Nuclear protein in testis (NUT) midline carcinoma (NMC) is an exceedingly rare malignant tumor with poorly epithelial differentiation which mostly affected children and young adults. It has several synonyms: t(15;19) carcinoma, aggressive t(15;19)-positive carcinoma, midline lethal carcinoma, midline carcinoma with NUT rearrangement, and BET-rearranged (bromodomain and extra-terminal) carcinoma.1 The most common location is head and neck region, thorax, and mediastinum. The highly aggressive clinical course, unique pathological feature and exclusive genetic alteration with the rearrangement of NUT gene makes it different from other tumors. Herein, we report a case of NMC.
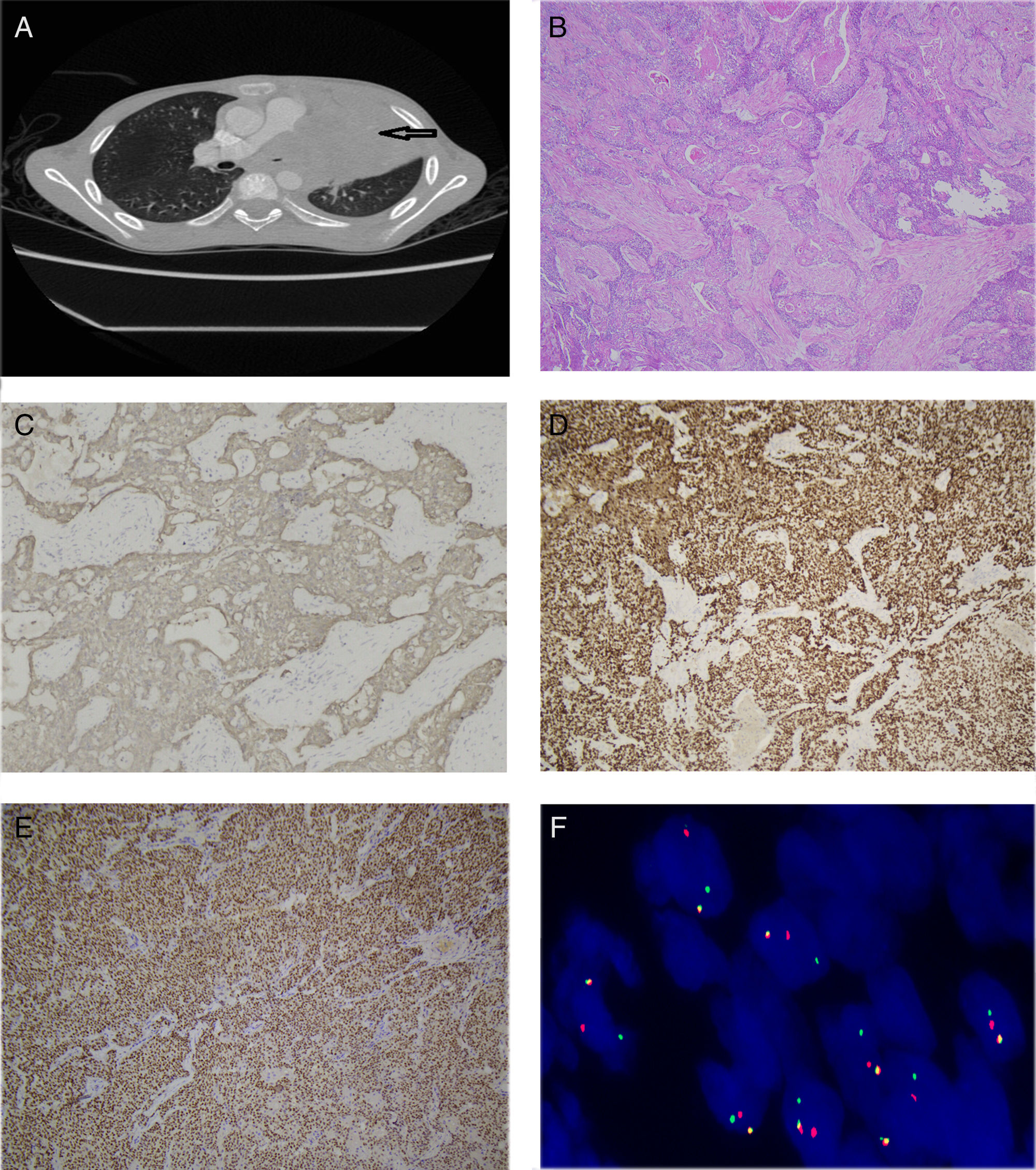
A ten-year-old boy presented to our hospital with the complaints of fever and coughing for more than one month. The child was diagnosed with pneumonia and administered antibiotic therapy for fifteen days in local hospital. The treatment protocol did not have any effect. CT scan revealed a giant mass measuring 10×10×8cm with uneven density in the left superior mediastinum which was heterogeneously enhanced by iv contrast (Fig. 1A). The mass wrapped the aorta and the left pulmonary artery and vein. The left lung was compressed. Multiple bone metastases were demonstrated by the X-ray and bone scan. Radical resection was unachievable and tumor biopsy was done. Morphological examination showed nests of primitive small round cells with islands of squamous epithelium and keratinization in the center suggesting a poorly differentiated carcinoma (Fig. 1B). Obvious necrosis and the lung invasion could also be noticed in the tumor. Pan-cytokeratin, p63, and NUT diffuse positivity on immunohistochemistry (Fig. 1C–E) and the break-apart positivity of the NUT probe by FISH (Fig. 1F) confirmed the final diagnosis of NMC. The child passed away one month after biopsy for the rapid procedure and also the lack of valid treatment strategies. Written informed consent was got from the child's parent.
