Tuberculosis (TB) remains a serious public health problem, and about one-third of world's population has active or latent TB. In Europe, there are 49 new cases and 7 deaths from TB every hour.1 In Portugal, the incidence has been decreasing in recent years, and in 2014 the annual incidence was 20/1,00,000.2
Drug-resistant Mycobacterium tuberculosis has become a major threat to the control of TB and, among all first-line drugs, resistance is greatest to isoniazid (INH).3,4 In Portugal, INH resistance was 10.5% in 2014 among TB cases in whom susceptibility testing was performed.2 In fact, there has been an increasing resistance to INH, despite the decreasing number of TB cases.2
INH is a first-line anti-TB drug because of its potent early bactericidal activity against rapidly dividing cells.3,5 However, treatment of active TB requires multiple anti-TB drugs along with INH to prevent selection and emergence of a drug-resistant population of M. tuberculosis. According to current World Health Organization (WHO) recommendations, INH mono-resistant TB should be treated with 6–9 months of rifampicin, ethambutol, and pyrazinamide, plus or minus a fluoroquinolone.6 These are also the current treatment guidelines in Portugal.
TB is a notifiable disease in Portugal, so clinicians report all cases to National-Tuberculosis-Surveillance-System (SVIG-TB) that has data on patient demographics, comorbidities, risk behaviors, and clinical, radiological, and microbiological information, as well as treatment outcomes.7
The objectives of this study were identify factors associated with INH mono-resistance, compare treatment outcomes of INH mono-resistant patients with drug-susceptible patients and understand the causes of unsuccessful treatment among INH mono-resistance TB cases.
To achieve the goals, data from Portuguese SVIG-TB were retrospectively analyzed from 1/January/2008 to 31/December/2014. INH mono-resistant cases were compared with drug-susceptible cases.
Culture-confirmed cases tested against first-line anti-TB drugs were included. INH mono-resistant TB cases were defined as having resistance to INH, but susceptibility to all other first-line anti-TB drugs. Drug-susceptible cases were those that had documented sensitivity to INH, rifampin, pyrazinamide, and ethambutol.
Susceptibility testing was carried out according to international standards; the method of proportions (liquid medium in the MGIT 960 system) was used to determine susceptibility to anti-tuberculosis drugs on: isoniazid (0.1μg/ml), rifampicin (1μg/ml), ethambutol (5μg/ml), pyrazinamide (100μg/ml) and streptomycin (1μg/ml).
The WHO standard definitions were used for treatment outcomes.6 Unsuccessful treatment includes failure, death during treatment and default. Cured patients and those with completed treatment were defined as treatment success. Cases with unknown outcomes (“in treatment” and “transferred”) were excluded.
Data were summarized by descriptive statistics, consisting of absolute (relative) frequencies or median (minimum–maximum), according to nature of variables.
Comparisons of demographic and clinical variables between two groups used the Chi-squared test (or Fisher's test, as appropriate) for categorical variables and the Mann–Whitney U-test for continuous variables. A univariate analysis evaluated the effect of demographic characteristics and risk factors on treatment outcome for INH mono-resistance TB, through simple logistic regression.
Statistical analyses were performed with SPSS version 18.0 (PASW Statistic 18). Significance level was set at 0.05.
This study used surveillance data, with no possibility of linking patient records to patient personal data, so ethical approval was considered unnecessary.
Between 2008 and 2014, 18,429 TB cases were reported to SVIG-TB, from them 12,031 had culture confirmation and 10,588 of them were tested for sensitivity to first-line anti-TB drugs. According to drug susceptibility test, 7103 cases were susceptible to all first-line anti-TB drugs and 242 cases were INH mono-resistant. 3243 cases were excluded: 657 cases had resistance to rifampin and/or pyrazinamide and/or ethambutol and/or streptomycin, and 2061 had susceptibility to isoniazid and rifampin and no results for pyrazinamide, ethambutol and streptomycin.
