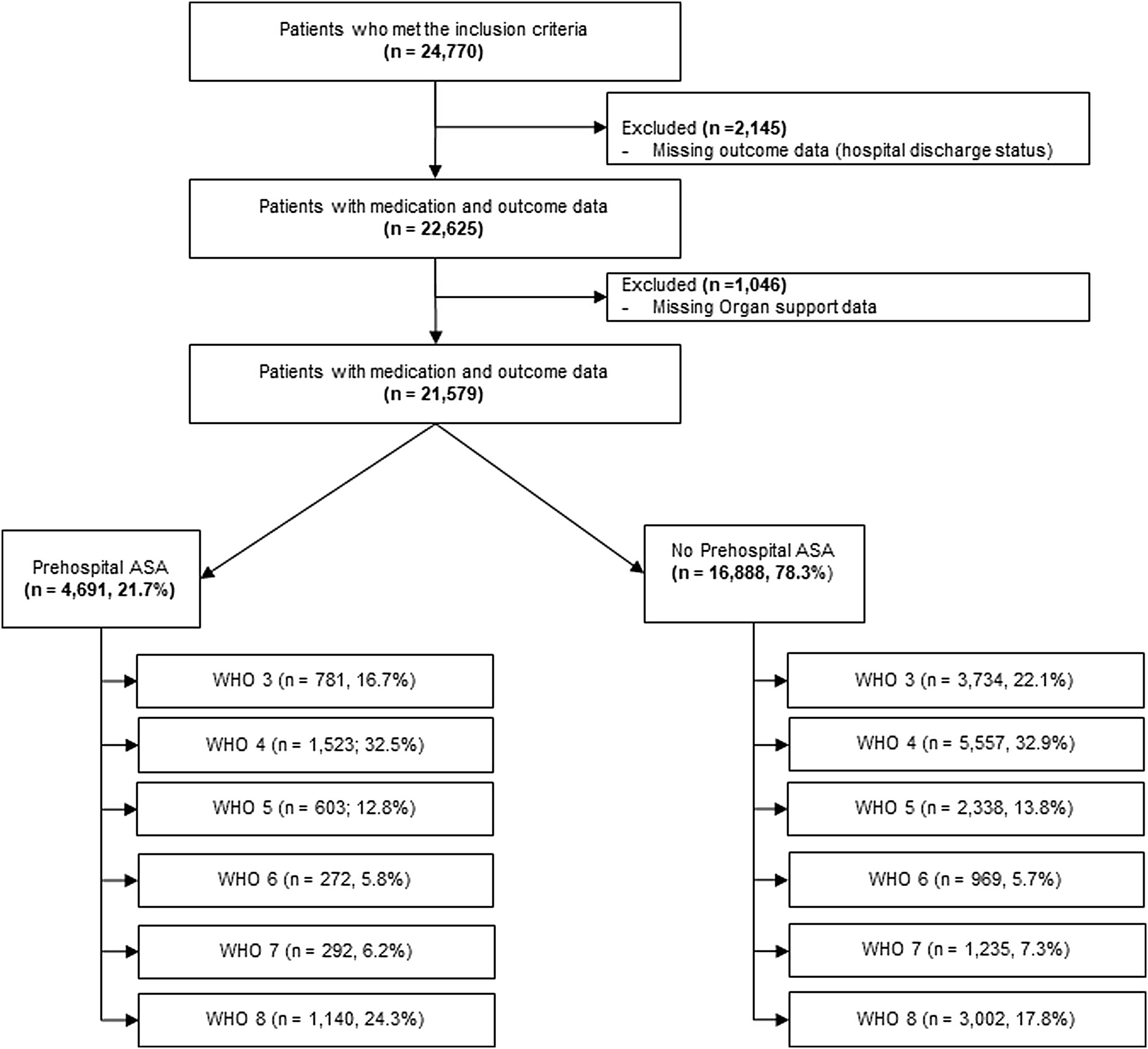
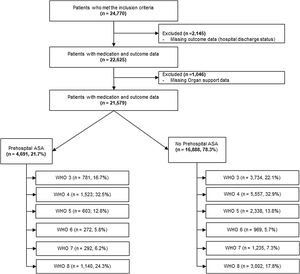
ResultsTwenty-one thousand five hundred and seventy-nine patients met eligibility criteria (described under methods section) and were included in the final analysis (Fig. 1). Of these, 4691 (21.7%) received pre-hospital ASA. Compared to those not receiving ASA, patients in the pre-hospital ASA group were older (median [IQR] 70 [61, 79] vs. 59 [46, 71] years, p<0.01), had higher proportion of males (58.7% vs. 56.0%, p≤0.01 for sex) and were more likely to be of White race (57.4% vs. 51.6%, p<0.01 for race; Table 1). Patients receiving ASA prior to hospitalization also had a higher prevalence of CAD (32.2% vs. 7.1%, p<0.01), HTN (78.2% vs. 48.7%, p<0.01), CHF (19.2% vs. 6.8%, p<0.01), CKD (26.2% vs. 10.5%, p<0.01), DM (51.9% vs. 30.8%, p<0.01), history of stroke (15.9% vs. 6.0%, p<0.01), obesity (21.5% vs. 17.5%, p<0.01), history of VTE (5.5% vs. 3.5%, p<0.01) and chronic pulmonary disorders (22.7% vs. 15.2%, p<0.01).
