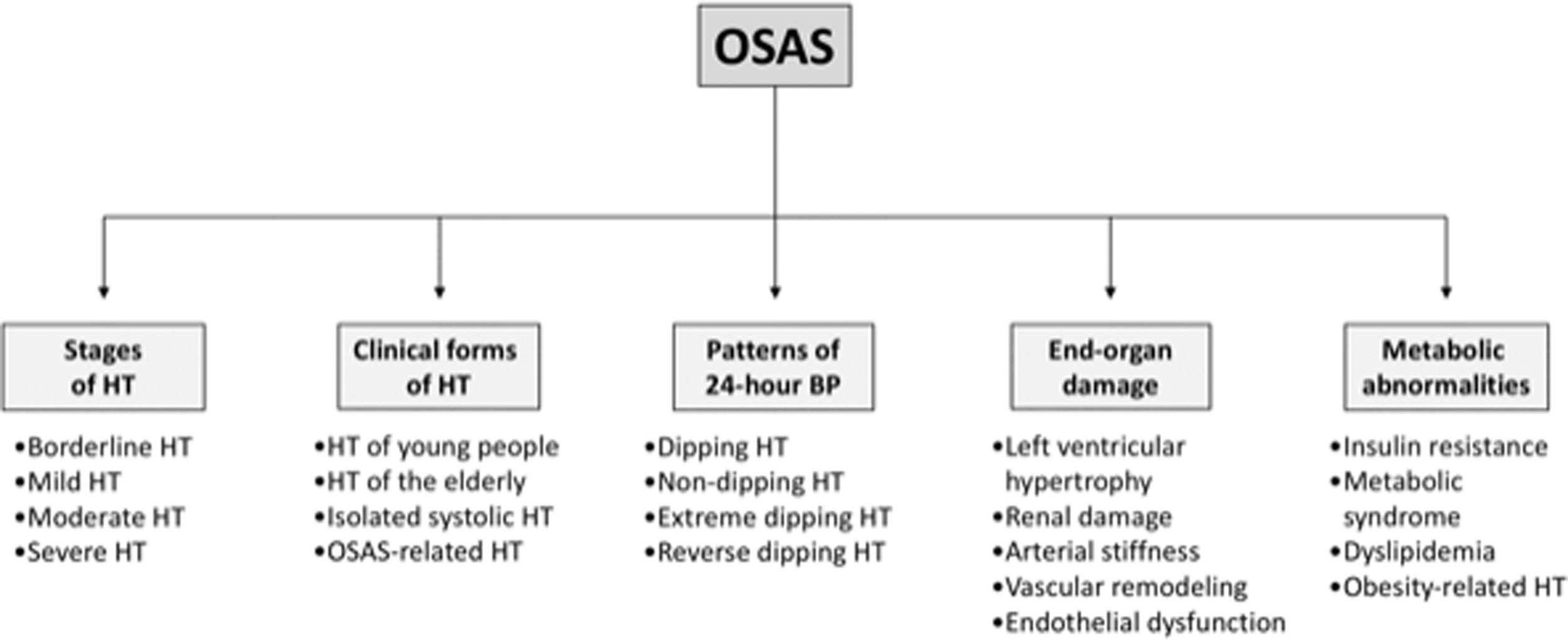
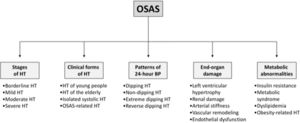
Obstructive Sleep Apnea Syndrome (OSAS) is characterized by repetitive upper-airway obstructions during sleep, leading to intermittent hypoxia, increased carbon dioxide concentrations, marked autonomic neural fluctuations and intrathoracic pressure alterations, leading to an increased risk of cardiovascular disease and all cause death, including sudden death, particularly during the nighttime period. This editorial will be focused on a cardiologist's view point on the relationships between OSAS and hypertension, affecting the stages of hypertension, the different hypertensive clinical phenotypes, the various patterns of 24-h blood pressure profile, the hypertension-related target organ damage and the metabolic abnormalities frequently detected in the high blood pressure state (Fig. 1). Emphasis will be given to three main aspects, i.e. (1) the epidemiological relevance of the association between high blood pressure and OSAS and its potential mechanisms, (2) the role of OSAS in initiating and favoring the progression of organ damage and (3) the impact of continuous positive airway pressure (CPAP) on the above mentioned alterations.
