To establish the diagnostic accuracy of pleural fluid (PF) CEA and CA 15-3 in identifying malignancy, and to determine the additional value of these markers in patients with malignant pleural effusions (MPEs) with false negative results from cytological fluid examination.
MethodsPF concentrations of CEA and/or CA 15-3 were determined in 1575 patients with non-purulent exudates, 549 of whom had confirmed MPEs, 284 probable MPEs, and 742 benign effusions. Tumor marker cut-off points were set to ensure 100% specificity for malignant effusion.
ResultsThe 41%, 40% and 60% of MPE patients had high PF levels of CEA (>45ng/mL), CA 15-3 (>77UI/l) or both, respectively. These percentages were 30%, 19% and 41% in MPEs with positive pleural biopsy and negative PF cytology; and 24%, 13% and 35% in clinical MPEs without histocytological confirmation. Tumor markers were of no value in lymphomas and mesotheliomas. The area-under-the-curve for CEA was 0.819 (95% CI: 0.793–0.845) and for CA 15-3, it was 0.822 (95% CI: 0.796–0.847). The use of tumor markers compared to cytology alone, increased the diagnosis of malignancy by 14%.
ConclusionsMeasurements of PF CEA and CA 15-3 may complement pleural cytology in the identification of MPEs.
Establecer la rentabilidad diagnóstica de la medición de CEA y CA 15-3 en el líquido pleural (LP) para identificar malignidad, así como el valor adicional de estos marcadores en pacientes con derrame pleural maligno (DPM) y citología pleural falsamente negativa.
MétodoSe determinaron las concentraciones de CEA o CA 15-3 en el LP de 1.575 pacientes con exudados no purulentos, de los que 549 tenían DPM demostrados, 284 derrames probablemente malignos y 742 derrames benignos. Se buscaron puntos de corte 100% específicos para dichos marcadores, de forma que no pudieran ser superados por ningún derrame benigno.
ResultadosEl 41%, 40% y el 60% de los pacientes con DPM tenían concentraciones pleurales elevadas de CEA (>45ng/mL), CA 15-3 (>77UI/l), o de alguno de los anteriores, respectivamente. Estos porcentajes fueron del 30%, 19% y 41% en los DPM con biopsia pleural positiva y estudios citológicos del LP negativos; y del 24%, 13% y 35% en los derrames considerados clínicamente malignos, pero sin demostración citohistológica. Los marcadores tumorales no tuvieron utilidad en linfomas ni mesoteliomas. El área bajo la curva de eficacia diagnóstica (AUC) del CEA fue de 0,819 (IC 95%: 0,793–0,845) y la del CA 15-3 de 0,822 (IC 95%: 0,796–0,847). Globalmente, el uso adicional de los marcadores tumorales incrementó el diagnóstico de malignidad un 14% respecto a la citología pleural de forma aislada.
ConclusionesLa determinación de CEA y CA 15-3 en LP puede complementar a la citología pleural en la identificación de los DPM.
The easiest way of diagnosing malignant pleural effusion (MPE) is to demonstrate the presence of tumor cells in the cytological examination of pleural fluid (PF).1 Unfortunately, pleural cytology has a maximum yield of around 60%, and figures are much lower in some tumor types, such as squamous cell lung cancer or mesothelioma (25%–30%).2 For this reason, extensive research has been conducted into obtaining a diagnosis of malignancy in patients with false negative results on pleural cytology, without the need to resort to invasive procedures such as biopsy by thoracoscopy. In this respect, measuring the concentrations of certain conventional tumor markers in pleural fluid is an attractive option, as this specimen is easy to obtain in clinical practice. However, there is no consensus on the most useful tumor markers and the most appropriate cutoff points to use.
Carcinoembryonic antigen (CEA) has been by far the most widely studied marker in PF, followed by others such as CYFRA 21-1 and carbohydrate antigen 15-3 (CA 15-3). Although evidence has shown that these markers can offer valuable information in some situations, neither the British3 nor the Spanish1 guidelines recommend their routine use in PF.
This study reports the largest single-center series published to date on the potential utility of determining CEA and CA 15-3 in PF for identifying malignancy in the context of non-purulent exudate. This validates our previous experience4 and shows the added value of determining these tumor markers in addition to pleural cytology.