Demographics and baseline characteristics.
| Demographics by aspirin use | p value | |||
|---|---|---|---|---|
| Total | No (N=16,888) | Yes (N=4691) | Total (N=21,579) | |
| Age (years) | <0.01 | |||
| Missing | 41 | |||
| N | 16,853 | 4685 | 21,538 | |
| Mean (SD) | 58.3 (17.6) | 69.3 (13.3) | 60.7 (17.3) | |
| Median (Q1, Q3) | 59.0 (46, 71) | 70.0 (61, 79) | 62.0 (49, 73) | |
| Sex | ||||
| Missing | 3 | <0.01 | ||
| Male | 9461 (56.0%) | 2756 (58.7%) | 12,217 (56.6%) | |
| Female | 7417 (43.9%) | 1934 (41.2%) | 9351 (43.3%) | |
| Intersex | 5 (0.03%) | 1 (0.02%) | 6 (0.03%) | |
| Transgender | 2 (0.01%) | 0 (0.0%) | 2 (0.01%) | |
| Race | <0.01 | |||
| Missing | 2963 | |||
| White | 7480 (51.6%) | 2361 (57.4%) | 9841 (52.9%) | |
| African American | 3366 (23.2%) | 1223 (29.7%) | 4589 (24.6%) | |
| Other | 3656 (25.2%) | 530 (12.9%) | 4186 (22.5%) | |
| Ethnic group | <0.01 | |||
| Missing | 177 | |||
| Unknown | 1156 (6.9%) | 240 (5.2%) | 1396 (6.5%) | |
| Hispanic | 3449 (20.6%) | 736 (15.9%) | 4185 (19.5%) | |
| Non-Hispanic | 10,619 (63.4%) | 3442 (74.2%) | 14,061 (65.7%) | |
| Not applicable | 1537 (9.2%) | 223 (4.8%) | 1760 (8.2%) | |
| Time since start of COVID | <0.01 | |||
| Missing | 7116 | |||
| Group 1: Jan–Mar 2020 | 596 (5.2%) | 115 (3.7%) | 711 (5.3%) | |
| Group 2: Apr–Jun 2020 | 3085 (27.0%) | 791 (26.0%) | 3876 (26.8%) | |
| Group 3: Jul–Sep 2020 | 3673 (32.2%) | 868 (28.5%) | 4541 (31.4%) | |
| Group 4: Oct–Dec 2020 | 2864 (25.1%) | 920 (30.2%) | 3784 (26.2%) | |
| Group 5: Jan 2021 onwards | 1200 (10.5%) | 351 (11.5%) | 1551 (10.7%) | |
| Comorbidities by aspirin usea | p value | |||
|---|---|---|---|---|
| No (N=16,888) | Yes (N=4,691) | Total (N=21,579) | ||
| Coronary artery disease, %y | 1194 (7.1%) | 1513 (32.2%) | 2707 (12.5%) | <0.01 |
| Hypertension, %y | 8231 (48.7%) | 3667 (78.2%) | 11,898 (55.1%) | <0.01 |
| Congestive heart failure, %y | 1148 (6.8%) | 901 (19.2%) | 2049 (9.5%) | <0.01 |
| CKD, %y | 1779 (10.5%) | 1230 (26.2%) | 3009 (13.9%) | <0.01 |
| Diabetes mellitus, %y | 5198 (30.8%) | 2435 (51.9%) | 7633 (35.4%) | <0.01 |
| Stroke, %y | 1007 (6.0%) | 746 (15.9%) | 1753 (8.1%) | <0.01 |
| Obesity, %y | 2959 (17.5%) | 1010 (21.5%) | 3969 (18.4%) | <0.01 |
| Venous thromboembolism – DVT/PE | 598 (3.5%) | 257 (5.5%) | 855 (4.0%) | <0.01 |
| Chronic pulmonary disorder (COPD+asthma+PCD) | 2567 (15.2%) | 1067 (22.7%) | 3634 (16.8%) | <0.01 |