The association between OSAS and hypertension has been investigated since several years, taking into account potential confounders such as obesity, age and gender. One of the first cross-sectional population-based studies evaluating the association between polysomnographic apnoea–hypopnea Index (AHI) and daytime hypertension was performed in large groups of Spanish patients while controlling for multiple potential confounding variables.1 Even among subjects with an AHI of less than 5 events per hour, those with AHI greater than zero had increased odds of hypertension (odds ratio 2.5; 95% CI 1.1–5.8) when compared to those with an AHI of zero. Similar results were obtained in the Sleep Heart Health Study in which the participants were 6132 of both gender.2 Age does not seem to be a variable capable to affect the relationship between OSAS and hypertension given the similar results obtained in younger (and less obese) participants in other population-based studies. The main findings showing the participation of OSAS at the development of hypertension come from the Wisconsin Sleep Cohort study.3 In this prospective investigation, even minimally elevated AHI values at baseline were independently associated with a 3-fold increased risk of developing hypertension over a 4-year follow-up period. Overall the results of these studies are consistent with those seen in other studies with different study designs recently included in a well structured meta-analysis.4 A high prevalence of OSAS is particularly evident in patients with drug-resistant hypertension suggesting that OSA might be one of the most important causes of refractory hypertension.5,6 The important role of OSAS as a determinant of high blood pressure levels has been further emphasized by recent European Society of Cardiology/European Society of Hypertension (ESC/ESH) guidelines, which also underline the improvement in hypertension management after correction of OSAS.7 As it will be briefly mentioned in the last part of this editorial, the best evidence for a causal role of OSA in the pathogenesis of hypertension has been obtained in humans by prospective randomized trials on the effects of CPAP therapy of OSA,8,9 demonstrating that nocturnal treatment of OSA is accompanied by lower daytime blood pressure.
Several mechanisms have been advanced for explaining the pro-hypertensive effects of OSAS. These include an endothelial dysfunction, an activation of inflammatory mediators, the occurrence of an oxidative stress reaction and a sympathetic overactivity coupled with a pronounced baroreflex impairment. The latter mechanism has been investigated in recent years in studies directly evaluating sympathetic nerve traffic via the microneurographic technique, which allows to overcome the limitations of other approaches used throughout the years to assess human sympathetic function, such as the assay of the circulating plasma venous levels of the neuroadrenergic transmitter norepinephrine and the power spectral analysis of the heart rate signal. The main results of these studies emphasize the central role of OSAS in favoring the sympathetic activation in patients with essential hypertension, metabolic syndrome and an obese state.10 These conclusions have been strengthened by the results of a recent meta-analysis,11 showing in addition that the sympathetic overdrive related to OSA (1) takes place in hypertension in absence of other comorbidities known to be characterized by an adrenergic activation and (2) is detectable not only in severe but also in mild and moderate OSA, a finding suggesting the early occurrence of the sympathetic overactivity in the clinical course of the disease.
A further intriguing aspect of the association between OSA and hypertension refers to the finding, confirmed by a recent meta-analysis,12 that about 1/4 of OSA patients display left ventricular hypertrophy and that this target organ alteration (1) is detectable even in absence of an obese state and (2) is potentiated by the presence of hypertension and (3) can be at least in part reversed by CPAP. Other end-organ damage alterations detectable in OSA include13 (1) left atrial enlargement, (2) right ventricular remodeling, (3) increased carotid intima-media thickness and development of carotid plaques, (4) large artery stiffening, (5) retinal microvascular alterations and (6) occurrence of microalbuminuria. Of particular relevance is the observation that many of the above mentioned cardiovascular alterations (which may take place concomitantly with metabolic abnormalities affecting glucose and insulin profile) can be favorably affected by CPAP and that they frequently are reported even when the presence of other comorbidities, including hypertension, is ruled out. This means that OSAS “per se” may favor the occurrence of target organ damage and that the presence of elevated blood pressure values acts as a “reinforcing element” of the adverse cardiovascular effects of OSAS.
As already mentioned, many of the pathophysiological alterations characterizing OSAS are favorably affected (and for some variables reversed and even normalized) by CPAP treatment.8,9 This is the case for the endothelial dysfunction and arterial stiffness alterations. This is also the case for the sympathetic overdrive and the concomitant baroreflex impairment.14,15 As a consequence, blood pressure values undergo some reduction, as also documented via the gold standard approach to assess the blood pressure lowering effects of a given therapeutic intervention, namely 24-h ambulatory blood pressure monitoring.8,9
In conclusion, from a cardiologist's point of view OSAS investigation appears to be a successful area of research work which has allowed throughout the years to translate pathophysiological information derived from experimental and clinical investigations in important therapeutic achievements useful to obtain significant cardiovascular risk reduction of the treated patient. Areas of controversy worthy of future investigations include the very long-term effects of C-PAP on OSAS and blood pressure, particularly in resistant hypertension, the impact of renal denervation on OSAS patients, considering the promising recent subset of data from SIMPLICITY 3 trial and the possibility to improve blood pressure control in a consistent fraction of hypertensive patients affected by OSAS throughout non-pharmacologic interventions.
Conflict of interestNone.