Materials and MethodsA retrospective review was conducted of all consecutive patients with pleural effusion referred by their treating physician to the Pleural Medicine Department of our hospital between August 2003 and August 2016 to undergo diagnostic thoracentesis with determination of CEA or CA 15-3 in the PF. The study was approved by the local ethics committee (CEIC-1713).
Diagnostic Variables and CriteriaDemographic data, results of the PF analysis, pathology studies and the final diagnosis of each patient were collected. Pleural effusion was defined as malignant if malignant cells were found in PF or in a pleural biopsy. Probably malignant pleural effusion (PMPE) was considered if 2 of the following conditions were met: (a) evidence of a primary tumor or extrapleural metastases, and (b) existence of a pleural exudate with negative cytological studies, not explained by causes other than tumor invasion of pleura, after performance of the clinical tests considered relevant by the treating physician and a sufficient follow-up period. Histological confirmation was always required for the diagnosis of pleural mesothelioma. Generally accepted criteria were used for diagnosing benign pleural effusion (BPE).5
Measuring Tumor Markers in Pleural FluidCEA and CA 15-3 in PF were measured in an Electsys Roche electrochemiluminescence analyzer with a commercially available kit (Roche Diagnostics, Mannheim, Germany), according to the instructions of the manufacturer. Analyses were performed within hours of extraction of the PF specimen. In our laboratory, normal serum values are 0.2–5ng/mL for CEA, and 1–30IU/mL for CA 15-3.
Statistical AnalysisData were expressed as number and percentage with their corresponding 95% confidence intervals (CI), or a median and 25%–75% quartiles. An analysis of the diagnostic efficacy curves (ROC) determined the yield of the tumor markers for identifying malignancy, seeking cutoff points with 100% specificity (i.e., no BPE above this threshold); these calculations were obtained excluding PMPE. Qualitative and quantitative variables between patients with MPE, PMPE, and BPE were compared using the Kruskal–Wallis and chi-squared tests, respectively. The frequency of elevated tumor markers among the different tumor types was compared using the chi-squared test with subsequent analysis of standardized residuals. Sensitivity and odds ratios were calculated using a 2×2 table. Calculations were performed using the SPSS statistical package, version 22.0.
ResultsStudy PopulationDuring the study period, 2580 patients referred for CEA or CA 15-3 determinations in PF were identified. After exclusion of 191 empyemas (gross pus) and 814 individuals who met Light's criteria for transudate,6 the study population consisted of 1575 patients with non-purulent pleural exudates. Of these, 549 had cytologically demonstrated MPE, 284 had PMPE, and 742 had BPE (Table 1).
Causes of Pleural Exudate Among the Study Population.
| Etiology | No. (%) |
|---|---|
| Malignant pleural effusion | 549 (35) |
| Lung | 214a |
| Breast | 96 |
| Unknown origin | 57 |
| Ovarian | 45 |
| Gastrointestinal | 43 |
| Lymphoma | 34 |
| Mesothelioma | 17 |
| Miscellaneous | 43 |
| Probably malignant pleural effusion | 284 (18) |
| Lung | 125b |
| Gastrointestinal | 42 |
| Breast | 33 |
| Unknown origin | 16 |
| Lymphoma | 14 |
| Ovarian | 11 |
| Miscellaneous | 43 |
| Benign pleural effusion | 742 (47) |
| Pneumonia | 177 |
| Heart failurec | 129 |
| Tuberculosis | 117 |
| Pericardial diseases | 66 |
| Idiopathic | 62 |
| Post-heart, chest or abdominal surgery | 55 |
| Pulmonary embolism | 29 |
| Trauma | 25 |
| Connective tissue diseases | 10 |
| Miscellaneous | 72 |
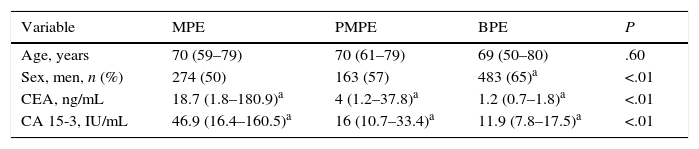
Median concentrations of CEA and CA 15-3 in PE were significantly higher in MPE than in the other groups, and also significantly higher in PMPE than in BPE (Table 2).