In univariate analyses (Table 2), patients receiving pre-hospital ASA had higher hospital mortality (24.3% vs. 17.8%, p<0.01), ICU mortality (44% vs. 32%, p<0.01), longer hospital LOS (7.8 [4.1, 14.3] vs. 7.0 [4, 14] days, p=0.10) and higher rates of progression to severe disease or death [WHO 6 or higher] (36% vs. 31%, p<0.01). Patients in ASA group had significantly lower rates of ICU admission (41% vs. 44%, p<0.01) (Table 2). In unadjusted proportional hazards modeling (Table 3), patients in the ASA group had higher risk of overall hospital mortality (HR 1.48, 95% CI: 1.37–1.60, p<0.01) and higher hazard of severe disease progression or death (HR 1.28, 95% CI 1.20–1.37, p<0.01). The ASA group had lower hazard for receiving ICU levels of care (HR 0.85, 95% CI: 0.80–0.91, p<0.01). With regards to hospital complications, the ASA group had higher hazard for CVA (HR 1.40, 95% CI: 1.06–1.86, p<0.02), gastrointestinal (GI) hemorrhage (HR 1.65, 95% CI: 1.25–2.18, p<0.01), cardiac arrest (HR 1.58, 95% CI: 1.39–1.81, p<0.01) and myocardial infarction (HR 1.95, 95% CI; 1.70–2.25). Patients in the ASA group were also found to have higher risk of needing packed red blood cell (pRBC) transfusion (HR 1.29, 95% CI: 1.03–1.61, p=0.03).
Unadjusted outcomes between ASA and no ASA groups.
| LOS and mortality outcomes, by aspirin use and overall | No (N=16,888) | Yes (N=4691) | Total (N=21,579) | p value |
|---|---|---|---|---|
| Disease severity, N (%) | ||||
| Hospitalized but did not receive any oxygen support (WHO 3) | 3734 (22.1%) | 781 (16.7%) | 4515 (20.9%) | <0.01 |
| Oxygen by face mask or nasal prongs (WHO 4) | 5557 (32.9%) | 1523 (32.5%) | 7080 (32.8%) | |
| Non-invasive ventilation or high-flow oxygen (WHO 5) | 2338 (13.8%) | 603 (12.8%) | 2941 (13.6%) | |
| Intubation and mechanical ventilation (WHO 6) | 969 (5.7%) | 272 (5.8%) | 1241 (5.7%) | |
| Ventilation and additional organ support (pressors, RRT, or ECMO) (WHO 7) | 1235 (7.3%) | 292 (6.2%) | 1527 (7.1%) | |
| Death (WHO 8) | 3002 (17.8%) | 1140 (24.3%) | 4142 (19.2%) | <0.01 |
| Progression to severe disease (IMV) or death | ||||
| N (%) | 5206 (30.8%) | 1704 (36.3%) | 6910 (32%) | <0.01 |
| IMV free days | ||||
| Median [IQR] | 18.0 [9, 24] | 18.7 [10.7, 24.0] | 0.38 | |
| Mean (SD) | 16.1 (9.3) | 16.6 (8.8) | ||
| Need for ICU admission | ||||
| N (%) | 7289 (44.5%) | 1862 (40.6%) | 9151 (43.6) | <0.01 |
| ICU-free days (out of 28 days) | ||||
| Median [IQR] | 15.0 [0.0, 23] | 0.0 [0.0, 23.0] | <0.01 | |
| Mean (SD) | 12.3 (11.1) | 10.1 (11.4) | ||
| Hospital-free days (out of 28) | ||||
| Median [IQR] | 19.2 [3, 23.4] | 18.3 [0, 23.1] | <0.01 | |
| Mean (SD) | 15.3 (9.9) | 14.1 (10.3) | ||
| Hospital length of stay (LOS) | ||||
| Median [IQR] | 7.0 [4,14] | 7.8 [4.1, 14.3] | 0.10 | |
| Mean (SD) | 11.6 (13.5) | 11.5 (13.1) | ||
| ICU mortality | ||||
| Died | 2455 (32.3%) | 882 (44.0%) | 3337 (34.7%) | <0.01 |
| Hospital mortality | ||||
| Died | 3002 (17.8%) | 1140 (24.3%) | 4142 (18.3%) | <0.01 |
Adjust outcomes between ASA and no ASA groups.
