Acute exacerbations (AE) of Interstitial Lung Diseases (ILD) are associated with poor outcomes, including in Hypersensitivity Pneumonitis (HP). Recent developments have been made on the characterization and standardization of the diagnosis of HP, with publication of new guidelines.1,2
Environmental exposure has a prominent role in ILD development and progression.1,2 Consequently, the identification of the inciting antigen related to HP in susceptible individuals is crucial and leads to better outcomes.3,4 There is little evidence regarding risk factors and outcomes in AE5 of HP (AE-HP).
A retrospective analysis of patients with HP diagnosis, older than 18 years-old, followed at our center between 2012 and 2017 was performed.
Patient demographics, medical history, smoking habits, occupational history, environmental antigen exposure assessed through a locally adapted questionnaire, pharmacological treatments, laboratory, imaging, pulmonary function and pathologic data were collected.
HP diagnosis was considered in case of compatible exposure, clinical features (dyspnea or cough, asthenia, fever, weight loss, and absence of an alternative diagnosis), typical chest high-resolution computed tomography (HRCT), and bronchoalveolar lavage (BAL) high lymphocytosis (>30%). Patients without definitive diagnosis after the previous diagnostic approach were submitted to transbronchial lung cryobiopsy and/or surgical lung biopsy. All diagnosis were asserted in Multidisciplinary Meeting. According with clinical condition and evolution the patients were evaluated with a 3–4 months appointment follow-up.
AE was defined as worsening of dyspnea within a 30-day period with new bilateral lung infiltration and no evidence of other causes of dyspnea, leading to hospitalization.7
Baseline descriptive statistics were calculated and reported as mean±sd or percentages. Student's t-test or Mann–Whitney test were used for continuous variables, while chi-squared test and Fisher's exact test were used to compare categorical variables. Multivariable logistic regression modeling was conducted using the independent variables identified as being statistically and clinically significant from the univariate analysis. All p-values were two-tailed, and p-values<0.05 were considered statistically significant. This study had the approval of local Ethics Committee.
A total of 102 patients with fibrotic HP were included, with median age of 69 years (min 34–max 89); 55.9% (n=57) were female and 64.3%(n=63) were never-smokers. Regarding the radiologic pattern, 60.2% (n=62) patients had Usual Interstitial Pneumonia “like” radiological pattern on HRCT.
Exposure to birds was reported by 74.5% (n=76) and to molds in the home or occupational environment by 32.3% (n=31). A total of 22 patients had multiple exposures and 17 were cryptogenic (no reported exposure).
Immunosuppressive treatment was prescribed in 87.3% (n=89) patients.
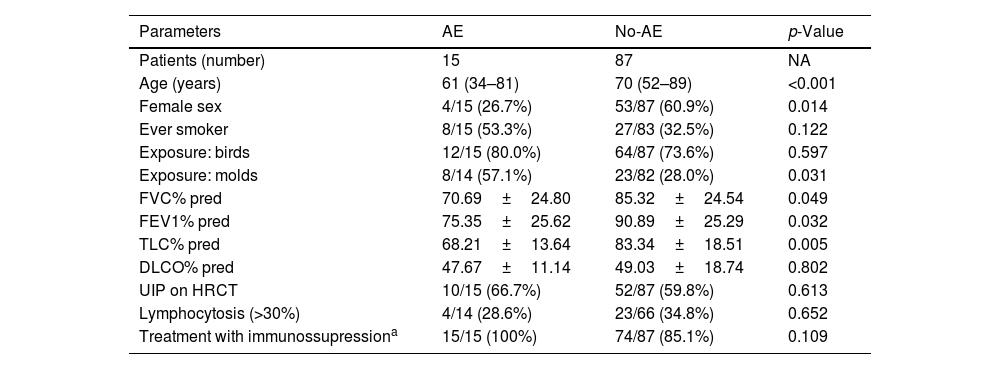
Fifteen patients (14.7%) developed AE during a median follow-up of 36 months. Patients with AE were younger (61 vs 70 years, p<0.01), had lower FVC % predicted (70.7±24.8 vs 85.3±24.5, p=0.049), lower FEV1% predicted (75.4±25.6 vs 90.9±25.3, p=0.032) and lower TLC% predicted (68.2±13.6 vs 83.3±18.5, p=0.005) at the time of diagnosis. For additional information on the distinct characterization of patients with and without AE see Table 1.