Demographic Characteristics and Pleural Concentrations of Tumor Markers Among the Study Groups.
| Variable | MPE | PMPE | BPE | P |
|---|---|---|---|---|
| Age, years | 70 (59–79) | 70 (61–79) | 69 (50–80) | .60 |
| Sex, men, n (%) | 274 (50) | 163 (57) | 483 (65)a | <.01 |
| CEA, ng/mL | 18.7 (1.8–180.9)a | 4 (1.2–37.8)a | 1.2 (0.7–1.8)a | <.01 |
| CA 15-3, IU/mL | 46.9 (16.4–160.5)a | 16 (10.7–33.4)a | 11.9 (7.8–17.5)a | <.01 |
Data are expressed as median (quartiles) or number (percentage), as applicable.
BPE, benign pleural effusion; MPE, malignant pleural effusion; PMPE, probably malignant pleural effusion.
ROC analysis of the MPE and BPE populations established pleural concentrations of CEA>45ng/mL and CA 15-3>77IU/mL as 100% specific cutoff points. In 41% (95% CI: 37–45) and 40% (95% CI: 36–44) of subjects with MPE, respectively, levels of these markers were above these limits. Moreover, 60% (95% CI: 56–64) of MPE patients had elevated levels of either of the 2 markers. Although elevated markers in PF was a definitive factor for confirming MPE (infinite positive probability ratios), their absence did not substantially modify the probability of cancer (negative probability ratios of 0.59 and 0.60 for CEA and CA 15-3, respectively, implying an absolute reduction in probability of only 15% approximately). Areas under the curve (AUC), as a discriminatory measure of efficacy, were 0.819 (95% CI: 0.793–0.845) for CEA and 0.822 (95% CI: 0.796–0.847) for CA 15-3.
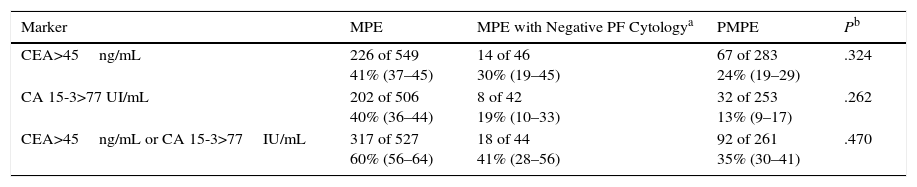
Tumor Markers in Malignant Effusions with Negative Cytological StudiesIn the subgroup of patients positive for MPE by pleural biopsy but negative by PF cytology, CEA, CA 15-3, or a combination of either CEA or CA 15-3 (where a result was considered positive if either of the 2 markers was higher than the diagnostic cutoff) correctly predicted malignant PE in 30%, 19% and 41% of cases, respectively. These percentages were not significantly different in the population of patients with PMPE who, by definition also had negative results from PF cytology studies (24%, 13% and 25%, respectively) (Table 3).
Sensitivity of Pleural CEA and CA 15-3 in Different Patient Groups with Malignant Effusion.
| Marker | MPE | MPE with Negative PF Cytologya | PMPE | Pb |
|---|---|---|---|---|
| CEA>45ng/mL | 226 of 549 41% (37–45) | 14 of 46 30% (19–45) | 67 of 283 24% (19–29) | .324 |
| CA 15-3>77 UI/mL | 202 of 506 40% (36–44) | 8 of 42 19% (10–33) | 32 of 253 13% (9–17) | .262 |
| CEA>45ng/mL or CA 15-3>77IU/mL | 317 of 527 60% (56–64) | 18 of 44 41% (28–56) | 92 of 261 35% (30–41) | .470 |
Data are expressed as absolute values and corresponding percentages, with 95% confidence intervals.
MPE, malignant pleural effusion; PF, pleural fluid; PMPE, probably malignant pleural effusion.