| Unadjusted | Adjusted | |||
|---|---|---|---|---|
| Outcome variables | HR (95% CI) | p | HR (95% CI) | p |
| Disease severity progression | ||||
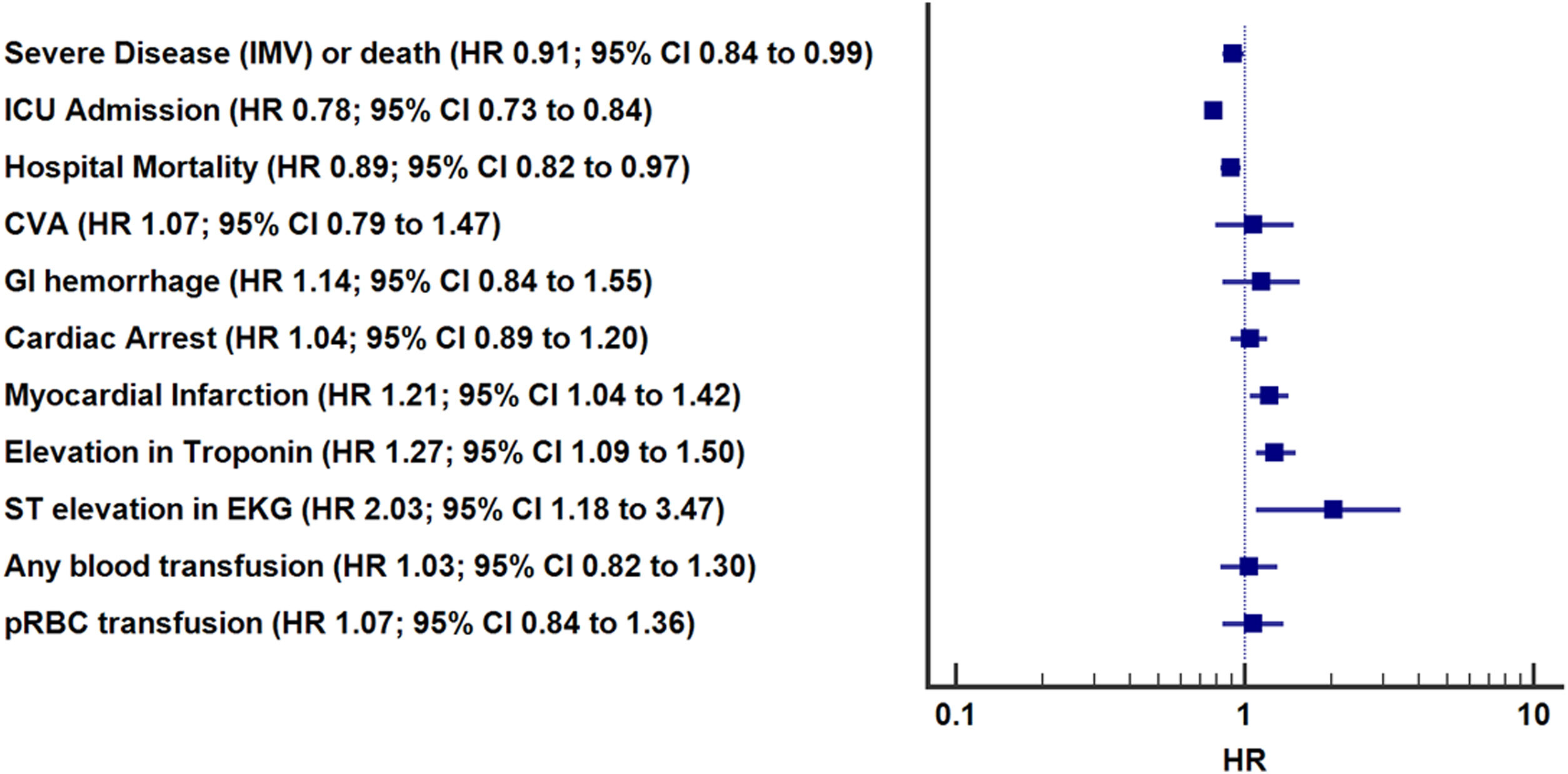
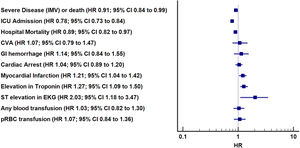
| Severe disease (IMV) or deatha | 1.28 (1.20–1.37) | <0.01 | 0.91 (0.84–0.99) | 0.02 |
| Outcome | ||||
| ICU admission | 0.85 (0.80–0.91) | <0.01 | 0.78 (0.73–0.84) | <0.01 |
| Hospital mortality | 1.48 (1.37–1.60) | <0.01 | 0.89 (0.82–0.97) | 0.01 |
| Complications | ||||
| CVA | 1.40 (1.06–1.86) | 0.02 | 1.07 (0.79–1.47) | 0.65 |
| GI hemorrhage | 1.65 (1.25–2.18) | <0.01 | 1.14 (0.84–1.55) | 0.40 |
| Myocardial infarctionb | 1.95 (1.70–2.25) | <0.01 | 1.21 (1.04–1.42) | 0.02 |
| Elevation in troponin | 1.93 (1.67–2.24) | <0.01 | 1.27 (1.09–1.50) | 0.03 |
| ST elevation in EKG | 2.56 (1.59–4.12) | <0.01 | 2.03 (1.18–3.47) | <0.01 |
| Need for transfusionc | ||||
| Any blood product transfusion (first 24h) | 1.21 (0.98–1.51) | 0.07 | 1.03 (0.82–1.30) | 0.79 |
| pRBC transfusion | 1.29 (1.03–1.61) | 0.03 | 1.07 (0.84–1.36) | 0.59 |
| Length of stay measures (not HR but days [95% CI]) | ||||
| Hospital-free days | −1.14 (−1.46 to −0.81) | <0.01 | 1.00 (0.66–1.35) | <0.01 |
| ICU-free days | −2.19 (−2.76 to −1.63) | <0.01 | 0.33 (−0.27 to 0.93) | 0.29 |
| IMV-free days | 0.50 (−0.13 to 1.13) | 0.12 | −0.27 (−0.96 to 0.40) | 0.42 |
Adjusted models were adjusted for age, sex, ethnicity, race, and comorbidities of CAD, HTN, CHF, stroke, chronic pulmonary diseases (COPD, asthma, pulmonary circulatory diseases), history of VTE, diabetes mellitus, chronic kidney disease and obesity.