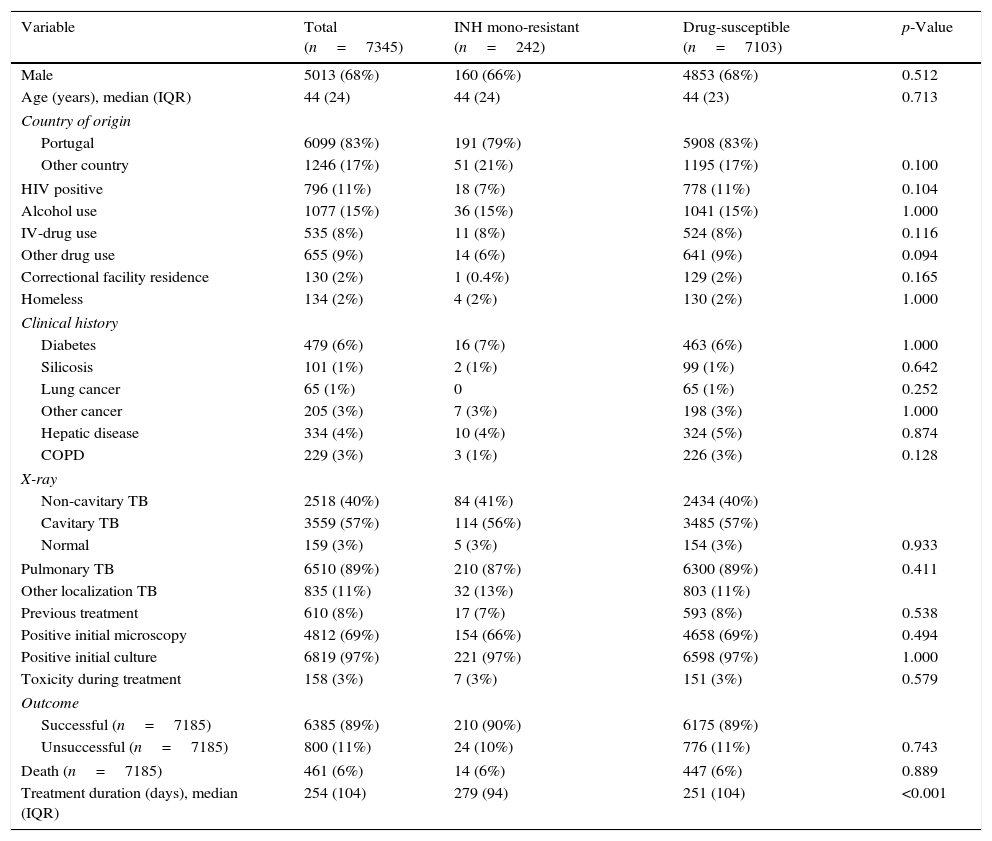
Table 1 shows the characteristics of INH mono-resistant, drug-susceptible, and all eligible TB cases. In all groups, median patient age was 44 years, men were more likely to be cases than women, and most patients were born in Portugal. Most of studied clinical factors were not statistically different between INH mono-resistant and drug-susceptible groups. Although the two groups had no significant difference in treatment outcome and toxicity during treatment, the median treatment duration was longer in INH mono-resistant group (279 vs. 251 days, p<0.001).
Demographic and clinical characteristics of enrolled TB patients.
| Variable | Total (n=7345) | INH mono-resistant (n=242) | Drug-susceptible (n=7103) | p-Value |
|---|---|---|---|---|
| Male | 5013 (68%) | 160 (66%) | 4853 (68%) | 0.512 |
| Age (years), median (IQR) | 44 (24) | 44 (24) | 44 (23) | 0.713 |
| Country of origin | ||||
| Portugal | 6099 (83%) | 191 (79%) | 5908 (83%) | |
| Other country | 1246 (17%) | 51 (21%) | 1195 (17%) | 0.100 |
| HIV positive | 796 (11%) | 18 (7%) | 778 (11%) | 0.104 |
| Alcohol use | 1077 (15%) | 36 (15%) | 1041 (15%) | 1.000 |
| IV-drug use | 535 (8%) | 11 (8%) | 524 (8%) | 0.116 |
| Other drug use | 655 (9%) | 14 (6%) | 641 (9%) | 0.094 |
| Correctional facility residence | 130 (2%) | 1 (0.4%) | 129 (2%) | 0.165 |
| Homeless | 134 (2%) | 4 (2%) | 130 (2%) | 1.000 |
| Clinical history | ||||
| Diabetes | 479 (6%) | 16 (7%) | 463 (6%) | 1.000 |
| Silicosis | 101 (1%) | 2 (1%) | 99 (1%) | 0.642 |
| Lung cancer | 65 (1%) | 0 | 65 (1%) | 0.252 |
| Other cancer | 205 (3%) | 7 (3%) | 198 (3%) | 1.000 |
| Hepatic disease | 334 (4%) | 10 (4%) | 324 (5%) | 0.874 |
| COPD | 229 (3%) | 3 (1%) | 226 (3%) | 0.128 |
| X-ray | ||||
| Non-cavitary TB | 2518 (40%) | 84 (41%) | 2434 (40%) | |
| Cavitary TB | 3559 (57%) | 114 (56%) | 3485 (57%) | |
| Normal | 159 (3%) | 5 (3%) | 154 (3%) | 0.933 |
| Pulmonary TB | 6510 (89%) | 210 (87%) | 6300 (89%) | 0.411 |
| Other localization TB | 835 (11%) | 32 (13%) | 803 (11%) | |
| Previous treatment | 610 (8%) | 17 (7%) | 593 (8%) | 0.538 |