Initial characterization of fibrotic HP patients according to the development of AE.
| Parameters | AE | No-AE | p-Value |
|---|---|---|---|
| Patients (number) | 15 | 87 | NA |
| Age (years) | 61 (34–81) | 70 (52–89) | <0.001 |
| Female sex | 4/15 (26.7%) | 53/87 (60.9%) | 0.014 |
| Ever smoker | 8/15 (53.3%) | 27/83 (32.5%) | 0.122 |
| Exposure: birds | 12/15 (80.0%) | 64/87 (73.6%) | 0.597 |
| Exposure: molds | 8/14 (57.1%) | 23/82 (28.0%) | 0.031 |
| FVC% pred | 70.69±24.80 | 85.32±24.54 | 0.049 |
| FEV1% pred | 75.35±25.62 | 90.89±25.29 | 0.032 |
| TLC% pred | 68.21±13.64 | 83.34±18.51 | 0.005 |
| DLCO% pred | 47.67±11.14 | 49.03±18.74 | 0.802 |
| UIP on HRCT | 10/15 (66.7%) | 52/87 (59.8%) | 0.613 |
| Lymphocytosis (>30%) | 4/14 (28.6%) | 23/66 (34.8%) | 0.652 |
| Treatment with immunossupressiona | 15/15 (100%) | 74/87 (85.1%) | 0.109 |
Data are presented as mean±standard deviation, median [minimum–maximum], or n (%).
In the overall HP population, lower age (OR=0.89; 95% CI, 0.82–0.95), FVC% predicted (OR=0.98; 95% CI, 0.95–1.00), FEV1% predicted (OR=0.97; 95% CI, 0.95–1.00), TLC% predicted (OR=0.95; 95% CI, 0.92–0.99), and exposure to molds (OR=3.42; 95% CI, 1.07–10.94) were risk factors for development of AE in a univariate analysis (p<0.05).
Patients who reported exposure to molds had a higher rate of AE (25.8% vs 9.4%), X2 (1, N=95)=4.49; p=0.034.
After a multiple logistic regression, exposure to molds remained a significant independent risk factor for AE even after adjusting for FVC% predicted, TLC% predicted and age (OR=5.06; 95% CI, 1.09–23.53; p=0.039).
Regarding exacerbation outcomes, 2 patients were submitted to invasive mechanical ventilation and one of those to extra-corporeal membrane oxygenation. A total of 8 patients died during the exacerbation admission (53.3%). From the remaining patients, 2 died within 2 and 50 months after the AE. One patient underwent a successful lung transplantation. The remaining 4 are on follow-up.
Acute exacerbations of ILDs remain a clinical challenge with high mortality and poor outcomes.6,7 Its incidence in IPF (AE-IPF) varies between 7 and 32%, with a mortality of about 50% and median survival of 3–4 months after AE-IPF.7 Our results in AE-HP, an entity with less evidence reported, are in line with previous reports, showing similar outcomes to AE-IPF.5–8
Primarily, it is important to state that all patients included had Fibrotic HP (which comprised 84.9% of all patients with HP at our center). A total of 60% of the patients had a UIP “like” radiological pattern, resembling results from a prospective trial with similar number of patients.9 This entity is now well characterized in the recent guidelines.1,2 Mechanisms associated with fibrosis and subsequent clinical and functional decline may have similarities with IPF and other PF-ILDs, despite a different underlying pathophysiology.8,10
The risk factors identified in this cohort related to impaired lung function (FVC% predicted, FEV1% predicted, TLC% predicted) are consistent with previous reports in fibrotic ILDs.5,6 Other potential associations reported in literature include Diffusing Capacity for Carbon Monoxide and PaO2 and UIP pattern.5 In our sample, UIP pattern was present in 60.8% of all patients, however it was not a risk factor for AE. A thorough characterization regarding radiology is needed in this context, namely fibrosis extension/severity or other signs of progression. Lower age was a risk factor, with a previous publication showing similar results in AE-IPF.11
Despite widely considered idiopathic, there are triggers to AE according to a working group definition6 such as infection, micro-aspiration or lung biopsy. Therefore, it is not fully understood whether AE represents an intrinsic deterioration of the underlying ILD or a response to an external stimulus. Given the distinct nature of HP, antigens could potentially be included in the “triggers”, as hypothesized here. It is known that identifying and removing the causal antigen is related to better survival in HP.3
In our sample, a questionnaire was applied to the patient and therefore the results are based on patient-reported exposure. Besides, the exposure reported may not necessarily indicate causality of HP. An evaluation from an environmental hygienist would probably increase the characterization of exposure in these patients. There is also no data regarding re-exposure.
Exposure to molds but not birds was associated with AE development. We hypothesized that patients with fungal, or other exposure difficult to identify and remove, have an increased risk to continuous exposure. This could explain the worst outcome, namely an AE increased risk, in this subset of patients (in which exposure cessation is more difficult). Despite previous reports referencing positive history of exposure in the characterization of AE-HP,5 this is the first report analyzing AE-HP occurrence in relation to the type of antigenic exposure.
In conclusion, our findings reveal that a lower age, declined lung function, and exposure to molds were factors associated with the AE occurrence. A better recognition of the risk factors may enable strategies for early recognition and management of AE in patients with HP. A methodologic approach may enable identification of the exposure etiologic factor and can elucidate on strategies to avoid exacerbations.
Funding sourceNone.
Conflicts of interestThe authors declare no conflicts of interest.