In adjusted analyses (Table 3, Fig. 2), the ASA group had significantly lower hazard of overall hospital mortality (HR 0.89, 95% CI: 0.82–0.97, p=0.01), progression to severe disease or death (HR 0.91, 95% CI: 0.84–0.99, p=0.02), and receiving ICU levels of care (HR 0.78, 95% CI: 0.73–0.84, p<0.01). Differences in incident CVA, GI hemorrhage, cardiac arrest, or need for pRBC transfusion were not significant. Patients receiving ASA had higher in-hospital myocardial infarction (HR 1.21, 95% CI: 1.04–1.42, p=0.02). Pre-hospital ASA use was associated with 1.0 more hospital free days (95% CI: 0.66–1.35, p≤0.01) but there was no difference in IMV free days (−0.27, 95% CI: −0.96 to 0.40, p value=0.42) or ICU free days (0.33, 95% CI: −0.27 to 0.93, p value=0.41) (Table 3).
We also performed a sensitivity analysis after adjusting for the time since pandemic (divided as the quarters of 2020 and 2021) variable added to our original model to assess if there was a difference in the outcomes since there were preventative measures (vaccination) and some therapeutics (steroids, monoclonal antibodies) introduced as the pandemic evolved (Supplementary Table). The overall direction and significance of the results remained the same. In this sensitivity analysis, the pre-hospital ASA group had lower risk of hospital mortality (HR 0.89, 95% CI: 0.81–0.97, p value ≤0.01), progression to severe disease or death (HR 0.92, 95% CI: 0.85–0.99, p=0.04) and lower risk for needing ICU level of care (HR 0.76, 95% CI: 0.71–0.82, p<0.01). Pre-hospital ASA group continued to demonstrate a higher risk of developing in hospital MI (HR 1.23, 95% CI: 1.05–1.44, p≤0.01).