| Positive initial microscopy | 4812 (69%) | 154 (66%) | 4658 (69%) | 0.494 |
| Positive initial culture | 6819 (97%) | 221 (97%) | 6598 (97%) | 1.000 |
| Toxicity during treatment | 158 (3%) | 7 (3%) | 151 (3%) | 0.579 |
| Outcome | ||||
| Successful (n=7185) | 6385 (89%) | 210 (90%) | 6175 (89%) | |
| Unsuccessful (n=7185) | 800 (11%) | 24 (10%) | 776 (11%) | 0.743 |
| Death (n=7185) | 461 (6%) | 14 (6%) | 447 (6%) | 0.889 |
| Treatment duration (days), median (IQR) | 254 (104) | 279 (94) | 251 (104) | <0.001 |
TB: tuberculosis; INH: isoniazid; IQR: interquartile range; HIV: human immunodeficiency virus; IV: intravenous; COPD: chronic obstructive pulmonary disease.
Several previous studies identified risk factors for INH mono-resistant TB, like previous TB treatment,5,8 younger age,9,10 foreign birth,10,11 and various social factors.11 However, as in Birmingham study,12 no predictive factors for INH mono-resistant TB were found.
We did find that INH mono-resistant cases had a longer mean treatment duration, in agreement with some previous studies.5,8 For example, a study in Georgia4 reported that the extended length of regimen may be associated with reduce adherence. A study in San Francisco5 suggested that new short-course treatment regimens are needed because of high incidence of drug toxicity. In contrast, a study in southern Mexico reported that treatment outcomes were similar for patients receiving a 6 month course and an extended course.13
In contrast to some studies,4,13,14 but in agreement with San Francisco study,5 we found no difference in the outcome of the two groups patients. This is in agreement with a previous report that INH mono-resistant treatment regimens have success rates of 95% or more.15
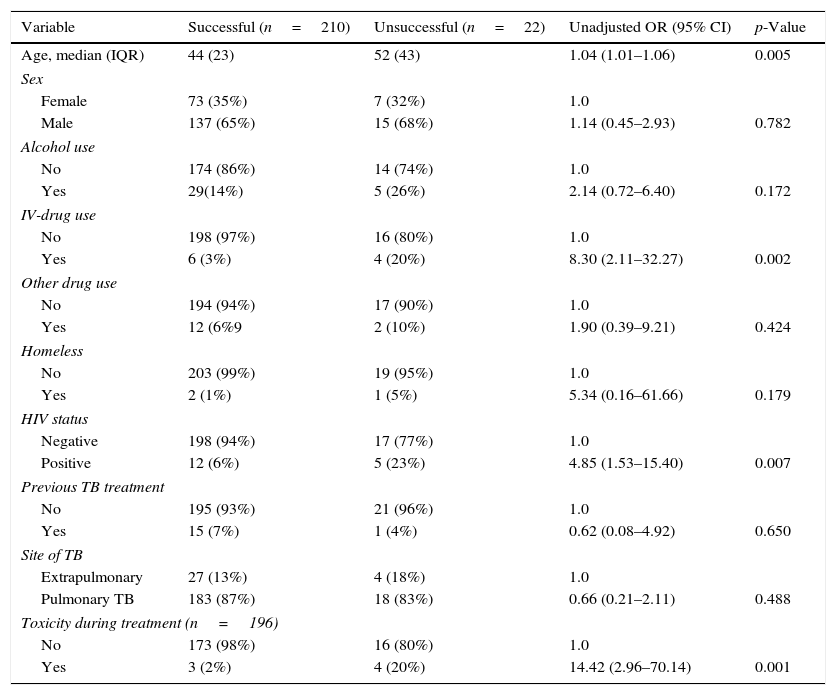
The treatment outcome of 232 INH mono-resistant cases were also analyzed (Table 2). Following the exclusion criteria, 10 of the INH mono-resistant cases without treatment results were excluded. The profile of these patients was similar to studied cases: median age was 46 years, 8 (80%) were male, 1 (10%) was HIV-positive, and 1 (12.5%) was an IV-drug user.
