The combination of 2 long-acting bronchodilators, a β2-agonist (LABA), and an antimuscarinic agent (LAMA) in a single inhalation device, known as double bronchodilation,1 has changed therapeutic strategies in chronic obstructive pulmonary disease (COPD). Studies evaluating the safety and efficacy of this modality have placed it at the forefront of current COPD treatment recommendations, while inhaled corticosteroids (ICS) are reserved as an alternative combined with double bronchodilation as a second treatment step in group C exacerbators or the more severe group D patients.2 In this context, the new GOLD 2017 strategy recommends the use of ICS in patients receiving double bronchodilation who continue presenting exacerbations. Consequently, there is a need to combine various drugs and inhalation devices to combine a LABA, a LAMA and an ICS to construct the so-called triple therapy using two inhalation devices.
Before the introduction of double bronchodilation, only 2 methods of administering this triple combination of drugs was available: either 3 drugs had to be administered via 3 different devices, or else a fixed ICS/LABA combination had to be used with a LAMA in another inhaler. The introduction of double bronchodilation now allows another alternative: a fixed LABA/LAMA combination together with an ICS in another inhaler, raising questions as to which strategy is the most appropriate.
On one hand, according to the current GOLD 2017 treatment algorithm, patients who are candidates for triple therapy have already been receiving double bronchodilation. So, while the GOLD 2017 document does not specify how this triple therapy should be administered, it seems logical to assume that, according to the GOLD 2017 recommendations, double bronchodilation in 1 device could be continued while adding an ICS in a separate device. However, on the other hand,3 suggesting that the triple therapy combination should be approached by adding the LAMA to the fixed ICS/LABA combination. Moreover, the real situation is more complex and real-world prescriptions do not always strictly follow recommendations,4 complicating the issue even further.
Although the preference of both the patient and the doctor for any given inhaler is certainly important when deciding which triple therapy to use in each case, there is a pressing need for scientific evidence on which drug combination strategy (LABA/ICS+LAMA or LABA/LAMA+ICS) is the most appropriate for treating COPD. Unfortunately, no clinical trials have prospectively evaluated the efficacy and safety of both treatment strategies, leaving us unable to make an evidence-based decision.
To make this decisions we need to take an indirect approach. This indirect approach leads us to ask about the real benefits of administering 2 drugs in a single inhaler. With regard to double bronchodilation, the BEACON study evaluated the clinical efficacy of the indacaterol/glycopyrronium combination versus the administration of indacaterol and glycopyrronium in 2 different devices.5 The results clearly showed that there was no difference between administering 2 bronchodilators together in a single device or separately. Therefore, to determine the advantages of administering 2 bronchodilators in a single inhaler, we must look beyond the clinical efficacy of bronchodilators and consider other outcomes, such as lower cost of treatment, and better treatment adherence, which in turn improves clinical efficacy.6