DiscussionIn this observational international study of more than 21,000 hospitalized adults with COVID-19 enrolled in the VIRUS: COVID-19 registry, pre-hospital ASA use was associated with reduced hospital mortality and progression to severe diseases or death (level 6 or above on WHO ordinal disease severity scale). These results suggest that pre-hospital use of ASA may have beneficial downstream effects during hospitalization. However, these associations should be interpreted as hypothesis-generating rather than causal in nature. Indeed, many factors may influence outcomes in patients with COVID-19 disease,17 and adverse events including death may be related to disease progression, complications of hospitalization, pre-existing patient frailty, and underlying medical comorbidities (with COVID-19 disease as an epiphenomenon). Nevertheless, aspirin therapy prior to hospital admission in this large and diverse patient cohort was associated with improvement in hospital outcomes. Differentiation between these pathways is important to interpret the results from observational data and devising strategies for improved outcome. In our study, we have attempted to control for multiple medical comorbidities that are independently linked to increased mortality and worse outcomes in COVID-19.13,18–27
COVID-19 associated ARDS has been associated with hypercoagulability and increased incidence of pulmonary microthrombosis,28,29 with platelets likely playing a key role.30–32 Preclinical data has suggested that antiplatelet agents could be a modulating factor for ARDS development,30,33 however randomized control trials have not shown protective effects of ASA therapy for prevention of non-COVID-19-related ARDS34–36. Similarly, a recent randomized control trial by the RECOVERY collaborative group did not show any benefit with regards to mortality or progression to severe disease (i.e. risk of progressing to IMV or death) in patients with COVID-19 randomized to ASA during hospitalization, although those receiving ASA had shorter hospital durations by approximately 1 day.10 It is important to note that patients in this trial were exposed to aspirin (150mg daily) only after being hospitalized, while patients in our study were receiving ASA therapy prior to admission, likely prior SARS-CoV-2 infection, and were therefore likely to have greater cumulative exposure. Additionally, patients in the RECOVERY study were approximately 10 years younger (RECOVERY 59 yrs. vs. VIRUS 69 yrs.) and with less comorbid illness burden (any comorbidities RECOVERY 43% vs. any comorbidity VIRUS 86%) when compared to those receiving ASA in our study, which may affect generalizability of study findings.
Importantly, the findings from our study are consistent with other observational studies assessing the relationships between pre-hospitalization ASA therapy and COVID-19 outcomes.11,12,37,38 These findings were also confirmed through recent meta-analyses on the use of ASA in COVID-19 patients.39,40 We hypothesize that pre-hospital ASA therapy could potentially be associated with improved hospital outcomes secondary to systemic antithrombotic effects which could potentially reduce pulmonary microthromboses, inflammation, and larger thromboembolic events. It is possible that ASA initiation during hospitalization, after the inflammatory state of COVID-19 has already been established, may not provide sufficient time to influence clinical outcomes. Future randomized trials evaluating the safety and efficacy of pre-hospital ASA therapy are clearly warranted in those at high-risk for COVID-19 pneumonia.
This study is notable as one of the largest studies from an international registry of hospitalized patients with COVID-19. However, there are limitations. First, data missingness, particularly with regards to pre-hospital medications, resulted in the exclusion of a significant number of patients. Second, we were unable to capture the dose of pre-hospital ASA therapy, and we cannot draw any conclusions with regards to potential dose-outcome relationships. Additionally, we cannot comment on the duration or indication of ASA therapy prior to hospitalization. Third, we were unable to assess the continuation of ASA use during hospitalization, as this was not documented through the registry. In conjunction this, the interaction of other medication such as use of statins, immunosuppression in cancer patients was not captured as part of this study. Prehospital use of antibiotics in patients with comorbidities such as chronic pulmonary diseases was not captured. However, our final model was adjusted for chronic pulmonary diseases to avoid any bias on the final outcomes. Fourthly, as a part of the design of the registry, we did not include the non-hospitalized patients. This could give rise of “healthy user bias”, which is a legitimate concern in observational studies. Therefore, these findings warrant a confirmation in a prospective experimental design. Finally, we were unable to adjust for variation in outcomes based on the availability of other therapeutic interventions (e.g., use of steroids, tocilizumab, etc.) and preventative measures (availability of vaccines during the second half of the study period).
ConclusionPre-hospital ASA use was associated with improved overall hospital mortality, lower risk of severe disease (disease progression to needing IMV or death), reduced need for ICU level of cares, and more hospital free days in hospitalized patients with COVID-19 pneumonia. Although these associations should be interpreted cautiously, well-designed clinical trials of pre-hospital ASA utilization may be warranted.