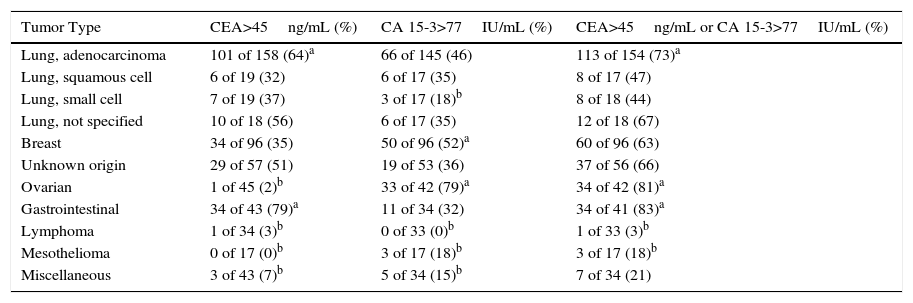
The yield of tumor markers in PF varied depending on the primary tumor (Table 4). The diagnostic sensitivity of CA 15-3 in ovarian tumors (79%), and of a combination of either CEA or CA 15-3 in gastrointestinal (83%), ovarian (81%), and lung (73%) adenocarcinomas was particularly notable. In contrast, the utility of both markers combined was practically zero in lymphomas and mesotheliomas (3% and 18%, respectively).
Sensitivity of Pleural Fluid CEA and CA 15-3 in Malignant Effusions, by Primary Tumor Type.
| Tumor Type | CEA>45ng/mL (%) | CA 15-3>77IU/mL (%) | CEA>45ng/mL or CA 15-3>77IU/mL (%) |
|---|---|---|---|
| Lung, adenocarcinoma | 101 of 158 (64)a | 66 of 145 (46) | 113 of 154 (73)a |
| Lung, squamous cell | 6 of 19 (32) | 6 of 17 (35) | 8 of 17 (47) |
| Lung, small cell | 7 of 19 (37) | 3 of 17 (18)b | 8 of 18 (44) |
| Lung, not specified | 10 of 18 (56) | 6 of 17 (35) | 12 of 18 (67) |
| Breast | 34 of 96 (35) | 50 of 96 (52)a | 60 of 96 (63) |
| Unknown origin | 29 of 57 (51) | 19 of 53 (36) | 37 of 56 (66) |
| Ovarian | 1 of 45 (2)b | 33 of 42 (79)a | 34 of 42 (81)a |
| Gastrointestinal | 34 of 43 (79)a | 11 of 34 (32) | 34 of 41 (83)a |
| Lymphoma | 1 of 34 (3)b | 0 of 33 (0)b | 1 of 33 (3)b |
| Mesothelioma | 0 of 17 (0)b | 3 of 17 (18)b | 3 of 17 (18)b |
| Miscellaneous | 3 of 43 (7)b | 5 of 34 (15)b | 7 of 34 (21) |
All P<.01
The overall sensitivity of PF cytology studies was 60% (95% CI: 57–64), while the sensitivity of a combination of either CEA or CA 15-3 was 49% (95% CI: 46–52). When the results of both tests (positive cytology or markers higher than the diagnostic cutoff) were taken into consideration, sensitivity for identifying the malignant nature of an effusion rose to 74% (95% CI: 71–77).
DiscussionThis study shows that the determination of CEA and CA 15-3 in PF can identify the tumor origin of over one third of MPE with negative cytology studies. These markers are more useful in the case of gastrointestinal, ovarian or lung adenocarcinomas that metastasize to the pleura, and negligible in the case of lymphomas and mesotheliomas.
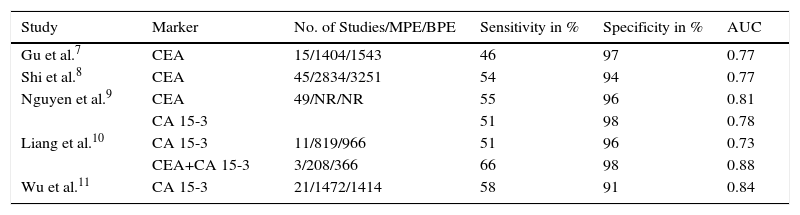
CEA has been the most widely studied tumor marker in PF. Three metaanalyses have been published on the potential utility of these biomarkers in diagnostics, comprising 15, 45, and 49 studies each.7–9 The results, along with those from another 3 metaanalyses on the use of pleural CA 15-3,9–11 are summarized in Table 5. Recently, a Chinese study of 601 patients with MPE and 538 with BPE found pleural CEA to have 69% sensitivity and 82% specificity, with an AUC of 0.740.12
Metaanalysis of Diagnostic Efficacy of Pleural CEA and CA 15-3 for Identifying Malignancy.