(A) CT scan showed a giant mass in the left superior mediastinum (arrow). (B) Microscopic findings revealed nests of small round cells with scattered squamous epithelial differentiation in the center (×50). (C–E) Pancytokeratin (×100), p63 (×100), NUT (×50) were diffusely positive. (F) The NUT probe detected the break-apart positivity by FISH.
Midline carcinoma was first reported in 1991 with the nomenclature of carcinoma with a translocation t(15;19).2 French discovered the genetic feature with the presence of NUT gene rearrangement in 2003 and designated it as NMC in 2008.3,4 NMC has a wide age range from infancy to 78 years according to the literatures.1 More than one hundred cases have been reported including about 40 pediatric cases.4–9 Most of the pediatric cases were above ten years old and the majority of them developed metastasis at diagnosis or in a short time after diagnosis and the most common metastasis site was bone and lymph node. The main complaint was a mass in the midline area with accompanying symptoms such as dyspnea and chest pain. The nosogenesis of NMC was still unknown. BRD–NUT oncoprotein was suggested to block the epithelial differentiation and promote the growth of tumor cells and thus derived the carcinogenesis.10 NMC depends on the pathological diagnosis. Typical morphological features include nests and sheets of undifferentiated small round cells with or without focal or prominent squamous differentiation in the core of the nests and fibrous cords in the mesenchymal stroma. Pan-cytokeratin, p63 and NUT diffuse positivity is very helpful on immunohistochemistry. Fluorescence in situ hybridization (FISH) can detect the BRD–NUT fusion which confirms the diagnosis. The genetic alteration include BRD4–NUT (70%), BRD3–NUT (6%), NSD3–NUT (only 6 cases reported), NUT-variant (the rest).1,11 Two unusual cases of 3-way chromosome translocations had also been reported.5,12 Unique immunohistochemical markers and genetic translocation make the diagnosis of NMC easier. FISH and monoclonal antibody for NUT have a high sensitivity and specificity and are recommended for the diagnosis of NMC. They should be considered in every poorly differentiated carcinoma in the midline structures. The prognosis is depressing with a median survival of 6.7 months.13 No treatment guideline has been established and the impossible radical resection in most cases and the lack of effective anticancer drugs make the tumor refractory to the treatment. Chemotherapy does not have a good response although combination chemotherapies were observed to have objective response to NMC to some extent in few cases.7 Luckily, together with the progress of target therapy in tumors with specific genetic alterations, small molecular inhibitors such as BET inhibitors and histone deacetylase inhibitors have improved the outcome and were still in the early clinical trials.14 What should be altered is that target therapy with BET inhibitors could change the cytopathologic and immunohistochemical features of the tumor cells and be deceivable in the estimation of tumor recurrence.15
In summary, we present an unusual case of NMC in a Chinese boy. NMC should be considered in the differential diagnosis of any undifferentiated carcinoma. The rapidly exacerbated course without effective therapy makes the prognosis dismal. The establishment of The International NUT Midline Carcinoma Registry in 2010 promoted the international cooperation and the clinical trial of target therapy was conducted which may bring the light of hope to this kind of patients.