Author contributionsA.L, J.P.D.G, A.K.K, M.A.W. O.G prepared the main manuscript text A.L, V.B, A.T, S.Z prepared the data files and tables A.L, J.P.D.G, A.K.K, M.A.W, O.G, R.K performed data analysis A.L, V.K, K.B, prepared the figures and supplementary material V.K, K.B, A.W, R.K provided administrative oversight A.L, V.B, A.T, S.Z, A.K.K, M.A.W, A.B.C, R.C.C, V.M.R, D.L.A, S.R.C, U.R, Y.T, J.L.D, N.D provided help with data collection and data quality control J.P.D.G, A.K.K, M.A.W, R.K, A.W, O.G provided critical review and expert advice All authors reviewed and approved the final version of the manuscript.
FundingThis publication was supported by NIH/NCRR/NCATS CTSA Grant Number UL1 TR002377. Its contents are solely the responsibility of the authors and do not necessarily represent the official views of the NIH. The registry is funded in part by the Gordon and Betty Moore Foundation, and Janssen Research & Development, LLC. They had no influence on the analysis, interpretation, and reporting of pooled data.
Conflict of interestAmos Lal: Supported by the National Center for Advancing Translational Sciences, No. UL1 TR002377.
Juan Pablo Domecq Garces: None.
Vikas Bansal: None.
Aysun Tekin: None.
Ashish K. Khanna: Research Grants from Edwards Lifesciences, DoD, NIH/BARDA, Retia Medical, Caretaker Medical, Trevena Pharmaceuticals, Masimo Corporation, Potrero Medical. Consulting Fees from Edwards Lifesciences, Medtronic, Philips North America, GE Healthcare, La Jolla Pharma, Retia Medical. Payment or Honoraria from Edwards Lifesciences, Medtronic, GE Healthcare, Merck. Leadership role Board of Directors SOCCA, Program Chair SCCM 2023, Vice Chair ASA Committee on CCM, SCCM Discovery Oversight Committee, Research Section Chair SCCM.
Matthew A. Warner: K23 award from National Heart, Lung, and Blood Institute.
Amy B. Christie: Navicent Health Foundation Grant-$70,800. This grant helped us obtain personnel support for data entry in the SCCM VIRUS registry project.
Rodrigo Cartin-Ceba: None.
Valerie M. Ranner-Goodspeed: None.
Donna Lee Armaignac: None.
Sreekanth R. Cheruku: SCCM VIRUS Data Automation Grant.
Umamaheswara Raju: None.
Yasir Tarabichi: Consulting Fees by Beckman Coulter To institution for research activities and personally for general consulting – both unrelated to this work.
Joshua L. Denson: Society of Critical Care Medicine – Research Grant; American Diabetes Association – Research Grant; U54 GM104940, NIH, Louisiana Clinical & Translational Science Center (LA CaTS) Roadmap Scholar Research Grant. Consulting Fees from Guidepoint Global Consulting. Payment or honoraria for lectures, presentations, speakers bureaus, manuscript writing or educational events from Astrazeneca. Participation on a Data Safety Monitoring Board or Advisory Board: ACTIV 6 COVID-19 Trial Advisory Committee.
Vishakha Kumar: Gordon and Betty Moore Foundation; Janssen and Janssen LLC. Both supporting Grant to institution – Society of Critical Care Medicine.
Allan J. Walkey: Gordon and Betty Moore Foundation.
Karen Boman: Gordon and Betty Moore Foundation; Janssen and Janssen LLC- none of them have any influence on data collection, interpretation, analysis or writing.
Neha Deo: None.
Rahul Kashyap: Gordon and Betty Moore Foundation; Janssen and Janssen LLC- none of them have any influence on data collection, interpretation, analysis or writing; Principal Investigator for SCCM Discovery VIRUS: COVID-19 Registry and core committee member.
Ognjen Gajic: None.
AL and OG had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
The study was performed in collaboration with or on behalf of The Society of Critical Care Medicine Discovery Viral Infection and Respiratory Illness Universal Study (VIRUS): COVID-19 Registry Investigator Group.