Regarding LABA/ICS combinations, if we evaluate the clinical efficacy of administering them combined or separately, the message is similar. A recent study assessed the functional efficacy of an ICS and a LABA in a single device compared to the same treatment at the same dose administered in 2 different devices. The results also showed a similar efficacy for both treatment modalities.7
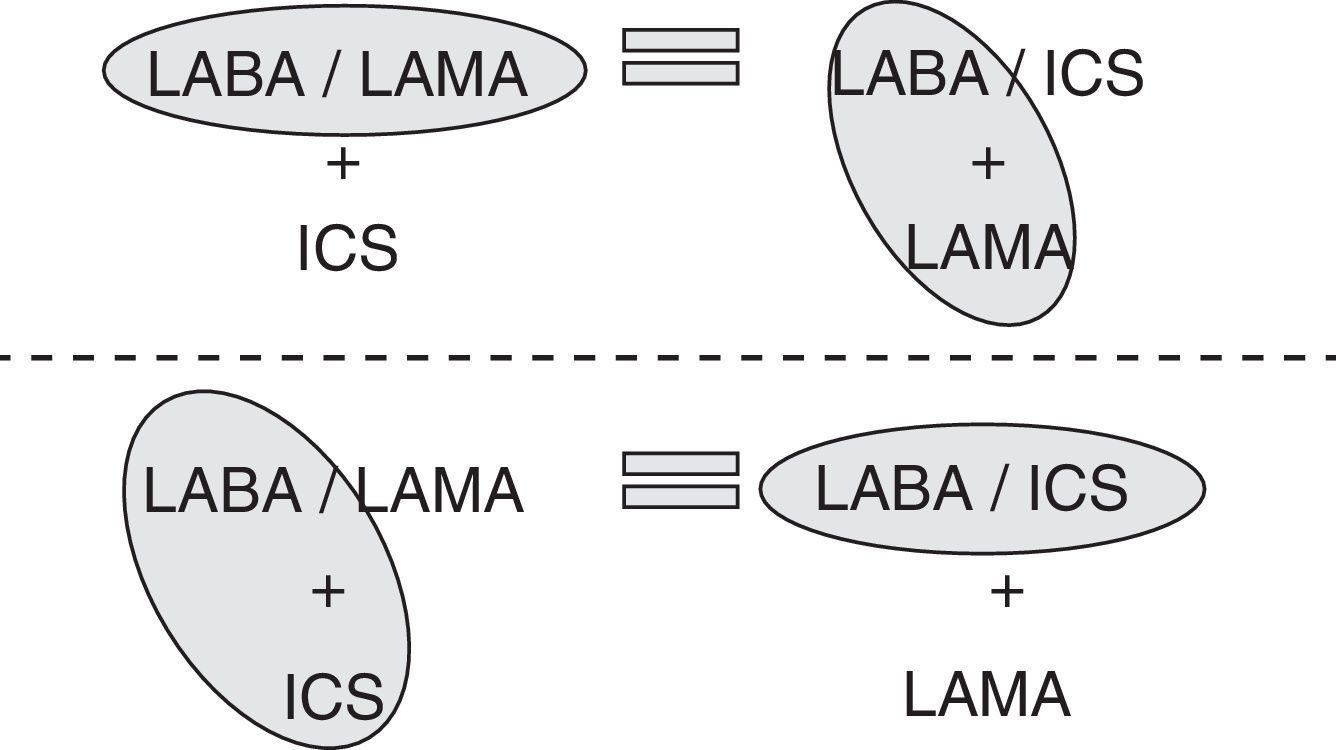
Accordingly, if giving 2 bronchodilators in different devices is the same as giving them separately, and giving an ICS/LABA in a single device is the same as giving them separately (Fig. 1), then both triple therapies (ICS/LABA+LAMA and LABA/LAMA+ICS) should be equally effective. This means that, strictly speaking in terms of efficacy, treating COPD patients with one or the other triple therapy should be the same.
However, it is important at this point to remember that in bronchial asthma, the combination ICS/LABA contributes significantly to improving patient control.8 In an inflammatory disease such as asthma, in which the essential treatment is the ICS, the combination of ICS/LABA improved control when they were administered together in the same device. However, the situation with COPD is different. COPD is an obstructive disease that is treated with bronchodilators. Indeed, the role of ICS in patients receiving LABA/LAMA is currently under debate.9 In COPD, then, we must guarantee appropriate administration of the bronchodilators, with a more controversial role for ICS.10 For this reason, due to the lack of clinical trials, in general terms, it seems more reasonable to use the combination of LABA/LAMA+ICS in COPD, while in asthma the ICS/LABA+LAMA option seems the most sensible.
The initial drawback is that, if once daily double bronchodilator therapies are used, there are currently no approved ICS not combined with an LABA for COPD with once-daily administration, which would have to be combined with administration of ICS every 12h. This scenario will probably change in the future when triple therapy is available in a single inhaler,11 since combined treatments have been shown to improve adherence in clinical practice, with the subsequent clinical and economic benefits.12
In summary, although no randomized clinical trials are available comparing both triple therapies, the results of previous studies seem to suggest that both forms of triple therapy must have a similar efficacy in COPD, bearing in mind that in COPD, it seems more reasonable to ensure double bronchodilation by administering it in a single inhaler and to add the ICS separately, thus permitting evaluation of the indication and the dose.
Please cite this article as: Lopez-Campos JL, Marquez-Martín E, Ortega-Ruiz F. Terapia triple vs tripe terapia en EPOC. Arch Bronconeumol. 2017;53:419–420.