| Study | Marker | No. of Studies/MPE/BPE | Sensitivity in % | Specificity in % | AUC |
|---|---|---|---|---|---|
| Gu et al.7 | CEA | 15/1404/1543 | 46 | 97 | 0.77 |
| Shi et al.8 | CEA | 45/2834/3251 | 54 | 94 | 0.77 |
| Nguyen et al.9 | CEA | 49/NR/NR | 55 | 96 | 0.81 |
| CA 15-3 | 51 | 98 | 0.78 | ||
| Liang et al.10 | CA 15-3 | 11/819/966 | 51 | 96 | 0.73 |
| CEA+CA 15-3 | 3/208/366 | 66 | 98 | 0.88 | |
| Wu et al.11 | CA 15-3 | 21/1472/1414 | 58 | 91 | 0.84 |
AUC, area under the curve; BPE, benign pleural effusion; MPE, malignant pleural effusion; NR, not reported.
Taken together, the pooled sensitivities of these markers are very similar to those of PF cytology, but unlike the latter, they achieve absolute specificities with the (various) cutoff points proposed. This creates a margin of uncertainty in the interpretation of the CEA and CA 15-3 values that reduces their clinical applicability. Moreover, in previous studies, the BPE group has always included a considerable percentage of cardiac transudates and empyemas, effusions which, in practice, do not confound the differential diagnosis with MPE. These 2 limitations, the selection of tumor marker cutoffs that do not have absolute specificity and the inclusion of effusions with etiologies easily identifiable from a clinical point of view, have been resolved in this study. Nevertheless, the selection of 100% specific cutoffs reduces the sensitivity of any test; in this case, the percentage of patients with MPE who exceeded the established threshold for each individual tumor marker was around 40%. However, the combined use of either one or other of the 2 tumor markers raised sensitivity for detecting MPE up to 60%.
Finally, very few studies have provided data on the value of CEA and CA 15-3 in PMPE or MPE with negative cytology results.4,13,14 Indeed, this is the subset of patients in which measuring tumor markers in PE is of practical interest, since in patients with positive pleural cytologies, the diagnosis of malignancy is definitively established. In this respect, the behavior of the tumor markers was similar in MPE with negative pleural cytology and in PMPE: a combination of either CEA or CA 15-3 in PF led to 41% and 35% of these cases being classified as malignant, respectively.
The strengths of this study do not conceal its limitations. This was a retrospective evaluation performed in a single center, and the results must be validated in independent populations in other centers. Nevertheless, we have been able to confirm the data that we obtained from a previous study conducted previously,4 and this, in a way, can be seen as a validation. Another potential limitation is the possibility that a “paramalignant” effusion (i.e., effusion not directly related with the tumor invading the pleura of a cancer patient) might have been erroneously included in the PMPE group. Although the clinical definition of PMPE was strict in order to avoid this occurrence, this patient group did not undergo a confirmatory pleuroscopy. In any case, similarities in the behavior of PF tumor markers in the MPE group with negative cytology and the PMPE group suggests that the classification of the latter was largely correct.
In conclusion, the determination of CEA and CA 15-3 in PF using 100% specific cutoffs contributes valuable clinical information by increasing our ability to identify MPE, notwithstanding the need for cytohistological confirmation in patients who may be candidates for receiving active oncological treatment.
Author ContributionsStudy conception and design: J.M. Porcel. Data collection and analysis: C. Civit, A. Esquerda, A. Salud, S. Bielsa. Data interpretation: J.M. Porcel, S. Bielsa. Manuscript draft: J.M. Porcel. Critical review and approval of the final version of the manuscript: J.M. Porcel, C. Civit, A. Esquerda, A. Salud, S. Bielsa.
Conflict of InterestThe authors state that they have no conflict of interests.
Please cite this article as: Porcel JM, Civit C, Esquerda A, Salud A, Bielsa S. Utilidad de la medición de CEA y CA 15-3 en los exudados pleurales no purulentos para diagnosticar malignidad: experiencia de un único centro. Arch Bronconeumol. 2017;53:427–431.