Atrium Health Navicent: Amy B. Christie, Dennis W. Ashley, Rajani Adiga
Mayo Clinic Arizona: Rodrigo Cartin-Ceba, Ayan Sen, Fahimeh Talaei
King Faisal Specialist Hospital & Research Centre – Riyadh: Marwa Ridha Amer, Mohammed Abdullah Bawazeer, Talal I. Dahhan, Eiad Kseibi, Abid Shahzad Butt, Syed Moazzum Khurshid, Muath Rabee, Mohammed Abujazar, Razan Alghunaim, Maal Abualkhair, Abeer Turki AlFirm, Eiad Kseibi, Syed Moazzum Khurshid, Muath Rabee, Mohammed Abujazar, Razan Alghunaim
Baptist Health South Florida: Donna Lee Armaignac, Don Parris, Maria Pilar Zuniga, Ilea Vargas, Viviana Boronat, Anneka Hutton, Navneet Kaur, Prashank Neupane, Nohemi Sadule-Rios, Lourdes M. Rojas, Aashish Neupane, Priscilla Rivera, Carlos Valle Carlos, Gregory Vincent
Wake Forest University School of Medicine; Wake Forest Baptist Health Network: Ashish K. Khanna, Lynne Harris, Bruce Cusson, Jacob Fowler, David Vaneenenaam, Brandon Reeves, Imoh Udoh, Kathleen Johnson, Jessica Fanelli, Nataya Disher, Anusha Samant, Christian DeGroot, Evan Youshock, R. Miller Ligon, Katherine McCartney, Julio Garcia, Chidi Ilobachie, Kelsey Flores, Lauren Sands, Samuel Robinson, Nia Sweatt, Madeline Fram, Easton Howard, Lilian Nosow, Lucy Winesett Howard, Quan Duc Minh Pham, Aman Irfanullah, Tiffany Ong, Chukwunyelu Henry Enwezor, Hannah Dabagian, Rafael Mendoza
Clements University Hospital at UT Southwestern Medical Center: Sreekanth Cheruku, Farzin Ahmed, Christopher Deonarine, Ashley Jones, Mohammad-Ali Shaikh, David Preston, Jeanette Chin
Gandhi Medical College and Hospital, Hyderabad: Umamaheswara Raju, Janaki Manduva, Naresh Kolakani, Shreeja Sripathi, Sheetal Chaitanya
MetroHealth Medical Center: Yasir Tarabichi, Adam Perzynski, Christine Wang, Dhatri Kotekal
Tulane University Medical Center and University Medical Center New Orleans: Joshua L. Denson, A. Scott Gillet, Margo Brown, Rachael Stevens, Andrew Wetherbie, Kevin Tea, Mathew Moore
Medical College of Wisconsin: Rahul S Nanchal, Paul A Bergl, Jennifer L Peterson
Valleywise Health (formerly Maricopa Medical Center): Murtaza Akhter, Rania Abdul Rahman, Mary Mulrow
Marmara University: Uluhan Sili, Huseyin Bilgin, Pinar Ay
Kuban State Medical University with affiliation Territorial Hospital #2: Igor Borisovich Zabolotskikh, Tatiana Sergeevna Musaeva
St. Joseph Mercy Ann Arbor, Ann Arbor: Harry L. Anderson, III, Dixy Rajkumar, Ali Abunayla, Jerrilyn Heiter
Mayo Clinic, Florida: Devang Sanghavi, Pramod Guru, Pablo Moreno Franco, Karthik Gnanapandithan, Hollie Saunders, Zachary Fleissner, Juan Garcia, Alejandra Yu Lee Mateus, Siva Naga Yarrarapu, Nirmaljot Kaur, Abhisekh Giri, Mohammed Mustafa Hasan, Ashrita Donepudi
Mayo Clinic Rochester: Rahul Kashyap, Juan Pablo Domecq, Ognjen Gajic, Vikas Bansal, Aysun Tekin, Amos Lal, John C. O’Horo, Neha N. Deo, Mayank Sharma, Shahraz Qamar, Romil Singh, Diana J. Valencia Morales
Hospital Universitario La Paz: Santiago Y. Teruel, Belen C. Martin
BSES MG Hospital, Mumbai: Girish Vadgaonkar, Rekha Ediga, Shilpa Basety, Shwetha Dammareddy, Phani Sreeharsha Kasumalla
University of Cincinnati: Dina Gomaa B.S., Michael Goodman, Devin Wakefield, Anthony Spuzzillo, John O. Shinn II
Panimalar Medical College Hospital & Research Institute: Surapaneni Krishna Mohan, Ekambaram Jyothisree
Bangalore Medical College and Research Institute: Smitha S. Segu, Tuhin Chakraborty, Epcebha Joyce