Univariate analysis of the risk (odds ratio, OR) for unsuccessful treatment of TB patients with INH mono-resistant disease.
| Variable | Successful (n=210) | Unsuccessful (n=22) | Unadjusted OR (95% CI) | p-Value |
|---|---|---|---|---|
| Age, median (IQR) | 44 (23) | 52 (43) | 1.04 (1.01–1.06) | 0.005 |
| Sex | ||||
| Female | 73 (35%) | 7 (32%) | 1.0 | |
| Male | 137 (65%) | 15 (68%) | 1.14 (0.45–2.93) | 0.782 |
| Alcohol use | ||||
| No | 174 (86%) | 14 (74%) | 1.0 | |
| Yes | 29(14%) | 5 (26%) | 2.14 (0.72–6.40) | 0.172 |
| IV-drug use | ||||
| No | 198 (97%) | 16 (80%) | 1.0 | |
| Yes | 6 (3%) | 4 (20%) | 8.30 (2.11–32.27) | 0.002 |
| Other drug use | ||||
| No | 194 (94%) | 17 (90%) | 1.0 | |
| Yes | 12 (6%9 | 2 (10%) | 1.90 (0.39–9.21) | 0.424 |
| Homeless | ||||
| No | 203 (99%) | 19 (95%) | 1.0 | |
| Yes | 2 (1%) | 1 (5%) | 5.34 (0.16–61.66) | 0.179 |
| HIV status | ||||
| Negative | 198 (94%) | 17 (77%) | 1.0 | |
| Positive | 12 (6%) | 5 (23%) | 4.85 (1.53–15.40) | 0.007 |
| Previous TB treatment | ||||
| No | 195 (93%) | 21 (96%) | 1.0 | |
| Yes | 15 (7%) | 1 (4%) | 0.62 (0.08–4.92) | 0.650 |
| Site of TB | ||||
| Extrapulmonary | 27 (13%) | 4 (18%) | 1.0 | |
| Pulmonary TB | 183 (87%) | 18 (83%) | 0.66 (0.21–2.11) | 0.488 |
| Toxicity during treatment (n=196) | ||||
| No | 173 (98%) | 16 (80%) | 1.0 | |
| Yes | 3 (2%) | 4 (20%) | 14.42 (2.96–70.14) | 0.001 |
TB: tuberculosis; INH: isoniazid; IQR: interquartile range; IV: intravenous; HIV: human immunodeficiency virus; TB: tuberculosis.
A univariate analysis showed that age (OR=1.04; 95%CI=1.01–1.06; p=0.005), intravenous (IV)-drug use (OR=8.30; 95%CI=2.11–32.27; p=0.002), HIV-positivity (OR=4.85; 95%CI=1.53–15.40; p=0.007) and toxicity during treatment (OR=14.42; 95%CI=2.96–70.14; p=0.001) were significantly associated with unsuccessful treatment outcome.
The Georgia study4 reported treatment outcomes were worse for older patients among those who had INH mono-resistance. The Tanzania study14 reported similar results, and also that HIV-infected individuals were more likely to experience unsuccessful treatment. The increased toxicity during treatment may be related to longer treatment duration, as described in San Francisco study.5
The main limitation of our study was the low number of unsuccessful treatments (n=22), which made multivariate analysis unfeasible.
Our findings reinforce the need for susceptibility testing and monitoring the treatment of INH mono-resistant cases, especially among the elderly, cases with HIV co-infection, IV drug users, and cases with toxicity during treatment.
Rita Gaio was partially supported by CMUP (UID/MAT/00144/2013), which is funded by FCT (Portugal) with national (MEC) and European structural funds (FEDER), under the partnership agreement PT2020.